
Concept explainers
a)
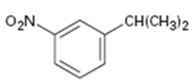
Interpretation:
The
Concept introduction:
The benzene ring is inert to even strong oxidizing agents like KMnO4 and Na2Cr2O7. However the alkyl side chains with benzylic hydrogen atoms are readily oxidized by these reagents into carboxyl groups. Compounds having no benzylic hydrogen atoms in the side chain are not oxidized.
To give:
The aromatic substance obtained as the product, when m-nitroisopropyl benzene is oxidized with KMnO4.
b)
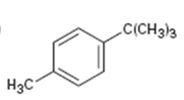
Interpretation:
The aromatic substance obtained as the product, when p-tert-butyltoluene is oxidized with KMnO4, is to be given.
Concept introduction:
The benzene ring is inert to even strong oxidizing agents like KMnO4 and Na2Cr2O7. However the alkyl side chains with benzylic hydrogen atoms are readily oxidized by these reagents into carboxyl groups. Compounds having no benzylic hydrogen atoms in the side chain are not oxidized.
To explain:
The aromatic substance obtained as the product when p-tert-butyl toluene is oxidized with KMnO4.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- 9. Consider the following galvanic cell: Fe (s) | Fe(NO3)2 (aq) || Sn(NO3)2 (aq) | Sn (s) a. Write an equation for the half reactions occurring at the anode and cathode. b. Calculate the standard cell potential Show all of your work. c. Draw and label the galvanic cell, including the anode and cathode, direction of electron flow, and direction of ion migration.arrow_forwardpls help asaparrow_forward11. Use the equation below to answer the following questions: 2 Al(s) + 3 Cd(NO3)2 (aq) → 2 Al(NO3)3 (aq) + 3 Cd(s) a. What is the net ionic equation for the reaction? b. Which species is a spectator ion in this reaction? Define a spectator ion. c. Identify the oxidizing agent and the reducing agent.arrow_forward
- Label each of the seven designated regions of the following multi-component, solid-liquid phase diagram for the Zinc - Magnesium system.arrow_forward22arrow_forwardPLEASE READ!!! I DONT WANT EXAMPLES, I DONT WANT WORDS OR PARAGRAPHS FOR THE MECHANISM!!! THANKS First image: QUESTION 6. I have to show, with ARROWS and STRUCTURES, the mechanism of the reaction at the bottom. Also I have to show by mecanism why the reaction wouldn't work if the alcohol was primary. I also tried to draw the mechanism, tell me what to change. Please note that its an AMIDE thats formed not an AMINE the nitrogen has ONE hydrogen and one Phenyl-C-Phenyl. I already asked for this mechanism and got as a final product ...-NH2 not whats shown on the picture, thank you Ths second part. QUESTION 3. I just need a way to synthesize the lactone A, I already started please continue from where I left it Second image: I simply need the products, substrates or reagents, thank youarrow_forward
- Indicate how to prepare a 10% sodium hydroxide (NaOH) solution to a slightly alkaline pH.arrow_forwardCH, CH CH₂ CH₂ Phytyl side chain 5. What is the expected order of elution of compounds A-D below from a chromatography column packed with silica gel, eluting with hexane/ethyl acetate? C D OHarrow_forwardPlease analze my gel electrophoresis column of the VRK1 kinase (MW: 39.71 kDa). Attached is the following image for the order of column wells and my gel.arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

