
Study Guide with Selected Solutions for Stoker's General, Organic, and Biological Chemistry, 7th
7th Edition
ISBN: 9781305081086
Author: STOKER, H. Stephen
Publisher: Brooks Cole
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 25.9, Problem 3QQ
Interpretation Introduction
Interpretation:
The total numbers of molecules of acetyl CoA that are consumed in the production of a cholesterol molecule have to be determined.
Concept introduction:
Cholesterol is an important component of cell membranes. It is used as the precursor for sex hormones, bile salts, and adrenal hormones.
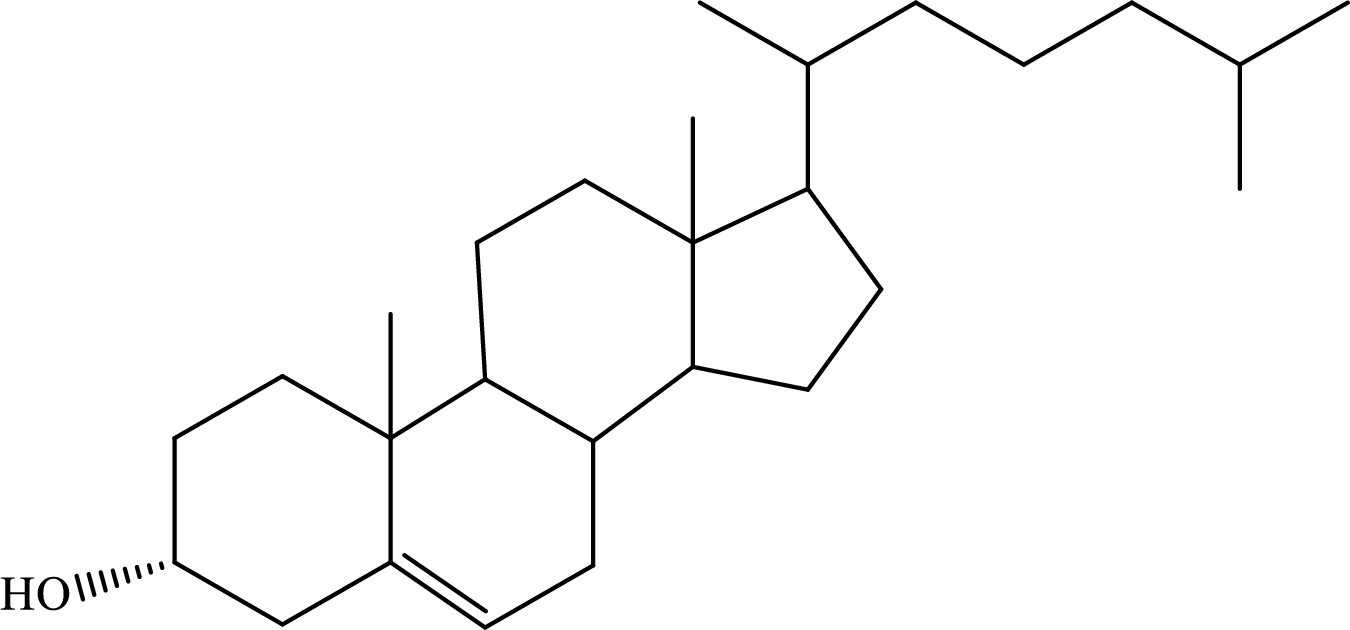
The biosynthesis of cholesterol occurs in the liver. It is a C27 molecule. The structure of cholesterol is as follows:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Lab Data
The distance entered is out of the expected range.
Check your calculations and conversion factors.
Verify your distance. Will the gas cloud be closer to the cotton ball with HCI or NH3?
Did you report your data to the correct number of significant figures?
- X
Experimental Set-up
HCI-NH3
NH3-HCI
Longer Tube
Time elapsed (min)
5 (exact)
5 (exact)
Distance between cotton balls (cm)
24.30
24.40
Distance to cloud (cm)
9.70
14.16
Distance traveled by HCI (cm)
9.70
9.80
Distance traveled by NH3 (cm)
14.60
14.50
Diffusion rate of HCI (cm/hr)
116
118
Diffusion rate of NH3 (cm/hr)
175.2
175.2
How to measure distance and calculate rate
For the titration of a divalent metal ion (M2+) with EDTA, the stoichiometry of the reaction is typically:
1:1 (one mole of EDTA per mole of metal ion)
2:1 (two moles of EDTA per mole of metal ion)
1:2 (one mole of EDTA per two moles of metal ion)
None of the above
Please help me solve this reaction.
Chapter 25 Solutions
Study Guide with Selected Solutions for Stoker's General, Organic, and Biological Chemistry, 7th
Ch. 25.1 - Which of the following statements about digestion...Ch. 25.1 - Prob. 2QQCh. 25.1 - The major function of bile released during...Ch. 25.1 - The two major products of triacylglycerol...Ch. 25.1 - Prob. 5QQCh. 25.2 - Hormone-sensitive lipase needed for...Ch. 25.2 - Prob. 2QQCh. 25.2 - Which of the following is not a product of...Ch. 25.3 - Prob. 1QQCh. 25.3 - What is the intermediate compound in the two-step...
Ch. 25.3 - Prob. 3QQCh. 25.4 - Prob. 1QQCh. 25.4 - Prob. 2QQCh. 25.4 - Prob. 3QQCh. 25.4 - Prob. 4QQCh. 25.4 - Prob. 5QQCh. 25.4 - Prob. 6QQCh. 25.5 - Prob. 1QQCh. 25.5 - Prob. 2QQCh. 25.5 - Prob. 3QQCh. 25.6 - Prob. 1QQCh. 25.6 - Prob. 2QQCh. 25.6 - Prob. 3QQCh. 25.6 - Prob. 4QQCh. 25.6 - Prob. 5QQCh. 25.6 - Prob. 6QQCh. 25.7 - Prob. 1QQCh. 25.7 - Prob. 2QQCh. 25.7 - Prob. 3QQCh. 25.7 - Prob. 4QQCh. 25.7 - The reducing agent needed in the process of...Ch. 25.7 - Prob. 6QQCh. 25.8 - Prob. 1QQCh. 25.8 - Prob. 2QQCh. 25.9 - Prob. 1QQCh. 25.9 - Prob. 2QQCh. 25.9 - Prob. 3QQCh. 25.9 - Prob. 4QQCh. 25.10 - Which of the following substances cannot be...Ch. 25.10 - Prob. 2QQCh. 25.10 - Which of the following processes occurs within the...Ch. 25.11 - Prob. 1QQCh. 25.11 - Prob. 2QQCh. 25.11 - Prob. 3QQCh. 25 - Indicate whether each of the following aspects of...Ch. 25 - Indicate whether each of the following aspects of...Ch. 25 - Indicate whether each of the following pairings of...Ch. 25 - Prob. 25.4EPCh. 25 - Indicate whether each of the following statements...Ch. 25 - Prob. 25.6EPCh. 25 - Prob. 25.7EPCh. 25 - What is a chylomicron?Ch. 25 - What are the products of the complete hydrolysis...Ch. 25 - What are the major products of the incomplete...Ch. 25 - Prob. 25.11EPCh. 25 - At what location are free fatty acids and...Ch. 25 - Prob. 25.13EPCh. 25 - Prob. 25.14EPCh. 25 - Prob. 25.15EPCh. 25 - Prob. 25.16EPCh. 25 - Prob. 25.17EPCh. 25 - Prob. 25.18EPCh. 25 - Prob. 25.19EPCh. 25 - Prob. 25.20EPCh. 25 - Prob. 25.21EPCh. 25 - Prob. 25.22EPCh. 25 - Prob. 25.23EPCh. 25 - Prob. 25.24EPCh. 25 - Prob. 25.25EPCh. 25 - Prob. 25.26EPCh. 25 - Prob. 25.27EPCh. 25 - Identify the oxidizing agent needed in Step 3 of a...Ch. 25 - Prob. 25.29EPCh. 25 - Prob. 25.30EPCh. 25 - Prob. 25.31EPCh. 25 - Prob. 25.32EPCh. 25 - Prob. 25.33EPCh. 25 - Prob. 25.34EPCh. 25 - Prob. 25.35EPCh. 25 - Prob. 25.36EPCh. 25 - Prob. 25.37EPCh. 25 - Prob. 25.38EPCh. 25 - Prob. 25.39EPCh. 25 - Prob. 25.40EPCh. 25 - Prob. 25.41EPCh. 25 - Prob. 25.42EPCh. 25 - How many turns of the -oxidation pathway would be...Ch. 25 - How many turns of the -oxidation pathway would be...Ch. 25 - Prob. 25.45EPCh. 25 - Prob. 25.46EPCh. 25 - Prob. 25.47EPCh. 25 - Prob. 25.48EPCh. 25 - Prob. 25.49EPCh. 25 - Explain why fatty acids cannot serve as fuel for...Ch. 25 - Prob. 25.51EPCh. 25 - Prob. 25.52EPCh. 25 - Prob. 25.53EPCh. 25 - Prob. 25.54EPCh. 25 - Prob. 25.55EPCh. 25 - Prob. 25.56EPCh. 25 - Prob. 25.57EPCh. 25 - Prob. 25.58EPCh. 25 - Prob. 25.59EPCh. 25 - Prob. 25.60EPCh. 25 - Prob. 25.61EPCh. 25 - Why does a deficiency of carbohydrates in the diet...Ch. 25 - Prob. 25.63EPCh. 25 - Prob. 25.64EPCh. 25 - Prob. 25.65EPCh. 25 - Prob. 25.66EPCh. 25 - Prob. 25.67EPCh. 25 - Prob. 25.68EPCh. 25 - Prob. 25.69EPCh. 25 - Prob. 25.70EPCh. 25 - Prob. 25.71EPCh. 25 - Prob. 25.72EPCh. 25 - Prob. 25.73EPCh. 25 - Prob. 25.74EPCh. 25 - Prob. 25.75EPCh. 25 - Severe ketosis situations produce acidosis....Ch. 25 - Prob. 25.77EPCh. 25 - Prob. 25.78EPCh. 25 - Prob. 25.79EPCh. 25 - Prob. 25.80EPCh. 25 - Prob. 25.81EPCh. 25 - Prob. 25.82EPCh. 25 - Prob. 25.83EPCh. 25 - Prob. 25.84EPCh. 25 - Prob. 25.85EPCh. 25 - Prob. 25.86EPCh. 25 - Prob. 25.87EPCh. 25 - Prob. 25.88EPCh. 25 - Prob. 25.89EPCh. 25 - Prob. 25.90EPCh. 25 - Prob. 25.91EPCh. 25 - Prob. 25.92EPCh. 25 - Prob. 25.93EPCh. 25 - Prob. 25.94EPCh. 25 - What role does molecular oxygen, O2, play in fatty...Ch. 25 - Prob. 25.96EPCh. 25 - Prob. 25.97EPCh. 25 - Prob. 25.98EPCh. 25 - Prob. 25.99EPCh. 25 - Prob. 25.100EPCh. 25 - Prob. 25.101EPCh. 25 - Prob. 25.102EPCh. 25 - Prob. 25.103EPCh. 25 - Prob. 25.104EPCh. 25 - Prob. 25.105EPCh. 25 - Prob. 25.106EPCh. 25 - Prob. 25.107EPCh. 25 - Prob. 25.108EPCh. 25 - Prob. 25.109EPCh. 25 - Prob. 25.110EPCh. 25 - Prob. 25.111EPCh. 25 - Prob. 25.112EPCh. 25 - Prob. 25.113EPCh. 25 - Prob. 25.114EP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forwardSynthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIf possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forward
- Synthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forward
- Indicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,