
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.SE, Problem 24VC
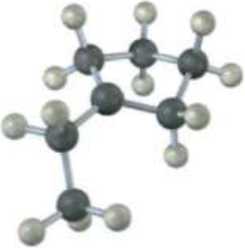
The following carbocation is an intermediate in the electrophilic addition reaction of HCl with two different
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Using the graphs could you help me explain the answers. I assumed that both graphs are proportional to the inverse of time, I think. Could you please help me.
Synthesis of Dibenzalacetone
[References]
Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone below.
Question 1
1 pt
Question 2
1 pt
Question 3
1 pt
H
Question 4
1 pt
Question 5
1 pt
Question 6
1 pt
Question 7
1pt
Question 8
1 pt
Progress:
7/8 items
Que Feb 24 at
You do not have to consider stereochemistry.
. Draw the enolate ion in its carbanion form.
• Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
⚫ Separate multiple reactants using the + sign from the drop-down menu.
?
4
Shown below is the mechanism presented for the formation of biasplatin in reference 1 from the Background and Experiment document. The amounts used of each reactant are shown. Either draw or describe a better alternative to this mechanism. (Note that the first step represents two steps combined and the proton loss is not even shown; fixing these is not the desired improvement.) (Hints: The first step is correct, the second step is not; and the amount of the anhydride is in large excess to serve a purpose.)
Chapter 7 Solutions
Organic Chemistry
Ch. 7.2 - Calculate the degree of unsaturation in each of...Ch. 7.2 - Prob. 2PCh. 7.2 - Diazepam, marketed as an antianxiety medication...Ch. 7.3 - Give IUPAC names for the following compounds:Ch. 7.3 - Prob. 5PCh. 7.3 - Prob. 6PCh. 7.3 - Prob. 7PCh. 7.4 - Prob. 8PCh. 7.4 - Prob. 9PCh. 7.4 - Prob. 10P
Ch. 7.5 - Which member in each of the following sets ranks...Ch. 7.5 - Prob. 12PCh. 7.5 - Prob. 13PCh. 7.5 - Prob. 14PCh. 7.6 - Prob. 15PCh. 7.8 - Prob. 16PCh. 7.8 - Prob. 17PCh. 7.9 - Show the structures of the carbocation...Ch. 7.9 - Draw a skeletal structure of the following...Ch. 7.10 - Prob. 20PCh. 7.11 - On treatment with HBr, vinylcyclohexane undergoes...Ch. 7.SE - Prob. 22VCCh. 7.SE - Prob. 23VCCh. 7.SE - The following carbocation is an intermediate in...Ch. 7.SE - Prob. 25VCCh. 7.SE - Predict the major product and show the complete...Ch. 7.SE - Prob. 27MPCh. 7.SE - When 1, 3-butadiene reacts with one mole of HBr,...Ch. 7.SE - When methyl vinyl ether reacts with a strong acid,...Ch. 7.SE - Addition of HCl to 1-isopropylcyclohexene yields a...Ch. 7.SE - Addition of HCl to...Ch. 7.SE - Limonene, a fragrant hydrocarbon found in lemons...Ch. 7.SE - Prob. 33MPCh. 7.SE - Calculate the degree of unsaturation in the...Ch. 7.SE - Prob. 35APCh. 7.SE - Prob. 36APCh. 7.SE - Name the following alkenes:Ch. 7.SE - Draw structures corresponding to the following...Ch. 7.SE - Prob. 39APCh. 7.SE - Prob. 40APCh. 7.SE - Prob. 41APCh. 7.SE - Prob. 42APCh. 7.SE - Prob. 43APCh. 7.SE - Draw and name the 17 alkene isomers, C6H12,...Ch. 7.SE - Prob. 45APCh. 7.SE - Prob. 46APCh. 7.SE - Which of the following E, Z designations are...Ch. 7.SE - Prob. 48APCh. 7.SE - trans-2-Butene is more stable than cis-2-butene by...Ch. 7.SE - Prob. 50APCh. 7.SE - Normally, a trans alkene is more stable than its...Ch. 7.SE - trans-Cyclooctene is less stable than...Ch. 7.SE - Prob. 53APCh. 7.SE - Prob. 54APCh. 7.SE - Use Hammond’s Postulate to determine which...Ch. 7.SE - Prob. 56APCh. 7.SE - Predict the major product in each of the following...Ch. 7.SE - Prob. 58APCh. 7.SE - Prob. 59APCh. 7.SE - Prob. 60APCh. 7.SE - Allene (1,2-propadiene), H2C=C=CH2, has two...Ch. 7.SE - The heat of hydrogenation for allene (Problem...Ch. 7.SE - Retin A, or retinoic acid, is a medication...Ch. 7.SE - Prob. 64APCh. 7.SE - tert-Butyl esters [RC02C(CH3)3] are converted into...Ch. 7.SE - Vinylcyclopropane reacts with HBr to yield a...Ch. 7.SE - Prob. 67APCh. 7.SE - Prob. 68APCh. 7.SE - Prob. 69APCh. 7.SE - Prob. 70APCh. 7.SE - Prob. 71APCh. 7.SE - Reaction of 2, 3-dimethyl-l-butene with HBr leads...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Hi I need help on the question provided in the image.arrow_forwardDraw a reasonable mechanism for the following reaction:arrow_forwardDraw the mechanism for the following reaction: CH3 CH3 Et-OH Et Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron-flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H± EXP. L CONT. י Α [1] осн CH3 а CH3 :Ö Et H 0 N о S 0 Br Et-ÖH | P LL Farrow_forward
- 20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY