
Concept explainers
(a)
Interpretation:
The spatial arrangement for the
Concept Introduction:
Hydrocarbons are the organic compounds that contain only hydrogen and carbon atoms. Hydrocarbon derivatives are the one in which the compounds contain hydrogen and carbon atoms along with one or more additional elements. The additional elements that can be present in hydrocarbon derivatives are oxygen, nitrogen, sulphur, chlorine, bromine etc.
Hydrocarbons are further classified into two categories. They are saturated hydrocarbons and
Saturated hydrocarbons are
Alkane has general molecular formula as
Considering the geometry of carbon atoms, the carbon atoms that have double bonds will have trigonal planar geometry. The carbon atoms that have only single bonds attached to it will have tetrahedral geometry. The carbon atoms that have a triple bond attached to it will have a linear geometry.
(a)
Answer to Problem 2.37EP
The spatial arrangement is identified as tetrahedral.
Explanation of Solution
Given structure is,

Looking into the left most carbon atom present in the given structure, it is not bonded to any double bonds or triple bond. This carbon atom has only four single bonds (three with hydrogen and one with carbon atom). Therefore, the spatial arrangement of the left-most carbon atom is tetrahedral.
The spatial arrangement of the left-most carbon atom is identified.
(b)
Interpretation:
The spatial arrangement for the chemical bonds in the left‑most carbon atom in the given structure has to be identified.
Concept Introduction:
Hydrocarbons are the organic compounds that contain only hydrogen and carbon atoms. Hydrocarbon derivatives are the one in which the compounds contain hydrogen and carbon atoms along with one or more additional elements. The additional elements that can be present in hydrocarbon derivatives are oxygen, nitrogen, sulphur, chlorine, bromine etc.
Hydrocarbons are further classified into two categories. They are saturated hydrocarbons and unsaturated hydrocarbons. The hydrocarbons that contain single bonds between carbon atoms in the entire molecule is known as saturated hydrocarbon. The hydrocarbons that contain atleast one double or triple bond between two carbon atoms in the entire molecule is known as unsaturated hydrocarbon.
Saturated hydrocarbons are alkanes. Unsaturated hydrocarbons are alkene, alkyne and aromatic hydrocarbons.
Alkane has general molecular formula as
Considering the geometry of carbon atoms, the carbon atoms that have double bonds will have trigonal planar geometry. The carbon atoms that have only single bonds attached to it will have tetrahedral geometry. The carbon atoms that have a triple bond attached to it will have a linear geometry.
(b)
Answer to Problem 2.37EP
The spatial arrangement is identified as tetrahedral.
Explanation of Solution
Given structure is,

Looking into the left most carbon atom present in the given structure, it is not bonded to any double bonds or triple bond. This carbon atom has only four single bonds (three with hydrogen and one with carbon atom). Therefore, the spatial arrangement of the left-most carbon atom is tetrahedral.
The spatial arrangement of the left-most carbon atom is identified.
(c)
Interpretation:
The spatial arrangement for the chemical bonds in the left‑most carbon atom in the given structure has to be identified.
Concept Introduction:
Hydrocarbons are the organic compounds that contain only hydrogen and carbon atoms. Hydrocarbon derivatives are the one in which the compounds contain hydrogen and carbon atoms along with one or more additional elements. The additional elements that can be present in hydrocarbon derivatives are oxygen, nitrogen, sulphur, chlorine, bromine etc.
Hydrocarbons are further classified into two categories. They are saturated hydrocarbons and unsaturated hydrocarbons. The hydrocarbons that contain single bonds between carbon atoms in the entire molecule is known as saturated hydrocarbon. The hydrocarbons that contain atleast one double or triple bond between two carbon atoms in the entire molecule is known as unsaturated hydrocarbon.
Saturated hydrocarbons are alkanes. Unsaturated hydrocarbons are alkene, alkyne and aromatic hydrocarbons.
Alkane has general molecular formula as
Considering the geometry of carbon atoms, the carbon atoms that have double bonds will have trigonal planar geometry. The carbon atoms that have only single bonds attached to it will have tetrahedral geometry. The carbon atoms that have a triple bond attached to it will have a linear geometry.
(c)
Answer to Problem 2.37EP
The spatial arrangement is identified as trigonal planar.
Explanation of Solution
Given structure is,

Looking into the left most carbon atom present in the given structure, it is bonded to one double bond. This carbon atom has only two single bonds with hydrogen and a double bond with carbon atom. Therefore, the spatial arrangement of the left-most carbon atom is trigonal planar.
The spatial arrangement of the left-most carbon atom is identified.
(d)
Interpretation:
The spatial arrangement for the chemical bonds in the left‑most carbon atom in the given structure has to be identified.
Concept Introduction:
Hydrocarbons are the organic compounds that contain only hydrogen and carbon atoms. Hydrocarbon derivatives are the one in which the compounds contain hydrogen and carbon atoms along with one or more additional elements. The additional elements that can be present in hydrocarbon derivatives are oxygen, nitrogen, sulphur, chlorine, bromine etc.
Hydrocarbons are further classified into two categories. They are saturated hydrocarbons and unsaturated hydrocarbons. The hydrocarbons that contain single bonds between carbon atoms in the entire molecule is known as saturated hydrocarbon. The hydrocarbons that contain atleast one double or triple bond between two carbon atoms in the entire molecule is known as unsaturated hydrocarbon.
Saturated hydrocarbons are alkanes. Unsaturated hydrocarbons are alkene, alkyne and aromatic hydrocarbons.
Alkane has general molecular formula as
Considering the geometry of carbon atoms, the carbon atoms that have double bonds will have trigonal planar geometry. The carbon atoms that have only single bonds attached to it will have tetrahedral geometry. The carbon atoms that have a triple bond attached to it will have a linear geometry.
(d)
Answer to Problem 2.37EP
The spatial arrangement is identified as tetrahedral.
Explanation of Solution
Given structure is,
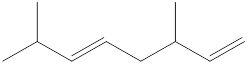
Looking into the left most carbon atom present in the given structure, it is not bonded to any double bonds or triple bond. This carbon atom has only four single bonds (three with hydrogen and one with carbon atom). Therefore, the spatial arrangement of the left-most carbon atom is tetrahedral.
The spatial arrangement of the left-most carbon atom is identified.
Want to see more full solutions like this?
Chapter 2 Solutions
Organic And Biological Chemistry
- oalmitic acid is a 16 carbon acid. In a balanced equation, the products of the sponification of tripalmitin (glyceryl tripalmitate are blank.arrow_forwardWrite the esterification reaction mechanism of salicylic acid and acetic acid to produce aspirin (acetylsalicylic acid). Note: salicylic acid will act as the alcoholarrow_forwardWhat type of interaction would you expect between the following R groups in the tertiary structure of a protein? O -CH2-CO and -CH2-CH2-CH2-CH2-NH3+ a. disulfide bonds b. salt bridges c. hydrogen bonds HO abios vist anisinoo tedt bigil s ai loistaslor sale! 10 OUT d. hydrophobic interactions e. peptide bondsarrow_forward
- 4. True or false: This skeletal structure represents a saturated fatty acid. Ini to 0 fale) me OH faistong starrow_forwardBy malonic or acetylacetic synthesis, synthesize 5-Methyl-2-hexanone (with the formulas of the compounds).arrow_forwardQUESTION: Answer Question 5: 'Calculating standard error of regression' by filling in all the empty green boxes *The values are all provided in the first photo attached*arrow_forward
- Draw the formula for 3-chlorobenzoic acetic anhydride.arrow_forwardBy malonic or acetylacetic synthesis, synthesize 2-methylbutanoic acid (indicate the formulas of the compounds).arrow_forwardObtain 2-methylbutanoic acid by malonic or acetylacetic synthesis (indicate the formulas of the compounds involved).arrow_forward
- EFFICIENTS SAMPLE READINGS CONCENTRATIONS Pigiadient) TOMATO SAUCE (REGULAR) TOMATO (REDUCED SALT) TOMATO SAUCE (REGULAR) TOMATO (REDUCED SALT) 58 6.274 3.898 301.7 151.2 14150 5.277 3.865 348.9 254.8 B 5.136 3.639 193.7 85.9 605 4.655 3.041 308.6 199.6 05 5.135 3.664 339.5 241.4 0139 4.676 3.662 160.6 87.6 90148 5.086 3.677 337.7 242.5 0092 6.348 3.775 464.7 186.4 PART3 5.081 3.908 223.5 155.8 5.558 3.861 370.5 257.1 4.922 3.66 326.6 242.9 4.752 3.641 327.5 253.3 50 5.018 3.815 336.1 256.0 84 4.959 3.605 317.9 216.6 38 4.96 3.652 203.8 108.7 $3 5.052 3.664 329.8 239.0 17 5.043 3.767 221.9 149.7 052 5.058 3.614 331.7 236.4 5.051 4.005 211.7 152.1 62 5.047 3.637 309.6 222.7 5.298 3.977 223.4 148.7 5.38 4.24 353.7 278.2 5 5.033 4.044 334.6 268.7 995 4.706 3.621 305.6 234.4 04 4.816 3.728 340.0 262.7 16 4.828 4.496 304.3 283.2 0.011 4.993 3.865 244.7 143.6 AVERAGE STDEV COUNT 95% CI Confidence Interval (mmol/L) [Na+] (mg/100 mL) 95% Na+ Confidence Interval (mg/100 mL)arrow_forwardIf we have two compounds: acetone (CH₃COCH₃) and acetic acid (CH₃COOH), applying heat to them produces an aldol condensation of the two compounds. If this is correct, draw the formula for the final product.arrow_forwardIf we have two compounds: acetone (CH3COCH3) and acetic acid (CH3COOH); if we apply heat (A), what product(s) are obtained?arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





