
a)
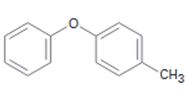
Interpretation:
The position and the ring in which the compound shown is expected to undergo electrophilic substitution is to be stated.
Concept introduction:
In
To state:
The position and the ring in which the compound shown is expected to undergo electrophilic substitution.
b)
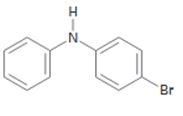
Interpretation:
The position and the ring in which the compound shown is expected to undergo electrophilic substitution is to be stated.
Concept introduction:
In aromatic electrophilic substitution reactions both -NH- and halogens are ortho and para directing as they stabilize the carbocation intermediates for these attacks. If two alternate options exist, the electrophile will enter into the more activated ring. The orientation of the incoming electrophile will be decided by the stronger of the two substituent groups already present.
To state:
The position and the ring in which the compound shown is expected to undergo electrophilic substitution.
c)
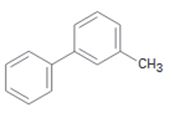
Interpretation:
The position and the ring in which the compound shown is expected to undergo electrophilic substitution is to be stated.
Concept introduction:
In aromatic electrophilic substitution reactions, both aryl and alkyl are ortho and para directing as they stabilize the carbocation intermediates for these attacks. If two alternate options exist, the electrophile will enter into the more activated ring. The orientation of the incoming electrophile will be decided by the stronger of the two substituent groups already present.
To state:
The position and the ring in which the compound shown is expected to undergo electrophilic substitution.
d)
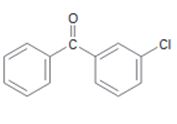
Interpretation:
The position and the ring in which the compound shown is expected to undergo electrophilic substitution is to be stated.
Concept introduction:
In aromatic electrophilic substitution reactions –C=O group is meta directing while halogens are ortho and para directing. If two alternate options exist the electrophile will enter into the more activated ring. The orientation of the incoming electrophile will be decided by the stronger of the two substituent groups already present.
To state:
The position and the ring in which the compound shown is expected to undergo electrophilic substitution.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- How can you prepare a 6 mL solution of 6% H2O2, if we have a bottle of 30% H2O2?arrow_forwardHow many mL of H2O2 from the 30% bottle must be collected to prepare 6 mL of 6% H2O2.arrow_forwardIndicate the product(s) B and C that are formed in the reaction: HN' OCH HC1 B + mayoritario C minoritario OCH3arrow_forward
- Indicate the product(s) that are formed in the reaction: NH-NH, OCH3 -H₂O OCH3arrow_forward21.38 Arrange the molecules in each set in order of increasing acidity (from least acidic to most acidic). OH OH SH NH2 8 NH3 OH (b) OH OH OH (c) & & & CH3 NO2 21.39 Explain the trends in the acidity of phenol and the monofluoro derivatives of phenol. OH OH OH OH PK 10.0 PK 8.81 PK 9.28 PK 9.81arrow_forwardidentify which spectrum is for acetaminophen and which is for phenacetinarrow_forward
- The Concept of Aromaticity 21.15 State the number of 2p orbital electrons in each molecule or ion. (a) (b) (e) (f) (c) (d) (h) (i) DA (k) 21.16 Which of the molecules and ions given in Problem 21.15 are aromatic according to the Hückel criteria? Which, if planar, would be antiaromatic? 21.17 Which of the following structures are considered aromatic according to the Hückel criteria? ---0-0 (a) (b) (c) (d) (e) (h) H -H .8.0- 21.18 Which of the molecules and ions from Problem 21.17 have electrons donated by a heteroatom?arrow_forward1. Show the steps necessary to make 2-methyl-4-nonene using a Wittig reaction. Start with triphenylphosphine and an alkyl halide. After that you may use any other organic or inorganic reagents. 2. Write in the product of this reaction: CH3 CH₂ (C6H5)₂CuLi H₂O+arrow_forward3. Name this compound properly, including stereochemistry. H₂C H3C CH3 OH 4. Show the step(s) necessary to transform the compound on the left into the acid on the right. Bri CH2 5. Write in the product of this LiAlH4 Br H₂C OHarrow_forward
- What are the major products of the following reaction? Please provide a detailed explanation and a drawing to show how the reaction proceeds.arrow_forwardWhat are the major products of the following enolate alkylation reaction? Please include a detailed explanation as well as a drawing as to how the reaction proceeds.arrow_forwardA block of zinc has an initial temperature of 94.2 degrees celcius and is immererd in 105 g of water at 21.90 degrees celcius. At thermal equilibrium, the final temperature is 25.20 degrees celcius. What is the mass of the zinc block? Cs(Zn) = 0.390 J/gxdegrees celcius Cs(H2O) = 4.18 J/gx degrees celcusarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


