
Concept explainers
Interpretation:
The pair of structural isomers needs to be identified from the given condensed structural formulas.
Concept introduction:
The molecules having same molecular formula but differ in structural arrangement of atoms are said to be isomers of each other.
Answer to Problem 69A
The pair of structural isomers in the given compound are:
- a and b.
- a and c.
- a and d.
- c and d.
- c and b.
Explanation of Solution
Given:
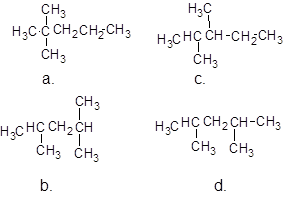
1. The parent (longest) continuous carbon chain is identified.
2. The ending of the parent chain for
3. Name should be written in alphabetical order and numbering should be done in such a way that the substituent group gets lowest number.
4. Hyphen is used to connect the number to the name.
For number of carbons atoms in alkane chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The numbering is done in such a way that the substituents gets lower number and the compound has longest carbon chain as a parent chain as:
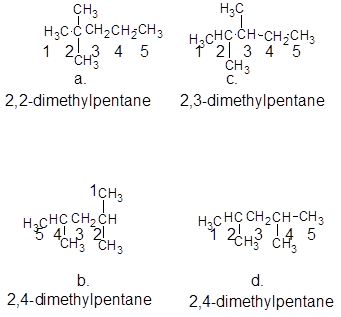
The molecular formula of all the compounds is same that is, 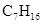 and the name of compounds a and c are different and of compound b and d are same, compound b and d are same but are isomers to a and c and a and c are isomers to each other also.
and the name of compounds a and c are different and of compound b and d are same, compound b and d are same but are isomers to a and c and a and c are isomers to each other also.
Hence, the pair of structural isomers in the given compound are:
- a and b.
- a and c.
- a and d.
- c and d.
- c and b.
Chapter 21 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Brock Biology of Microorganisms (15th Edition)
Applications and Investigations in Earth Science (9th Edition)
Biology: Life on Earth (11th Edition)
College Physics: A Strategic Approach (3rd Edition)
Genetic Analysis: An Integrated Approach (3rd Edition)
Anatomy & Physiology (6th Edition)
- Steps and explanation please. Add how to solve or target similar problems.arrow_forwardWould the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forwardThis organic molecule is dissolved in an acidic aqueous solution: OH OH A short time later sensitive infrared spectroscopy reveals the presence of a new C = O stretch absorption. That is, there must now be a new molecule present with at least one C = O bond. In the drawing area below, show the detailed mechanism that could convert the molecule above into the new molecule. Videos 849 Explanation Check C Click and drag to start dwing a structure. # 3 MAR 23 Add/Remove steparrow_forward||| 7:47 ull 57% ← Problem 19 of 48 Submit Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the curved arrows to draw the product of this carbocation rearrangement. Include all lone pairs and charges as appropriate. H 1,2-alkyl shift +arrow_forwardWould the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forwardBelow is the SN1 reaction of (S)-3-chlorocyclohexene and hydroxide (OH). Draw the missing curved arrows, lone pairs of electrons, and nonzero formal charges. In the third box, draw the two enantiomeric products that will be produced. 5th attempt Please draw all four bonds at chiral centers. Draw the two enantiomeric products that will be produced. Draw in any hydrogen at chiral centers. 1000 4th attempt Feedback Please draw all four bonds at chiral centers. 8. R5 HO: See Periodic Table See Hint H Cl Br Jid See Periodic Table See Hintarrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





