
Concept explainers
(a)
Interpretation :
Shape of ammonia molecule must be predicted.
Concept Introduction :
Shape of a molecule is determined by the total number of electron pairs in the valence shell of the central atom.
Ammonia molecule has total 4 atoms in which N is the central atom.
(a)
Answer to Problem C7.2RE
Shape of ammonia molecule is trigonal pyramidal.
Explanation of Solution
The structure of ammonia molecule is represented as follows:

N has total 4 pairs of electrons in the valence shell.
There are three bond pairs and lone pairs of electrons. N atom is sp3 hybridized and thus NH3 has trigonal pyramidal shape due to presence of one lone pair electrons.
b)
Interpretation :
Shape of silicon tetrachloride must be predicted.
Concept Introduction :
Shape of a molecule is determined by the total number of electron pairs in the valence shell of the central atom.
Silicon tetrachloride molecule has total 5 atoms in which Si is the central atom.
b)
Answer to Problem C7.2RE
Shape of silicon tetrachloride molecule is tetrahedral.
Explanation of Solution
The structure is represented as follows:
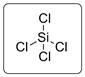
Si has total 4 pairs of electrons in the valence shell.
There are four bond pairs and no lone pairs of electrons. Si atom is sp3 hybridized and thus SiCl4 has tetrahedral shape.
c)
Interpretation :
Shape of hydrogen sulfide must be predicted.
Concept Introduction :
Shape of a molecule is determined by the total number of electron pairs in the valence shell of the central atom.
Hydrogen sulfide molecule has total 3 atoms in which S is the central atom.
c)
Answer to Problem C7.2RE
Shape of hydrogen molecule is angular.
Explanation of Solution
The structure of H2S is represented as follows:
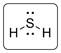
S has total 4 pairs of electrons in the valence shell.
There are two bond pairs and two lone pairs of electrons. S atom is sp3 hybridized and thus H2S has angular shape.
d)
Interpretation :
Shape of hydrogen cyanide must be predicted.
Concept Introduction :
Shape of a molecule is determined by the total number of electron pairs in the valence shell of the central atom.
Hydrogen cyanide molecule has total 3 atoms in which C is the central atom.
d)
Answer to Problem C7.2RE
Shape of hydrogen cyanide is linear.
Explanation of Solution
The structure is represented as follows:
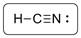
C has total 4 pairs of electrons in the valence shell.
There are four bond pairs and no lone pairs of electrons. C atom is triple bonded with N and carbon is sp hybridized and thus HCN has linear shape.
e)
Interpretation :
Shape of formaldehyde must be predicted.
Concept Introduction :
Shape of a molecule is determined by the total number of electron pairs in the valence shell of the central atom.
Formaldehyde molecule has total 4 atoms in which C is the central atom.
e)
Answer to Problem C7.2RE
Shape of formaldehyde is trigonal planar.
Explanation of Solution
The structure of formaldehyde is represented as follows:
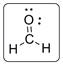
C has total 4 pairs of electrons in the valence shell.
There are four bond pairs and no lone pairs of electrons. C atom is double bonded with O and single bonded with two hydrogen atoms. Carbon is sp2 hybridized and thus CH2N has trigonal planar shape.
Chapter U2 Solutions
Living by Chemistry
Additional Science Textbook Solutions
Campbell Biology (11th Edition)
Introductory Chemistry (6th Edition)
Applications and Investigations in Earth Science (9th Edition)
Campbell Biology in Focus (2nd Edition)
Microbiology with Diseases by Body System (5th Edition)
Organic Chemistry (8th Edition)
- b. CH3 H3C CH3 CH3 H3C an unexpected product, containing a single 9- membered ring the expected product, containing two fused rings H3C-I (H3C)2CuLi an enolatearrow_forwardb. H3C CH3 1. 2. H3O+ H3C MgBr H3Carrow_forwardPredict the major products of this reaction: excess H+ NaOH ? A Note that the first reactant is used in excess, that is, there is much more of the first reactant than the second. If there won't be any products, just check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privarrow_forward
- 1. For each of the reaction "railroads" below, you are either asked to give the structure(s) of the starting material(s) or product(s), or provide reagents/conditions to accomplish the transformation, as indicated by the boxes. a. NaOMe H+ .CO,H HO₂C MeOH (excess) MeOH H3C Br يع CH3 1. LiAlH4 2. H3O+ 3. PBг3 H3C 1. Et-Li 2. H3O+ -CO₂Me -CO₂Me OH CH3 CH3 ল CH3arrow_forwardPredict the intermediate 1 and final product 2 of this organic reaction: NaOMe ག1, ད།་, - + H You can draw 1 and 2 in any arrangement you like. 2 work up Note: if either 1 or 2 consists of a pair of enantiomers, just draw one structure using line bonds instead of 3D (dash and wedge) bonds at the chiral center. Explanation Check Click and drag to start drawing a structure. Х © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Parrow_forwardWhat is the total energy cost associated with the compound below adopting the shown conformation? CH3 HH DH CH3arrow_forward
- ΗΝ, Draw Final Product C cyclohexanone pH 4-5 Edit Enamine H3O+ CH3CH2Br THF, reflux H Edit Iminium Ionarrow_forwardHow many hydrogen atoms are connected to the indicated carbon atom?arrow_forwardIdentify the compound with the longest carbon - nitrogen bond. O CH3CH2CH=NH O CH3CH2NH2 CH3CH2C=N CH3CH=NCH 3 The length of all the carbon-nitrogen bonds are the samearrow_forward
- Identify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer below. Η H's+ 6Η Η Η Η Η Ηδ Η Ο Ο HH +Η Η +Η Η Η -8+ CIarrow_forwardH H:O::::H H H HH H::O:D:D:H HH HH H:O:D:D:H .. HH H:O:D:D:H H H Select the correct Lewis dot structure for the following compound: CH3CH2OHarrow_forwardRank the following compounds in order of decreasing boiling point. ннннн -С-С-Н . н-с- ННННН H ΗΤΗ НННН TTTĪ н-с-с-с-с-о-н НННН НН C' Н н-с-с-с-с-н НН || Ш НННН H-C-C-C-C-N-H ННННН IVarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





