
a)
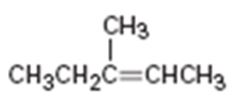
Interpretation:
Assuming Markovnikov’s rule is valid, the major alcohol product obtainable by the acid-catalyzed addition of water to 3-methyl-3-butene is to be predicted.
Concept introduction:
Markovnikov’s rule can be used to decide on the orientation in electrophilic addition reactions. According to this rule, in the addition of HX to an
To state:
The major alcohol product obtainable by the acid-catalyzed addition of water to 3-methyl-3-butene assuming that Markovnikov’s rule is valid.
b)
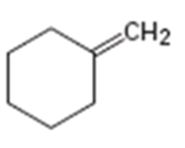
Interpretation:
Assuming Markovnikov’s rule is valid, the major alcohol product obtainable by the acid-catalyzed addition of water to methylidenecyclohexene is to be predicted.
Concept introduction:
Markovnikov’s rule can be used to decide on the orientation in electrophilic addition reactions. According to this rule, in the addition of HX to an alkene, the H attaches itself with the carbon with fewer alkyl substituents and the X attaches itself with the carbon with more alkyl substituents.
To state:
The major alcohol product obtainable by the acid-catalyzed addition of water to methylidenecyclohexene assuming that Markovnikov’s rule is valid.
c)
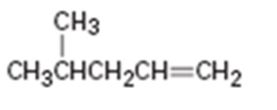
Interpretation:
Assuming Markovnikov’s rule is valid, the major alcohol product obtainable by the acid-catalyzed addition of water to 4- methyl-1-pentene is to be predicted.
Concept introduction:
Markovnikov’s rule can be used to decide on the orientation in electrophilic addition reactions. According to this rule, in the addition of HX to an alkene, the H attaches itself with the carbon with fewer alkyl substituents and the X attaches itself with the carbon with more alkyl substituents.
To state:
The major alcohol product obtainable by the acid-catalyzed addition of water to 4- methyl-1-pentene assuming that Markovnikov’s rule is valid.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- The cobalt mu-hydroxide complex cobaltate(III) of potassium is a dinuclear complex. Correct?arrow_forwardThe cobalt mi-hydroxide complex cobaltate(III) of potassium is a dinuclear complex. Correct?arrow_forward3. Arrange the different acids in Exercise B # 2 from the strongest (1) to the weakest acid (10). 1. 2. (strongest) 3. 4. 5. 6. 7. 8. 9. 10 10. (weakest)arrow_forward
- Name Section Score Date EXERCISE B pH, pOH, pка, AND PKD CALCULATIONS 1. Complete the following table. Solution [H+] [OH-] PH РОН Nature of Solution A 2 x 10-8 M B 1 x 10-7 M C D 12.3 6.8 2. The following table contains the names, formulas, ka or pka for some common acids. Fill in the blanks in the table. (17 Points) Acid Name Formula Dissociation reaction Ka pka Phosphoric acid H₂PO₁ H3PO4 H++ H₂PO 7.08 x 10-3 Dihydrogen H₂PO H₂PO H+ HPO 6.31 x 10-6 phosphate Hydrogen HPO₁ 12.4 phosphate Carbonic acid H2CO3 Hydrogen HCO 6.35 10.3 carbonate or bicarbonate Acetic acid CH,COOH 4.76 Lactic acid CH₂CHOH- COOH 1.38 x 10 Ammonium NH 5.63 x 10-10 Phenol CH₂OH 1 x 10-10 Protonated form CH3NH3* 3.16 x 10-11 of methylaminearrow_forwardIndicate whether it is true that Co(III) complexes are very stable.arrow_forwardMnO2 acts as an oxidant in the chlorine synthesis reaction.arrow_forward
- In Potassium mu-dihydroxydicobaltate (III) tetraoxalate K4[Co2(C2O4)4(OH)2], indicate whether the OH ligand type is bidentate.arrow_forwardImagine an electrochemical cell based on these two half reactions with electrolyte concentrations as given below: Oxidation: Pb(s) → Pb2+(aq, 0.10 M) + 2 e– Reduction: MnO4–(aq, 1.50 M) + 4 H+(aq, 2.0 M) + 3 e– → MnO2(s) + 2 H2O(l) Calculate Ecell (assuming temperature is standard 25 °C).arrow_forward: ☐ + Draw the Fischer projection of the most common naturally-occurring form of aspartate, with the acid group at the top and the side chain at the bottom. Important: be sure your structure shows the molecule as it would exist at physiological pH. Click and drag to start drawing a structure. ✓arrow_forward
- For a silver-silver chloride electrode, the following potentials are observed: E°cell = 0.222 V and E(saturated KCl) = 0.197 V Use this information to find the [Cl–] (technically it’s the activity of Cl– that’s relevant here, but we’ll just call it “concentration” for simplicity) in saturated KCl.arrow_forwardA concentration cell consists of two Sn/Sn2+ half-cells. The cell has a potential of 0.10 V at 25 °C. What is the ratio of [Sn2+] (i.e., [Sn2+left-half] / [Sn2+right-half])?arrow_forwardElectrochemical cell potentials can be used to determine equilibrium constants that would be otherwise difficult to determine because concentrations are small. What is Κ for the following balanced reaction if E˚ = +0.0218 V? 3 Zn(s) + 2 Cr3+(aq) → 3 Zn2+(aq) + Cr(s) E˚ = +0.0218 Varrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


