
Chemistry in Context
8th Edition
ISBN: 9780073522975
Author: American Chemical Society
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 11.12, Problem 11.27YT
Interpretation Introduction
Interpretation:
The structural formula for methyl bromide has to be drawn and the reason for it to be deplete the ozone layer has to be given.
Concept introduction:
Ozone: It is an allotrope of oxygen with chemical formula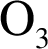 . Ozone is present in the stratosphere layer in the atmosphere, protecting the earth as a shield by absorbing ultra violet radiations from sun.
. Ozone is present in the stratosphere layer in the atmosphere, protecting the earth as a shield by absorbing ultra violet radiations from sun.
Ozone layer depletion is the destruction process of ozone layer by various chemicals mainly chlorofluorocarbons (CFC).
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please help me answer this question. I don't understand how or even if this can happen in a single transformation. Please provide a detailed explanation and a drawing showing how it can happen in a single transformation.
Add the necessary reagents and reaction conditions above and below the arrow in this organic reaction. If the products can't be made from the reactant with a single transformation, check the box under the drawing area instead.
2) Draw the correct chemical structure (using line-angle drawings / "line structures") from their
given IUPAC name:
a.
(E)-1-chloro-3,4,5-trimethylhex-2-ene
b. (Z)-4,5,7-trimethyloct-4-en-2-ol
C. (2E,6Z)-4-methylocta-2,6-diene
පිපිම
Draw curved arrows to represent the flow of electrons in the reaction on the left
Label the reactants on the left as either "Acid" or "Base"
(iii) Decide which direction the equilibrium arrows will point in each reaction, based on
the given pk, values
(a)
+
H-O
H
3-H
+
(c)
H"
H
+
H****H
000
44-00
NH₂
(e)
i
Дон
OH
Ө
NH
Chapter 11 Solutions
Chemistry in Context
Ch. 11.2 - You Decide A Lifetime of Food During your...Ch. 11.4 - Prob. 11.8CTCh. 11.4 - Prob. 11.10YTCh. 11.5 - Prob. 11.11YTCh. 11.6 - Prob. 11.12CTCh. 11.6 - Prob. 11.13CTCh. 11.7 - Prob. 11.14YTCh. 11.8 - Prob. 11.15CTCh. 11.8 - Prob. 11.16CTCh. 11.8 - Prob. 11.17CT
Ch. 11.8 - You Decide Sodium in Your Diet Compare the sodium...Ch. 11.8 - Prob. 11.19CTCh. 11.9 - Prob. 11.20SCCh. 11.9 - Prob. 11.21YTCh. 11.11 - Prob. 11.25YTCh. 11.12 - a. Ammonia (NH3) is applied to soil in the form of...Ch. 11.12 - Prob. 11.27YTCh. 11 - One theme in this chapter is that what you eat...Ch. 11 - Prob. 3QCh. 11 - Prob. 4QCh. 11 - Prob. 5QCh. 11 - Prob. 6QCh. 11 - Prob. 7QCh. 11 - Prob. 8QCh. 11 - Prob. 9QCh. 11 - Prob. 10QCh. 11 - Prob. 11QCh. 11 - Prob. 12QCh. 11 - Prob. 13QCh. 11 - Prob. 14QCh. 11 - Prob. 15QCh. 11 - Prob. 16QCh. 11 - Prob. 17QCh. 11 - Prob. 18QCh. 11 - Prob. 19QCh. 11 - Prob. 20QCh. 11 - Prob. 21QCh. 11 - Prob. 22QCh. 11 - Prob. 23QCh. 11 - Prob. 24QCh. 11 - Prob. 25QCh. 11 - Prob. 26QCh. 11 - Prob. 27QCh. 11 - Prob. 28QCh. 11 - Prob. 29QCh. 11 - Prob. 30QCh. 11 - Prob. 31QCh. 11 - Prob. 32QCh. 11 - Explain to a friend why it is impossible to go on...Ch. 11 - Prob. 34QCh. 11 - Prob. 35QCh. 11 - Prob. 36QCh. 11 - Prob. 37QCh. 11 - Prob. 38QCh. 11 - Prob. 39QCh. 11 - Here is the condensed structural formula for...Ch. 11 - Prob. 41QCh. 11 - Prob. 42QCh. 11 - Prob. 43QCh. 11 - Prob. 44QCh. 11 - Prob. 45QCh. 11 - Prob. 46QCh. 11 - Prob. 47QCh. 11 - Biodiesel is another example of a biofuel. Answer...Ch. 11 - Prob. 49QCh. 11 - This chapter (together with Section 5.15) provided...Ch. 11 - Prob. 52QCh. 11 - Prob. 53QCh. 11 - Consider this structural formula for one of the...Ch. 11 - Prob. 56QCh. 11 - Prob. 57QCh. 11 - Prob. 59QCh. 11 - Prob. 60Q
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 3) Label the configuration in each of the following alkenes as E, Z, or N/A (for non-stereogenic centers). 00 E 000 N/A E Br N/A N/A (g) E N/A OH E (b) Oz N/A Br (d) 00 E Z N/A E (f) Oz N/A E (h) Z N/Aarrow_forward6) Fill in the missing Acid, pKa value, or conjugate base in the table below: Acid HCI Approximate pK, -7 Conjugate Base H-C: Hydronium (H₂O') -1.75 H-O-H Carboxylic Acids (RCOOH) Ammonium (NH4) 9.24 Water (H₂O) H-O-H Alcohols (ROH) RO-H Alkynes R--H Amines 25 25 38 HOarrow_forward5) Rank the following sets of compounds in order of decreasing acidity (most acidic to least acidic), and choose the justification(s) for each ranking. (a) OH V SH я вон CH most acidic (lowst pKa) least acidic (highest pKa) Effect(s) Effect(s) Effect(s) inductive effect O inductive effect O inductive effect electronegativity electronegativity O electronegativity resonance polarizability resonance polarizability O resonance O polarizability hybridization Ohybridization O hybridization оarrow_forward
- How negatively charged organic bases are formed.arrow_forwardNonearrow_forward1) For the following molecules: (i) Label the indicated alkenes as either cis (Z), trans (E), or N/A (for non-stereogenic centers) by bubbling in the appropriate label on the molecule. (ii) Complete the IUPAC name located below the structure (HINT: Put the letter of the configuration in parentheses at the beginning of the name!) E z N/A ()-3,4,6-trimethylhept-2-ene E Oz O N/A ()-3-ethyl-1-fluoro-4-methylhex-3-ene E -+- N/A Me )-2,3-dimethylpent-2-ene (d) (b) E O N/A Br ()-5-bromo-1-chloro-3-ethyloct-4-ene ОЕ Z N/A Et (___)-3-ethyl-4-methylhex-3-ene E (f) Oz N/A z N/A HO (4.7)-4-(2-hydroxyethyl)-7-methylnona-4,7-dien-2-onearrow_forward
- O 9:21AM Tue Mar 4 ## 64% Problem 51 of 15 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. H :0: CI. AI :CI: :CI: Cl AI Select to Add Arrows Select to Add Arrows O: Cl :CI: :0: H CI: CI CO Select to Add Arrows Select to Add Arrows :O: CI :0: Cl. 10: AIarrow_forward(i) Draw in the missing lone pair(s) of electrons of the reactants on the left (ii) Draw (curved) arrows to show the flow of electrons in the acid/base reaction on the left (iii) Draw the products of the acid/base on the right (iv) Select the correct label for each product as either "conjugate acid" or "conjugate base" (a) JOH OH NH₂ acid base (b) De "H conjugate acid conjugate acid conjugate base conjugate base acid base conjugate acid conjugate base conjugate acid conjugate base acid basearrow_forwardCould someone answer this NMR and explain please Comment on the general features of the 1H-NMR spectrum of isoamyl ester provided below.arrow_forward
- Macmillan Learning Draw the acyl chloride that would give the ketone shown using the Friedel-Crafts acylation reaction. Select Draw Templates More с H о Cl 2Q Erase AICI₂arrow_forwardDraw the complete mechanism for this reaction: .OH مدید OH H2SO4 + H₂O To save you some time, the starting material has been copied into the first drawing area. However, you will still need to add any other reactants or catalysts that take part in the reaction. ན ི.. OH Add/Remove step Х ด ك Click and drag to start drawing a structure.arrow_forward9:27 AM Tue Mar 4 ← Problem 64 of 15 #63% Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. 0:0 0:0 :0: N. :0: :O :0: H H. :0: Select to Add Arrows O :0: H O :0: 0:0. S. H Select to Add Arrows S :0: :0: H Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning