
Concept explainers
Assign an IUPAC name to each of the following
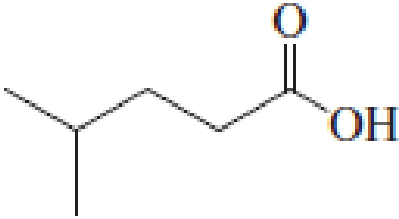
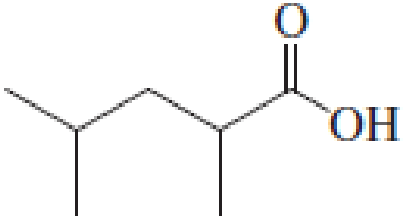
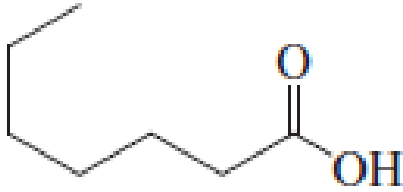
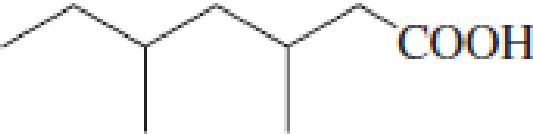
(a)
Interpretation:
IUPAC name for the carboxylic acid has to be given.
Concept Introduction:
For naming a carboxylic acid in IUPAC nomenclature, the suffix “-oic” is added to the parent alkane name.
IUPAC rules for naming a carboxylic acid:
- The longest parent carbon chain is identified that includes the carboxyl group.
- The parent chain name is changed by replacing the suffix “-e” with “-oic acid”.
- Numbering is done in a way that the carboxyl group is designated as number 1. This is not indicated in the part of the name because for aldehyde, the carboxyl carbon is always numbered 1.
- The identity and location of substituents if any has to be determined and this information has to be added in front of the IUPAC name.
- If the carboxyl functional group is attached to a ring of carbon atoms, the ring is named and “-carboxylic acid” is added as suffix.
In a line-angle structural formula the point at which two lines intersect and the end points are carbon atoms.
Answer to Problem 5.16EP
IUPAC name of the given carboxylic acid is 4-methylpentanoic acid.
Explanation of Solution
Given structure of carboxylic acid is,
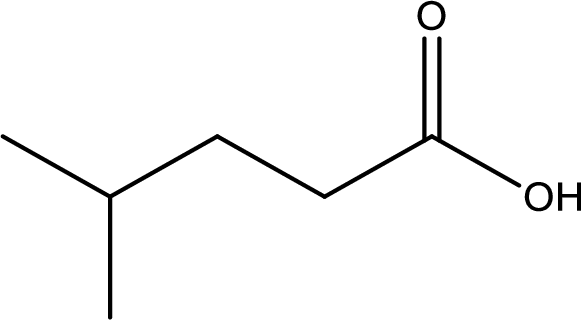
The longest continuous carbon chain has to be found out with the carboxyl group in it. In this it is a five carbon chain. Hence, the parent alkane is pentane. The carboxylic acid is named by replacing the suffix “-e” in the alkane name with “-oic acid”. This gives the name of carboxylic acid as pentanoic acid.
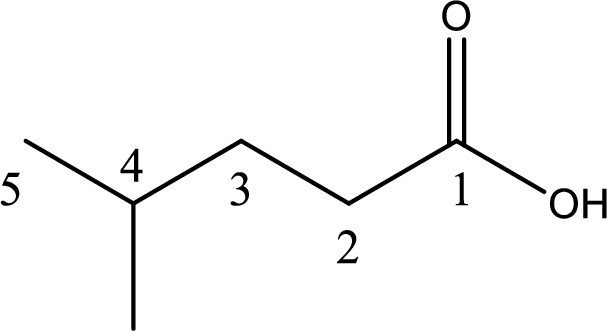
Looking for substituents it is found that there is a methyl group present on fourth carbon atom. Hence, the IUPAC name of the given carboxylic acid is 4-methylpentanoic acid.
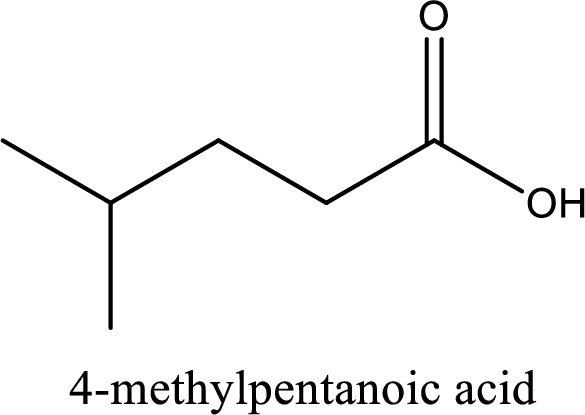
The IUPAC name of the given carboxylic acid is 4-methylpentanoic acid.
IUPAC name of the given carboxylic acid is found out.
(b)
Interpretation:
IUPAC name for the carboxylic acid has to be given.
Concept Introduction:
For naming a carboxylic acid in IUPAC nomenclature, the suffix “-oic” is added to the parent alkane name.
IUPAC rules for naming a carboxylic acid:
- The longest parent carbon chain is identified that includes the carboxyl group.
- The parent chain name is changed by replacing the suffix “-e” with “-oic acid”.
- Numbering is done in a way that the carboxyl group is designated as number 1. This is not indicated in the part of the name because for carboxylic acid, the carboxyl carbon is always numbered 1.
- The identity and location of substituents if any has to be determined and this information has to be added in front of the IUPAC name.
- If the carboxyl functional group is attached to a ring of carbon atoms, the ring is named and “-carboxylic acid” is added as suffix.
In a line-angle structural formula the point at which two lines intersect and the end points are carbon atoms.
Answer to Problem 5.16EP
IUPAC name of the given carboxylic acid is 2,4-dimethylpentanoic acid.
Explanation of Solution
Given structure of carboxylic acid is,
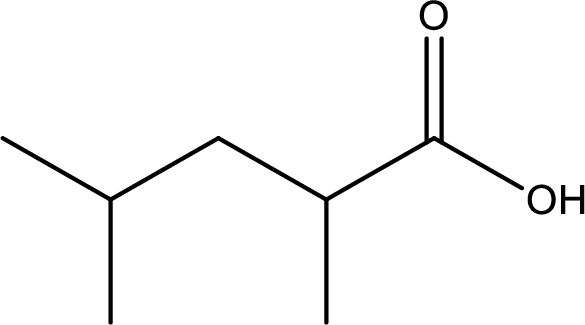
The longest continuous carbon chain has to be found out with the carboxyl group in it. In this it is a five carbon chain. Hence, the parent alkane is pentane. The carboxylic acid is named by replacing the suffix “-e” in the alkane name with “-oic acid”. This gives the name of carboxylic acid as pentanoic acid.
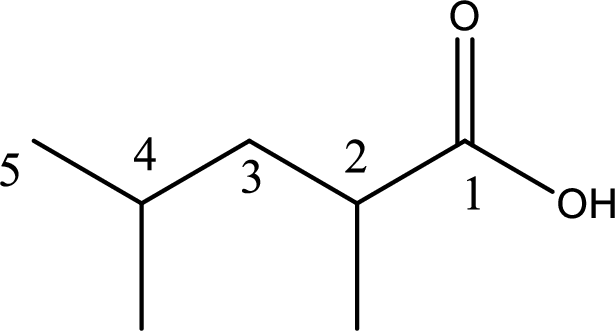
Looking for substituents it is found that there are two methyl groups present, each on second and fourth carbon atom. In the IUPAC name prefix “-di” has to be added before the alkyl name. Hence, the IUPAC name of the given carboxylic acid is 2,4-dimethylpentanoic acid.
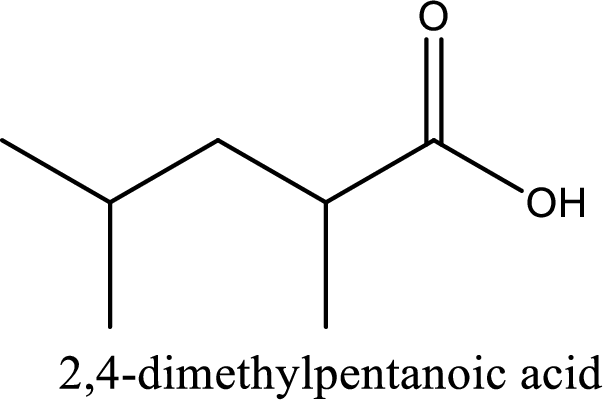
The IUPAC name of the given carboxylic acid is 2,4-dimethylpentanoic acid.
IUPAC name of the given carboxylic acid is found out.
(c)
Interpretation:
IUPAC name for the carboxylic acid has to be given.
Concept Introduction:
For naming a carboxylic acid in IUPAC nomenclature, the suffix “-oic” is added to the parent alkane name.
IUPAC rules for naming a carboxylic acid:
- The longest parent carbon chain is identified that includes the carboxyl group.
- The parent chain name is changed by replacing the suffix “-e” with “-oic acid”.
- Numbering is done in a way that the carboxyl group is designated as number 1. This is not indicated in the part of the name because for aldehyde, the carboxyl carbon is always numbered 1.
- The identity and location of substituents if any has to be determined and this information has to be added in front of the IUPAC name.
- If the carboxyl functional group is attached to a ring of carbon atoms, the ring is named and “-carboxylic acid” is added as suffix.
In a line-angle structural formula the point at which two lines intersect and the end points are carbon atoms.
Answer to Problem 5.16EP
IUPAC name of the given carboxylic acid is heptanoic acid.
Explanation of Solution
Given structure of carboxylic acid is,
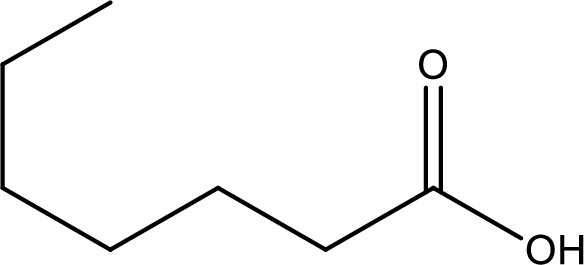
The longest continuous carbon chain has to be found out with the carboxyl group in it. In this it is a seven carbon chain. Hence, the parent alkane is heptane. The carboxylic acid is named by replacing the suffix “-e” in the alkane name with “-oic acid”. This gives the name of carboxylic acid as heptanoic acid.
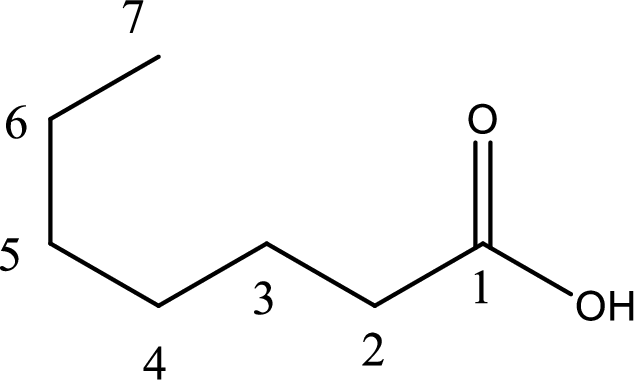
Looking for substituents it is found that there are no substituents present on the carbon chain. Hence, the IUPAC name of the given carboxylic acid is heptanoic acid.
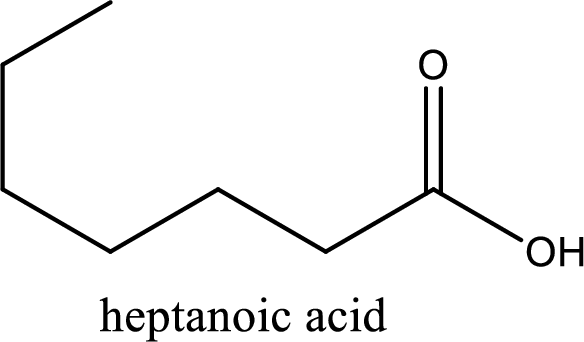
The IUPAC name of the given carboxylic acid is heptanoic acid.
IUPAC name of the given carboxylic acid is found out.
(d)
Interpretation:
IUPAC name for the carboxylic acid has to be given.
Concept Introduction:
For naming a carboxylic acid in IUPAC nomenclature, the suffix “-oic” is added to the parent alkane name.
IUPAC rules for naming a carboxylic acid:
- The longest parent carbon chain is identified that includes the carboxyl group.
- The parent chain name is changed by replacing the suffix “-e” with “-oic acid”.
- Numbering is done in a way that the carboxyl group is designated as number 1. This is not indicated in the part of the name because for aldehyde, the carboxyl carbon is always numbered 1.
- The identity and location of substituents if any has to be determined and this information has to be added in front of the IUPAC name.
- If the carboxyl functional group is attached to a ring of carbon atoms, the ring is named and “-carboxylic acid” is added as suffix.
In a line-angle structural formula the point at which two lines intersect and the end points are carbon atoms.
Answer to Problem 5.16EP
IUPAC name of the given carboxylic acid is 3,5-dimethylheptanoic acid.
Explanation of Solution
Given structure of carboxylic acid is,
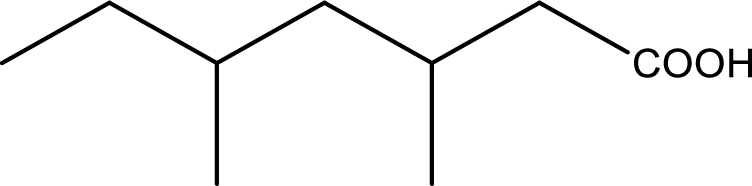
The longest continuous carbon chain has to be found out with the carboxyl group in it. In this it is a seven carbon chain. Hence, the parent alkane is heptane. The carboxylic acid is named by replacing the suffix “-e” in the alkane name with “-oic acid”. This gives the name of carboxylic acid as heptanoic acid.
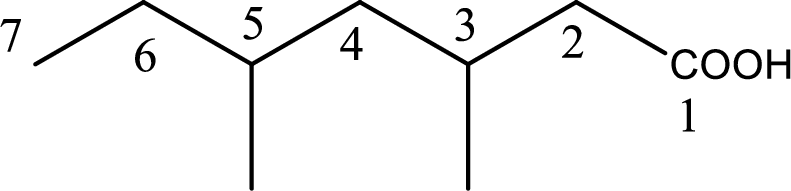
Looking for substituents it is found that there are two methyl groups present as substituents, each on third carbon atom and fifth carbon atom. Hence, the IUPAC name of the given carboxylic acid is 3,5-dimethylheptanoic acid.
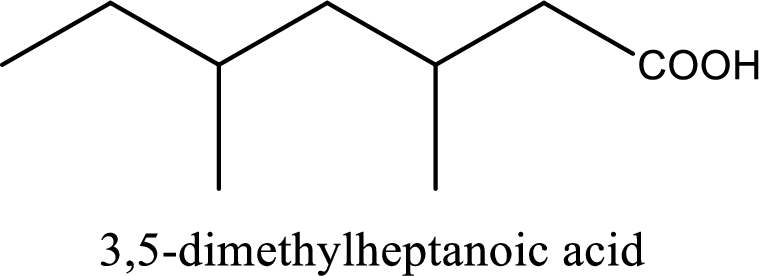
The IUPAC name of the given carboxylic acid is 3,5-dimethylheptanoic acid.
IUPAC name of the given carboxylic acid is found out.
Want to see more full solutions like this?
Chapter 5 Solutions
Organic And Biological Chemistry
- Q1: Answer the questions for the reaction below: ..!! Br OH a) Predict the product(s) of the reaction. b) Is the substrate optically active? Are the product(s) optically active as a mix? c) Draw the curved arrow mechanism for the reaction. d) What happens to the SN1 reaction rate in each of these instances: 1. Change the substrate to Br "CI 2. Change the substrate to 3. Change the solvent from 100% CH3CH2OH to 10% CH3CH2OH + 90% DMF 4. Increase the substrate concentration by 3-fold.arrow_forwardExperiment 27 hates & Mechanisms of Reations Method I visual Clock Reaction A. Concentration effects on reaction Rates Iodine Run [I] mol/L [S₂082] | Time mo/L (SCC) 0.04 54.7 Log 1/ Time Temp Log [ ] 13,20] (time) / [I] 199 20.06 23.0 30.04 0.04 0.04 80.0 22.8 45 40.02 0.04 79.0 21.6 50.08 0.03 51.0 22.4 60-080-02 95.0 23.4 7 0.08 0-01 1970 23.4 8 0.08 0.04 16.1 22.6arrow_forward(15 pts) Consider the molecule B2H6. Generate a molecular orbital diagram but this time using a different approach that draws on your knowledge and ability to put concepts together. First use VSEPR or some other method to make sure you know the ground state structure of the molecule. Next, generate an MO diagram for BH2. Sketch the highest occupied and lowest unoccupied MOs of the BH2 fragment. These are called frontier orbitals. Now use these frontier orbitals as your basis set for producing LGO's for B2H6. Since the BH2 frontier orbitals become the LGOS, you will have to think about what is in the middle of the molecule and treat its basis as well. Do you arrive at the same qualitative MO diagram as is discussed in the book? Sketch the new highest occupied and lowest unoccupied MOs for the molecule (B2H6).arrow_forward
- Q8: Propose an efficient synthesis of cyclopentene from cyclopentane.arrow_forwardQ7: Use compound A-D, design two different ways to synthesize E. Which way is preferred? Please explain. CH3I ONa NaOCH 3 A B C D E OCH3arrow_forwardPredict major product(s) for the following reactions. Note the mechanism(s) of the reactions (SN1, E1, SN2 or E2).arrow_forward
- (10 pts) The density of metallic copper is 8.92 g cm³. The structure of this metal is cubic close-packed. What is the atomic radius of copper in copper metal?arrow_forwardPredict major product(s) for the following reactions. Note the mechanism(s) of the reactions (SN1, E1, SN2 or E2).arrow_forwardPredict major product(s) for the following reactions. Note the mechanism(s) of the reactions (SN1, E1, SN2 or E2).arrow_forward
- Q3: Rank the following compounds in increasing reactivity of E1 and E2 eliminations, respectively. Br ca. go do A CI CI B C CI Darrow_forwardQ5: Predict major product(s) for the following reactions. Note the mechanism(s) of the reactions (SN1, E1, SN2 or E2). H₂O דיי "Br KN3 CH3CH2OH NaNH2 NH3 Page 3 of 6 Chem 0310 Organic Chemistry 1 HW Problem Sets CI Br excess NaOCH 3 CH3OH Br KOC(CH3)3 DuckDuckGarrow_forwardQ4: Circle the substrate that gives a single alkene product in a E2 elimination. CI CI Br Brarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

