
Concept explainers
(a)
Interpretation:
The IUPAC names of the product formed from hydrolysis of given hemiacetal in an acid solution has to be given.
Concept Introduction:
Aldehydes and ketones react with alcohol to form hemiacetal as the product. This reacts with further molecule of aldehyde or ketone to form acetal.
Acetals are stable in basic solution. But in acidic solution, they undergo hydrolysis. Hydrolysis is a
Hemiacetal undergoes hydrolysis in acidic solution to form one alcohol molecule and ketone or aldehyde molecule. The general reaction for hydrolysis of hemiacetal in acid solution,
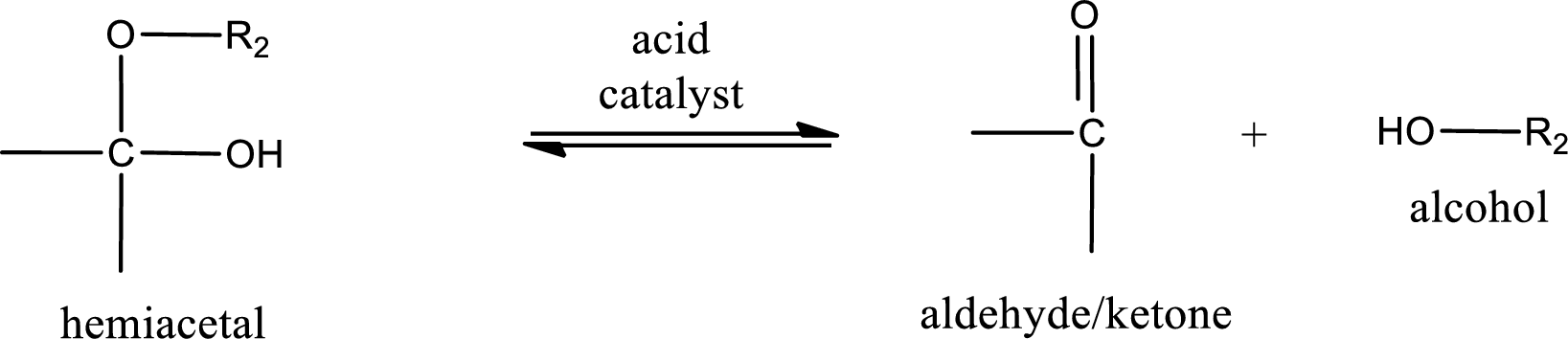
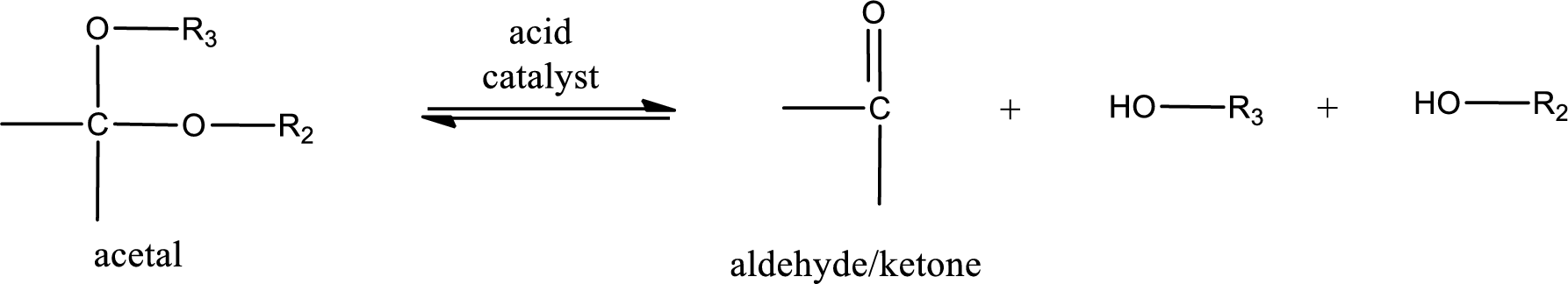
Acetal undergoes hydrolysis in acidic solution to form two alcohol molecules and ketone or aldehyde molecule. The general reaction for hydrolysis of acetal in acid solution,
(b)
Interpretation:
The IUPAC names of the product formed from hydrolysis of given acetal in an acid solution has to be given.
Concept Introduction:
Aldehydes contain a carbonyl group that is bonded to a hydrogen atom and a carbon atom. Ketones are compounds that contain a carbonyl group bonded to two carbon atoms. Aldehydes and ketones undergo addition reaction across the carbonyl group.
Aldehydes and ketones react with alcohol to form hemiacetal as the product. This reacts with further molecule of aldehyde or ketone to form acetal.
Acetals are stable in basic solution. But in acidic solution, they undergo hydrolysis. Hydrolysis is a chemical reaction in which the compound splits into two or more fragments when water is added to the compound in presence of acid or base as catalyst. Acetals undergo hydrolysis to give the respective starting materials from which it is formed.
Hemiacetal undergoes hydrolysis in acidic solution to form one alcohol molecule and ketone or aldehyde molecule. The general reaction for hydrolysis of hemiacetal in acid solution,
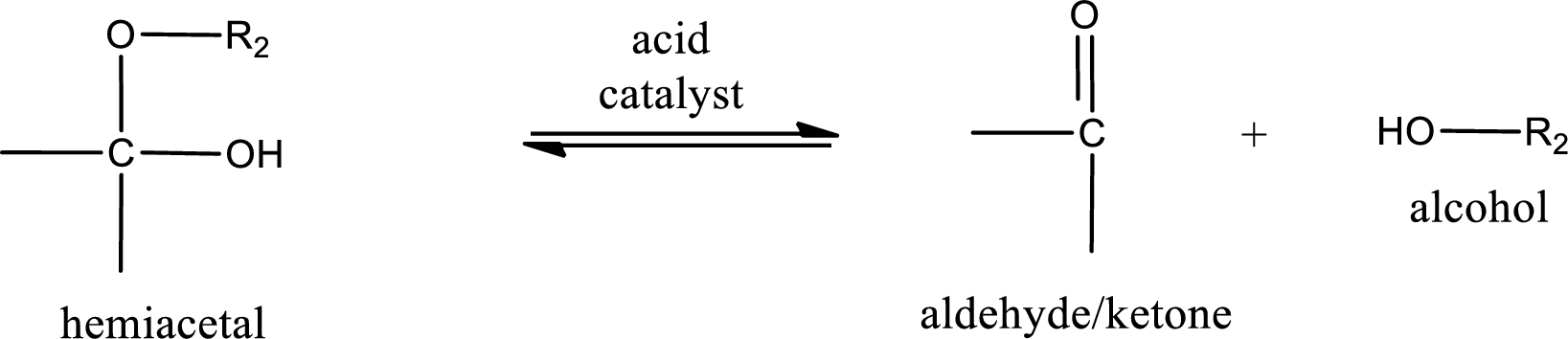
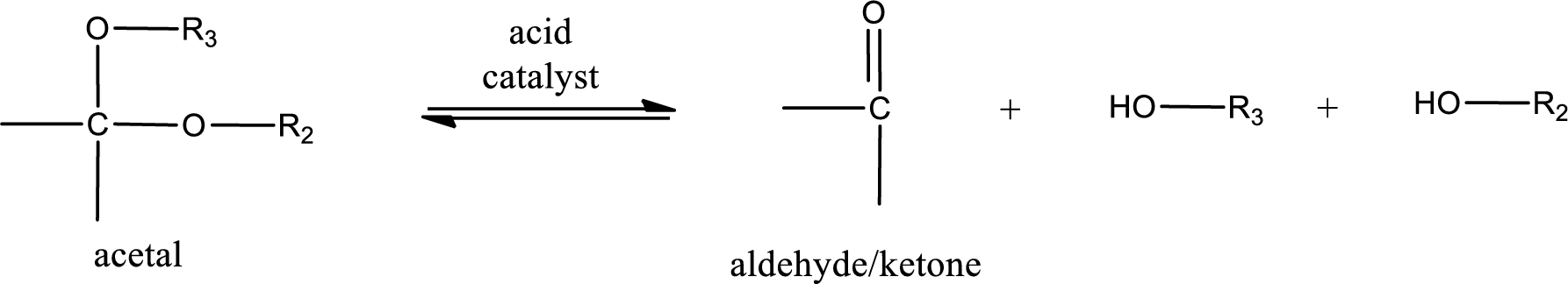
Acetal undergoes hydrolysis in acidic solution to form two alcohol molecules and ketone or aldehyde molecule. The general reaction for hydrolysis of acetal in acid solution,
(c)
Interpretation:
The IUPAC names of the product formed from hydrolysis of given hemiacetal in an acid solution has to be given.
Concept Introduction:
Aldehydes contain a carbonyl group that is bonded to a hydrogen atom and a carbon atom. Ketones are compounds that contain a carbonyl group bonded to two carbon atoms. Aldehydes and ketones undergo addition reaction across the carbonyl group.
Aldehydes and ketones react with alcohol to form hemiacetal as the product. This reacts with further molecule of aldehyde or ketone to form acetal.
Acetals are stable in basic solution. But in acidic solution, they undergo hydrolysis. Hydrolysis is a chemical reaction in which the compound splits into two or more fragments when water is added to the compound in presence of acid or base as catalyst. Acetals undergo hydrolysis to give the respective starting materials from which it is formed.
Hemiacetal undergoes hydrolysis in acidic solution to form one alcohol molecule and ketone or aldehyde molecule. The general reaction for hydrolysis of hemiacetal in acid solution,
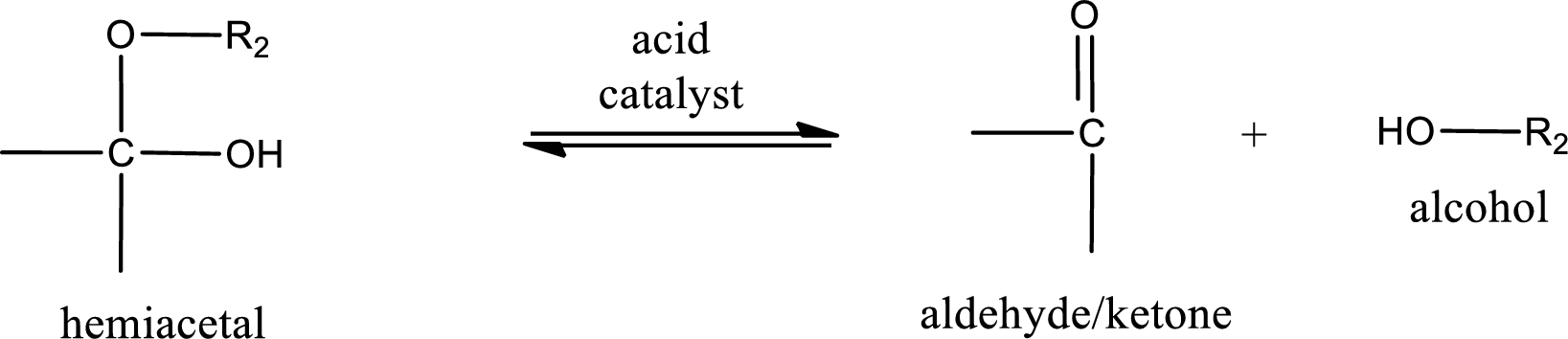
Acetal undergoes hydrolysis in acidic solution to form two alcohol molecules and ketone or aldehyde molecule. The general reaction for hydrolysis of acetal in acid solution,
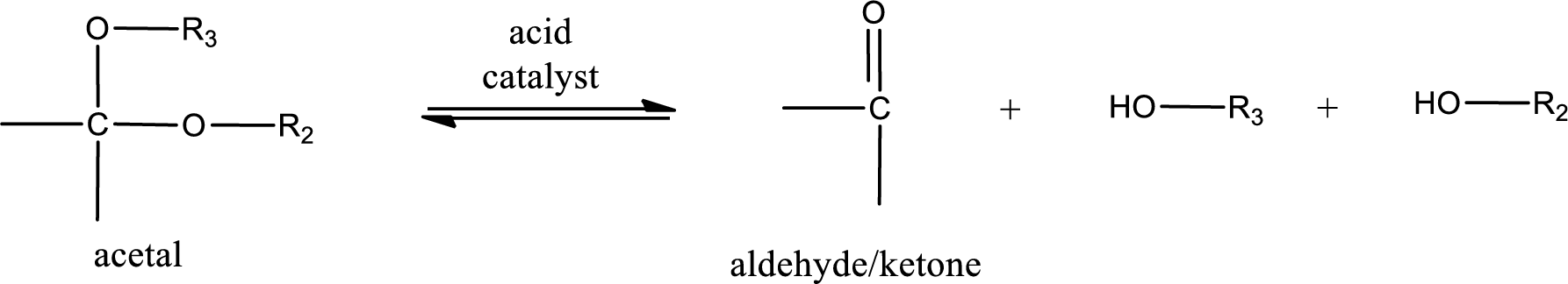
(d)
Interpretation:
The IUPAC names of the product formed from hydrolysis of given acetal in an acid solution has to be given.
Concept Introduction:
Aldehydes contain a carbonyl group that is bonded to a hydrogen atom and a carbon atom. Ketones are compounds that contain a carbonyl group bonded to two carbon atoms. Aldehydes and ketones undergo addition reaction across the carbonyl group.
Aldehydes and ketones react with alcohol to form hemiacetal as the product. This reacts with further molecule of aldehyde or ketone to form acetal.
Acetals are stable in basic solution. But in acidic solution, they undergo hydrolysis. Hydrolysis is a chemical reaction in which the compound splits into two or more fragments when water is added to the compound in presence of acid or base as catalyst. Acetals undergo hydrolysis to give the respective starting materials from which it is formed.
Hemiacetal undergoes hydrolysis in acidic solution to form one alcohol molecule and ketone or aldehyde molecule. The general reaction for hydrolysis of hemiacetal in acid solution,
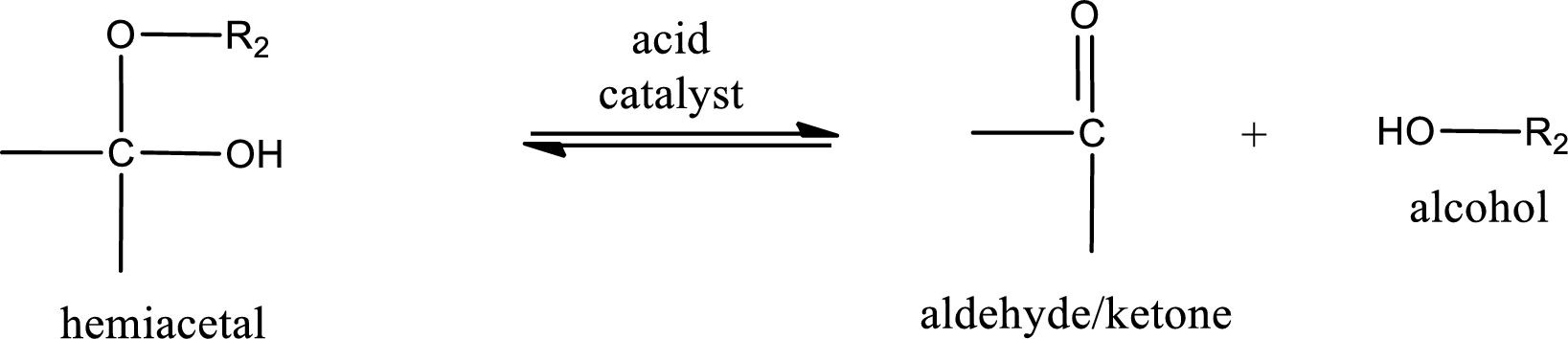
Acetal undergoes hydrolysis in acidic solution to form two alcohol molecules and ketone or aldehyde molecule. The general reaction for hydrolysis of acetal in acid solution,
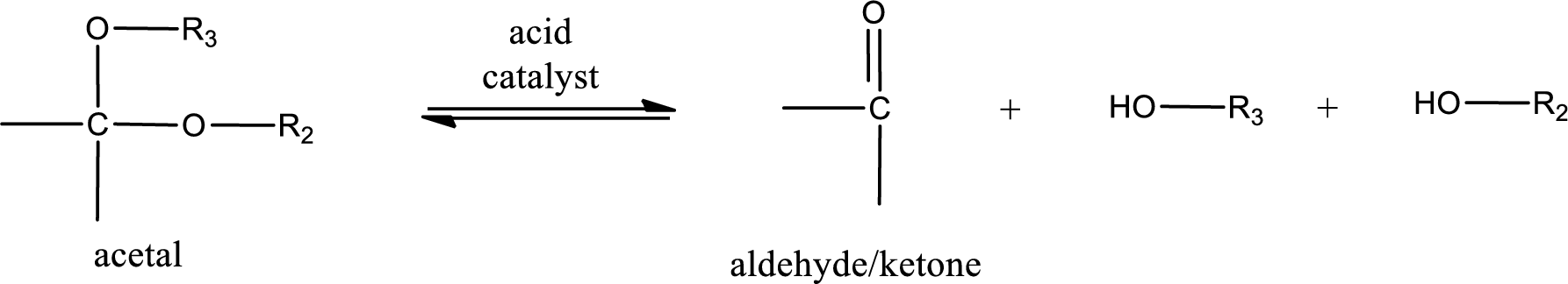
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic And Biological Chemistry
- Consider the reaction below to answer the following questions: 847 Acetoacetic ester can be prepared by the Claisen self-condensation reaction of ethyl acetate. H₁C 0 H 0 IL 유 || OCH2CH3 1. NaOEt, EtOH C 2. H₂O H3C CH₂ Cold not tobizmo. S OCH2CH3 A. Write the complete stepwise mechanism for this reaction. Show all electron flow with arrows and draw all intermediate structures. B. Ethyl acetate can be prepared from ethanol as the only organic starting material. Show all reagents and structures for all intermediates in this preparation. C. Give the structures of the ester precursors for the following Claisen condensation product and formulate the reaction. ou OELarrow_forwardA. What is the correct structure for a-D-glucopyranose? CH₂OH a HO HO- OH b HO HO- OH HOH₂C OH OH OH CH₂OH HO C. HO HO- OH OH CH₂OH OH OH B. Draw structures for the products you would expect to obtain from reaction of B-D-galactopyranose with each of the following reagents. Be sure to include all relevant stereochemistry. [FOUR only] A. CH, Ag₂O B. warm dilute HNO3 C. (CH3CO)20, pryridine D. NaBH in H₂O E. CH₂OH, HCI F. Br₂, H₂O HO CH₂OH HO- OH OH B-D-galactopyranosearrow_forwardGive the major organic product(s) for each of the following reactions or reaction sequences. CH₂CN 5% NaOEt, EIOH سجد سی . بلی H 1. NaOCH, CH,OH CH3 OCH3 2 H₂O*arrow_forward
- Draw the structures for each of the intermediates in the boxes provided for the synthesis below. 004 HNO F HO CHCO) D Dydre R.SO. 1.1 NO fe HO H.SO. 2. CC1 NOH HO MCL HNO, H.50.arrow_forward. Each of the following compounds can be prepared by a mixed aldol condensation reaction. Give the Cructures of the aldehyde and/or ketone precursors for each aldol product and formulate the reaction. 0 CH=CHCCH 3. Ph 1arrow_forward. Consider the reaction below to answer the following question: H NaOEt H BOH بلی H + H₂O A. Write the complete stepwise mechanism for the reaction above. Show all intermediate structures and all electron flow with arrows. B. This reaction is an example of: an intramolecular aldol condensation a. an intramolecular Claisen condensation b. C. d. a Robinson annulation a Michael reaction C. The product of this reaction is: a. b. C. d. a ẞ. y-unsaturated aldehyde an a, B-unsaturated ketone an a, B-unsaturated aldehyde an enolarrow_forward
- Classify each of the following nitrogen atoms in the following compounds as primary, secondary, tiary, or quaternary. A. B. C. CH3 HO-CHCHNHCH3 ephedrine CH CHCH3 amphetamine NH₂ D. CF H3C CH3 mapiquat chloride HO fexofenadine OH H3C CH3 CO₂Harrow_forward.. Name each of the following compounds by IUPAC rules. [three Only] A. 0 B. C. Cl NH₂ OCH N CH3 NHCH2CH3 D. CH O₁₂N NH₂arrow_forwardDraw the structure of the aldol self condensation product for each of the following compounds if a compound does not under go aldol self condensation explain whyarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





