
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.SE, Problem 63AP
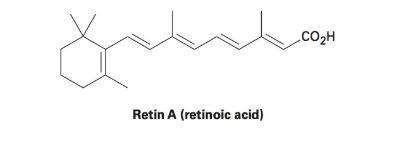
Retin A, or retinoic acid, is a medication commonly used to reduce wrinkles and treat severe acne. How many different isomers arising from double-bond isomerizations are possible?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Consider the solubility products (Ksp values) for the following compounds:SrSO4 (Ksp = 7.6 x 10−7), BaSO4 (Ksp = 1.5 x 10−9), SrCO3 (Ksp = 7.0 x 10−10), BaCO3 (Ksp = 1.6 x 10−9)Which anion is the harder base, CO32− or SO42−? Justify your answer.
Q1:
a) Arrange the compounds in order of decreasing pKa, highest first.
ОН
ΟΗ
ῸΗ
дон
ОН
ОН
CI
Br
(4 pts - 2 pts each part) A route that can be taken to prepare a
hydrophobic (water-repellent) aerogel is to start with
trichloromethylsilane, CH3SiCl3 as the silicon source.
a. What is the chemical reaction that this undergoes to form a
product with Si-OH groups? Write as complete of a chemical
equation as you can.
CI
CI-SI-CH3
CI
b. The formation of a byproduct is what drives this reaction - what is the byproduct (if you
didn't already answer it in part (a)) and how/why does it form?
Chapter 7 Solutions
Organic Chemistry
Ch. 7.2 - Calculate the degree of unsaturation in each of...Ch. 7.2 - Prob. 2PCh. 7.2 - Diazepam, marketed as an antianxiety medication...Ch. 7.3 - Give IUPAC names for the following compounds:Ch. 7.3 - Prob. 5PCh. 7.3 - Prob. 6PCh. 7.3 - Prob. 7PCh. 7.4 - Prob. 8PCh. 7.4 - Prob. 9PCh. 7.4 - Prob. 10P
Ch. 7.5 - Which member in each of the following sets ranks...Ch. 7.5 - Prob. 12PCh. 7.5 - Prob. 13PCh. 7.5 - Prob. 14PCh. 7.6 - Prob. 15PCh. 7.8 - Prob. 16PCh. 7.8 - Prob. 17PCh. 7.9 - Show the structures of the carbocation...Ch. 7.9 - Draw a skeletal structure of the following...Ch. 7.10 - Prob. 20PCh. 7.11 - On treatment with HBr, vinylcyclohexane undergoes...Ch. 7.SE - Prob. 22VCCh. 7.SE - Prob. 23VCCh. 7.SE - The following carbocation is an intermediate in...Ch. 7.SE - Prob. 25VCCh. 7.SE - Predict the major product and show the complete...Ch. 7.SE - Prob. 27MPCh. 7.SE - When 1, 3-butadiene reacts with one mole of HBr,...Ch. 7.SE - When methyl vinyl ether reacts with a strong acid,...Ch. 7.SE - Addition of HCl to 1-isopropylcyclohexene yields a...Ch. 7.SE - Addition of HCl to...Ch. 7.SE - Limonene, a fragrant hydrocarbon found in lemons...Ch. 7.SE - Prob. 33MPCh. 7.SE - Calculate the degree of unsaturation in the...Ch. 7.SE - Prob. 35APCh. 7.SE - Prob. 36APCh. 7.SE - Name the following alkenes:Ch. 7.SE - Draw structures corresponding to the following...Ch. 7.SE - Prob. 39APCh. 7.SE - Prob. 40APCh. 7.SE - Prob. 41APCh. 7.SE - Prob. 42APCh. 7.SE - Prob. 43APCh. 7.SE - Draw and name the 17 alkene isomers, C6H12,...Ch. 7.SE - Prob. 45APCh. 7.SE - Prob. 46APCh. 7.SE - Which of the following E, Z designations are...Ch. 7.SE - Prob. 48APCh. 7.SE - trans-2-Butene is more stable than cis-2-butene by...Ch. 7.SE - Prob. 50APCh. 7.SE - Normally, a trans alkene is more stable than its...Ch. 7.SE - trans-Cyclooctene is less stable than...Ch. 7.SE - Prob. 53APCh. 7.SE - Prob. 54APCh. 7.SE - Use Hammond’s Postulate to determine which...Ch. 7.SE - Prob. 56APCh. 7.SE - Predict the major product in each of the following...Ch. 7.SE - Prob. 58APCh. 7.SE - Prob. 59APCh. 7.SE - Prob. 60APCh. 7.SE - Allene (1,2-propadiene), H2C=C=CH2, has two...Ch. 7.SE - The heat of hydrogenation for allene (Problem...Ch. 7.SE - Retin A, or retinoic acid, is a medication...Ch. 7.SE - Prob. 64APCh. 7.SE - tert-Butyl esters [RC02C(CH3)3] are converted into...Ch. 7.SE - Vinylcyclopropane reacts with HBr to yield a...Ch. 7.SE - Prob. 67APCh. 7.SE - Prob. 68APCh. 7.SE - Prob. 69APCh. 7.SE - Prob. 70APCh. 7.SE - Prob. 71APCh. 7.SE - Reaction of 2, 3-dimethyl-l-butene with HBr leads...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- b) Circle the substrate that would not efficiently generate a Grignard reagent upon reaction with Mg in ether. CI Br ד c) Circle the Grignard reagents that contain incompatible functional groups. MgBr HO MgBr MgBr MgBr MgBr HO MgBrarrow_forwardQ2: Predict all organic product(s), including stereoisomers when applicable. PCC OH a) CH2Cl2 Page 2 of 5 Chem 0310 Organic Chemistry 1 HW Problem Sets b) .OH Na2Cr2O7, H+ OH PCC CH2Cl2 c) OHarrow_forwardd) Circle the substrates that will give an achiral product after a Grignard reaction with CH3MgBr. Harrow_forward
- Explain why the S-F bond strength is 367 kJ/mol in SF2 and 329 kJ/mol in SF6.arrow_forwardWould Si(CH3)3F react with AgCl? If so, write out the balanced chemical equation. If not,explain why no reaction would take place.arrow_forwardNH3 reacts with boron halides (BX3 where X = F, Cl, Br, or I) to form H3N-BX3 complexes.Which of these complexes will have the strongest N-B bond? Justify your answerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning 
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning



Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY