
Concept explainers
(a)
Interpretation: The number of lone pairs in TeCl2 molecule needs to be determined.
Concept Introduction:
Lewis dot structure or electron dot structure can be defined as the structure of a molecule in which all the valence electrons and lone pairs on each atom is represented with the help of line and dot or cross.
The Lewis structure is used to predict the hybridization and molecular geometry of the molecule as it gives complete idea about the bonding between bonded atoms in a molecule.
(a)
Answer to Problem 6E
8 lone pairs; 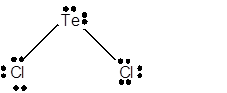
Explanation of Solution
The number of lone pairs in any molecule can be determined with the help of Lewis structure. In the Lewis structure of TeCl2 molecule;
Number of valence electrons in Te = 6 electrons
Number of valence electrons in Cl = 7
Total number of valence electrons = 6 + (2 x 7) = 20 electrons.
Thus the Lewis structure must be:
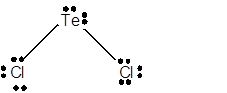
Hence in TeCl2 molecule there are 8 lone pairs.
(b)
Interpretation: The number of lone pairs in HI molecule needs to be determined.
Concept Introduction:
Lewis dot structure or electron dot structure can be defined as the structure of a molecule in which all the valence electrons and lone pairs on each atom is represented with the help of line and dot or cross.
The Lewis structure is used to predict the hybridization and molecular geometry of the molecule as it gives complete idea about the bonding between bonded atoms in a molecule.
(b)
Answer to Problem 6E
3 lone pairs; 
Explanation of Solution
The number of lone pairs in any molecule can be determined with the help of Lewis structure. In the Lewis structure of HI molecule;
Number of valence electrons in I = 7 electrons
Number of valence electrons in H= 1
Total number of valence electrons = 1 + (1 x 7) = 8 electrons.
Thus the Lewis structure must be:

Hence in HI molecule there are 3 lone pairs.
(c)
Interpretation: The number of lone pairs in AsBr3molecule needs to be determined.
Concept Introduction:
Lewis dot structure or electron dot structure can be defined as the structure of a molecule in which all the valence electrons and lone pairs on each atom is represented with the help of line and dot or cross.
The Lewis structure is used to predict the hybridization and molecular geometry of the molecule as it gives complete idea about the bonding between bonded atoms in a molecule.
(c)
Answer to Problem 6E
10 lone pairs; 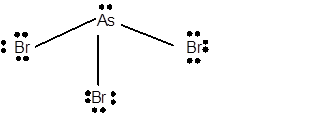
Explanation of Solution
The number of lone pairs in any molecule can be determined with the help of Lewis structure. In the Lewis structure of AsBr3 molecule;
Number of valence electrons in As = 5 electrons
Number of valence electrons in Br = 7
Total number of valence electrons = 5 + (3 x 7) = 26 electrons.
Thus the Lewis structure must be:
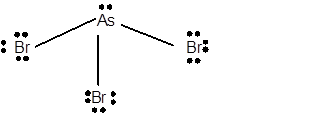
Hence in AsBr3 molecule there are 10 lone pairs.
(d)
Interpretation: The number of lone pairs in SiF4 molecule needs to be determined.
Concept Introduction:
Lewis dot structure or electron dot structure can be defined as the structure of a molecule in which all the valence electrons and lone pairs on each atom is represented with the help of line and dot or cross.
The Lewis structure is used to predict the hybridization and molecular geometry of the molecule as it gives complete idea about the bonding between bonded atoms in a molecule.
(d)
Answer to Problem 6E
12 lone pairs; 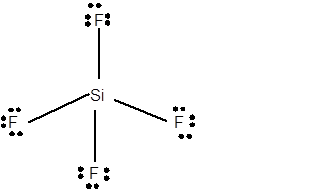
Explanation of Solution
The number of lone pairs in any molecule can be determined with the help of Lewis structure. In the Lewis structure of SiF4 molecule;
Number of valence electrons in Si = 4 electrons
Number of valence electrons in F = 7
Total number of valence electrons = 4 + (4 x 7) = 32 electrons.
Thus the Lewis structure must be:
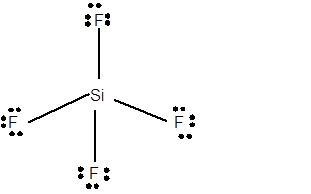
Hence in SiF4 moleculethere are 12 lone pairs.
(e)
Interpretation: The number of lone pairs in F2 molecule needs to be determined.
Concept Introduction:
Lewis dot structure or electron dot structure can be defined as the structure of a molecule in which all the valence electrons and lone pairs on each atom is represented with the help of line and dot or cross.
The Lewis structure is used to predict the hybridization and molecular geometry of the molecule as it gives complete idea about the bonding between bonded atoms in a molecule.
(e)
Answer to Problem 6E
6 lone pairs; 
Explanation of Solution
The number of lone pairs in any molecule can be determined with the help of Lewis structure. In the Lewis structure of F2 molecule;
Number of valence electrons in F = 7
Total number of valence electrons = (2x 7) = 14 electrons.
Thus the Lewis structure must be:

Hence in F2 molecule there are 6 lone pairs.
Chapter U2 Solutions
Living By Chemistry: First Edition Textbook
Additional Science Textbook Solutions
Campbell Biology (11th Edition)
Chemistry (7th Edition)
Chemistry: A Molecular Approach (4th Edition)
Introductory Chemistry (6th Edition)
Applications and Investigations in Earth Science (9th Edition)
Brock Biology of Microorganisms (15th Edition)
- 20. The Brusselator. This hypothetical system was first proposed by a group work- ing in Brussels [see Prigogine and Lefever (1968)] in connection with spatially nonuniform chemical patterns. Because certain steps involve trimolecular reac tions, it is not a model of any real chemical system but rather a prototype that has been studied extensively. The reaction steps are A-X. B+X-Y+D. 2X+ Y-3X, X-E. 305 It is assumed that concentrations of A, B, D, and E are kept artificially con stant so that only X and Y vary with time. (a) Show that if all rate constants are chosen appropriately, the equations de scribing a Brusselator are: dt A-(B+ 1)x + x²y, dy =Bx-x²y. diarrow_forwardProblem 3. Provide a mechanism for the following transformation: H₂SO A Me. Me Me Me Mearrow_forwardYou are trying to decide if there is a single reagent you can add that will make the following synthesis possible without any other major side products: xi 1. ☑ 2. H₂O хе i Draw the missing reagent X you think will make this synthesis work in the drawing area below. If there is no reagent that will make your desired product in good yield or without complications, just check the box under the drawing area and leave it blank. Click and drag to start drawing a structure. There is no reagent that will make this synthesis work without complications. : ☐ S ☐arrow_forward
- Predict the major products of this organic reaction: H OH 1. LiAlH4 2. H₂O ? Note: be sure you use dash and wedge bonds when necessary, for example to distinguish between major products with different stereochemistry. Click and drag to start drawing a structure. G C टेarrow_forwardFor each reaction below, decide if the first stable organic product that forms in solution will create a new C-C bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. NH2 CI MgCl ? Will the first product that forms in this reaction create a new CC bond? Yes No MgBr ? Will the first product that forms in this reaction create a new CC bond? Yes No G टेarrow_forwardFor each reaction below, decide if the first stable organic product that forms in solution will create a new CC bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. དྲ。 ✗MgBr ? O CI Will the first product that forms in this reaction create a new C-C bond? Yes No • ? Will the first product that forms in this reaction create a new CC bond? Yes No × : ☐ Xarrow_forward
- Predict the major products of this organic reaction: OH NaBH4 H ? CH3OH Note: be sure you use dash and wedge bonds when necessary, for example to distinguish between major products with different stereochemistry. Click and drag to start drawing a structure. ☐ : Sarrow_forwardPredict the major products of this organic reaction: 1. LIAIHA 2. H₂O ? Note: be sure you use dash and wedge bonds when necessary, for example to distinguish between major products with different stereochemistry. Click and drag to start drawing a structure. X : ☐arrow_forwardFor each reaction below, decide if the first stable organic product that forms in solution will create a new C - C bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. NH2 tu ? ? OH Will the first product that forms in this reaction create a new CC bond? Yes No Will the first product that forms in this reaction create a new CC bond? Yes No C $ ©arrow_forward
- As the lead product manager at OrganometALEKS Industries, you are trying to decide if the following reaction will make a molecule with a new C-C bond as its major product: 1. MgCl ? 2. H₂O* If this reaction will work, draw the major organic product or products you would expect in the drawing area below. If there's more than one major product, you can draw them in any arrangement you like. Be sure you use wedge and dash bonds if necessary, for example to distinguish between major products with different stereochemistry. If the major products of this reaction won't have a new CC bond, just check the box under the drawing area and leave it blank. Click and drag to start drawing a structure. This reaction will not make a product with a new CC bond. G marrow_forwardIncluding activity coefficients, find [Hg22+] in saturated Hg2Br2 in 0.00100 M NH4 Ksp Hg2Br2 = 5.6×10-23.arrow_forwardgive example for the following(by equation) a. Converting a water insoluble compound to a soluble one. b. Diazotization reaction form diazonium salt c. coupling reaction of a diazonium salt d. indacator properties of MO e. Diazotization ( diazonium salt of bromobenzene)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





