
Concept explainers
a)
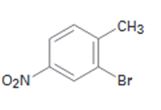
Interpretation:
Starting from benzene and assuming that the ortho and para isomers can be separated how the compound can be synthesized is to be shown.
Concept introduction:
The compound required is 2-bromo-4-nitrotoluene. Benzene can be converted into alkyl benzene by Friedal-Crafts alkylation. The alkylbenzene can be nitrated in the ortho and para positions as the alkyl groups are ortho and para directors. Nitro group is deactivating while alkyl group is activating in nature. Hence bromonation of the nitrotoluene will introduce the bromine in the ortho position to alkyl group.
To show:
Starting from benzene and assuming that the ortho and para isomers can be separated how 2-bromo-4-nitrotoluene can be synthesized.
b)
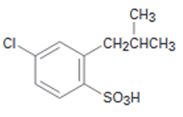
Interpretation:
Starting from benzene and assuming that the ortho and para isomers can be separated how the compound can be synthesized is to be shown.
Concept introduction:
The Friedal-Crafts acylation of benzene will yield an acyl benzene as the product. Chlorination of the acyl benzene will produce m-chloroacyl benzene as the acyl group is meta directing. Reduction of the acyl group to the corresponding alkyl group and sulfonation of the product will result in the product required.
To show:
Starting from benzene and assuming that the ortho and para isomers can be separated how the compound can be synthesized.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- Draw the products of this reduction of a ketone with sodium borohydride. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts. 1) NaBH4 2) HCI/H2O Select to Drawarrow_forwardWhy do you think people who live at high altitudes are advised to add salt to water when boiling food like pasta? What mole fraction of NaCl is needed to raise the boiling point of H2O by 3˚C? Does the amount of salt added to water (typically about one teaspoon to four quarts of water) substantially change the boiling point? (Kb (H2O) = 0.51˚C/molal.)arrow_forwardpls help asaparrow_forward
- pls help asaparrow_forward9. Consider the following galvanic cell: Fe (s) | Fe(NO3)2 (aq) || Sn(NO3)2 (aq) | Sn (s) a. Write an equation for the half reactions occurring at the anode and cathode. b. Calculate the standard cell potential Show all of your work. c. Draw and label the galvanic cell, including the anode and cathode, direction of electron flow, and direction of ion migration.arrow_forwardpls help asaparrow_forward
- 11. Use the equation below to answer the following questions: 2 Al(s) + 3 Cd(NO3)2 (aq) → 2 Al(NO3)3 (aq) + 3 Cd(s) a. What is the net ionic equation for the reaction? b. Which species is a spectator ion in this reaction? Define a spectator ion. c. Identify the oxidizing agent and the reducing agent.arrow_forwardpls help asaparrow_forwardpls help asaparrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

