
Concept explainers
a)
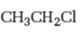
Interpretation:
Whether ethyl chloride is expected to undergo Friedal-Crafts reaction with or without rearrangement is to be stated and explained.
Concept introduction:
Friedal-Crafts reaction is an electrophilic substitution reaction in which an
To state and explain:
Whether ethyl chloride is expected to undergo Friedal- Crafts reaction with or without rearrangement.
b)
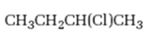
Interpretation:
Whether 2-chlorobutane is expected to undergo Friedal-Crafts reaction with or without rearrangement is to be stated and explained.
Concept introduction:
Friedal-Crafts reaction is an electrophilic substitution reaction in which an aromatic compound reacts with an alkyl chloride in the presence of AlCl3 to produce an alkylbenzene. The electrophile in this reaction is an alkyl carbocation. The skeletal rearrangement of the alkyl carbocation electrophile either through a hydride shift or alkyl shift (particularly when a primary alkyl halide is used) can lead to the formation of rearranged product also in this reaction.
To state and explain:
Whether 2-chlorobutane is expected to undergo Friedal-Crafts reaction with or without rearrangement.
c)
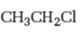
Interpretation:
Whether n-propyl chloride is expected to undergo Friedal-Crafts reaction with or without rearrangement is to be stated and explained.
Concept introduction:
Friedal-Crafts reaction is an electrophilic substitution reaction in which an aromatic compound reacts with an alkyl chloride in the presence of AlCl3 to produce an alkylbenzene. The electrophile in this reaction is an alkyl carbocation. The skeletal rearrangement of the alkyl carbocation electrophile either through a hydride shift or alkyl shift, particularly when a primary alkyl halide is used, can lead to the formation of rearranged product also in this reaction.
To state and explain:
Whether n-propyl chloride is expected to undergo Friedal-Crafts reaction with or without rearrangement.
d)
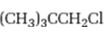
Interpretation:
Whether 1-chloro-2, 2-dimethylpropane is expected to undergo Friedal-Crafts reaction with or without rearrangement is to be stated and explained.
Concept introduction:
Friedal-Crafts reaction is an electrophilic substitution reaction in which an aromatic compound reacts with an alkyl chloride in the presence of AlCl3 to produce an alkylbenzene. The electrophile in this reaction is an alkyl carbocation. The skeletal rearrangement of the alkyl carbocation electrophile either through a hydride shift or alkyl shift, particularly when a primary alkyl halide is used, can lead to the formation of rearranged product also in this reaction.
To state and explain:
Whether 1-chloro-2, 2-dimethylpropane is expected to undergo Friedal-Crafts reaction with or without rearrangement.
e)
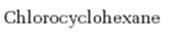
Interpretation:
Whether chlorocyclohexane is expected to undergo Friedal Crafts reaction with or without rearrangement is to be stated and explained.
Concept introduction:
Friedal-Crafts reaction is an electrophilic substitution reaction in which an aromatic compound reacts with an alkyl chloride in the presence of AlCl3 to yield an alkylbenzene. The electrophile in this reaction is an alkyl carbocation. The skeletal rearrangement of the alkyl carbocation electrophile either through a hydride shift or alkyl shift, particularly when a primary alkyl halide is used, can lead to the formation of rearranged product also in this reaction.
To state and explain:
Whether chlorocyclohexane is expected to undergo Friedal-Crafts reaction with or without rearrangement.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- Draw the mechanism (including all curved arrows for electron movement) showing how the maleicanhydride is attacked by the anthracene and formation of the final Diels Alder product.arrow_forwardProvide the missing information. *see imagearrow_forwardProvide the missing information. *see imagearrow_forward
- Provide the missing information. *see imagearrow_forwardI have a bottle of butanal that has been improperly used by lab workers. They allowed a traceamount NaOH (aq) to contaminate the bottle. What is now in my bottle of “butanal? What is the molecular name and functional group name? Draw the structure.arrow_forwardProvide the missing information. *see imagearrow_forward
- First image: Why can't the molecule C be formed in those conditions Second image: Synthesis for lactone C its not an examarrow_forwardFirst image: I have to show the mecanism for the reaction on the left, where the alcohol A is added fast in one portion Second image: I have to show the mecanism of the reaction at the bottom. Also I have to show by mecanism why the reaction wouldn't work if the alcohol was primaryarrow_forwardFirst image: I have to explain why the molecule C is never formed in those conditions. Second image: I have to propose a synthesis for the lactone Aarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





