
a)
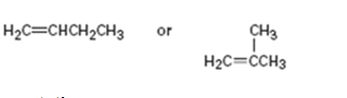
Interpretation:
The names of the
Concept introduction:
The longest carbon chain containing the double bond to be chosen. Based on the name of the parent compound - the alkene name ends with the suffix -ene. The chain is to be numbered from the end that gives the lowest number to the carbon in double bond. Substituents are to be numbered according to their positions in the chain and listed alphabetically. The position of the double bond is indicated by giving the number of the first alkene carbon before the name of the parent. The stability of alkenes can be explained using the concept of hyperconjucation or based on the bond strengths. Further a compound with more number of sp3-sp2 bonds is more stable.
To name:
The alkene given and to state which is more stable.
b)
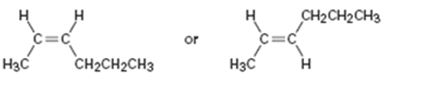
Interpretation:
The names of the alkenes given are to be given. Among the two compounds which is more stable also is to be stated.
Concept introduction:
The longest carbon chain containing the double bond to be chosen. Based on the name of the parent compound - the alkene name ends with the suffix -ene. The chain is to be numbered from the end that gives the lowest number to the carbon in double bond. Substituents are to be numbered according to their positions in the chain and listed alphabetically. The position of the double bond is indicated by giving the number of the first alkene carbon before the name of the parent. Among cis and trans isomers, the cis isomer is less stable than the trans isomer, because of the steric strain between the two larger substituents on the same side of the double bond.
To name:
The alkene given and to state which is more stable.
c)
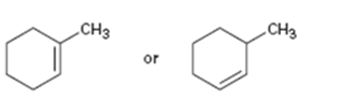
Interpretation:
The names of the alkenes given are to be given. Among the two compounds which is more stable also is to be stated.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane - the cycloalkene is named with the suffix -ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. The stability of cycloalkenes can be explained using the concept of hyper conjucation or based on the bond strengths.
To name:
The alkene given and to state which is more stable.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- Predict the major organic product(s) of the following reactions. Include stereochemistry when necessary. Write NR if no reaction, try to explain.arrow_forwardQ2: Explain why epoxides that react in an SN1 manner will not show any stereochemical inversion in the product. Q3: Rationalize why Alcohol B will react under the indicated reaction conditions, but Alcohol A will not. A ☑ OH B OH PBr3 R-Brarrow_forwardQ1: Predict the major organic product(s) of the following reactions. Include stereochemistry when necessary. Write NR if no reaction, try to explain. 1.) LDA, THF 2.) СОН CI OH H2SO4, heat OH m...... OH 1.) PCC, CH2Cl2 2.) CH3CH2MgBr, THF 3.) H3O+ 4.) TsCl, pyr 5.) tBuOK, tBuOH 1.) SOCI 2, CHCI 3 2.) CH3CH2ONA, DMF OH 1.) HBr 2.) Mg, THF 3.) H₂CO, THE 4.) H3O+ OH NaH, THFarrow_forward
- Problem 6-29 Identify the functional groups in the following molecules, and show the polarity of each: (a) CH3CH2C=N CH, CH, COCH (c) CH3CCH2COCH3 NH2 (e) OCH3 (b) (d) O Problem 6-30 Identify the following reactions as additions, eliminations, substitutions, or rearrangements: (a) CH3CH2Br + NaCN CH3CH2CN ( + NaBr) Acid -OH (+ H2O) catalyst (b) + (c) Heat NO2 Light + 02N-NO2 (+ HNO2) (d)arrow_forwardPredict the organic product of Y that is formed in the reaction below, and draw the skeletal ("line") structures of the missing organic product. Please include all steps & drawings & explanations.arrow_forwardPlease choose the best reagents to complete the following reactionarrow_forward
- Problem 6-17 Look at the following energy diagram: Energy Reaction progress (a) Is AG for the reaction positive or negative? Label it on the diagram. (b) How many steps are involved in the reaction? (c) How many transition states are there? Label them on the diagram. Problem 6-19 What is the difference between a transition state and an intermediate? Problem 6-21 Draw an energy diagram for a two-step reaction with Keq > 1. Label the overall AG°, transition states, and intermediate. Is AG° positive or negative? Problem 6-23 Draw an energy diagram for a reaction with Keq = 1. What is the value of AG° in this reaction?arrow_forwardProblem 6-37 Draw the different monochlorinated constitutional isomers you would obtain by the radical chlorination of the following compounds. (b) (c) Problem 6-39 Show the structure of the carbocation that would result when each of the following alkenes reacts with an acid, H+. (a) (b) (c)arrow_forwardPlease draw the major product of this reaction. Ignore inorganic byproducts and the carboxylic side productarrow_forward
