
Concept explainers
a)

Interpretation:
A structure for the molecule which meets the descriptions given is to be assigned.
C6H12O; IR: 1715 cm-1; 13CNMR: 8.0 δ (10), 18.5 δ (10), 33.5 δ (20), 40.6 δ (30), 214.0 δ (40)
Concept introduction:
In 13CNMR, carbons in double bond which are sp2 hybridized absorb from 110 to 220 δ. Saturated aldehydes and ketones usually absorb in the region from 200 to 215 δ While aromatic and unsaturated carbonyl compounds absorb in the 190 to 200 δ region. The primary alkyl carbon absorbs in the range 10-15 δ, a secondary alkyl radical in the range 16-25 δ while a tertiary alkyl in the range 25-35 δ. The signals will be observed at downfield if these alkyl groups are attached to electron withdrawing groups.
To assign:
A structure for the molecule which meets the descriptions given.
C6H12O; IR: 1715 cm-1; 13CNMR: 8.0 δ (10), 18.5 δ (10), 33.5 δ (20), 40.6 δ (30), 214.0 δ (40)
Answer:
Answer to Problem 69AP
A structure for the molecule which meets the spectral descriptions given is
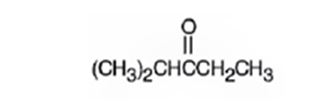
Explanation of Solution
The molecular formula of the compound is C6H12O. Ignoring oxygen the molecular formula of the parent compound becomes C6H14. Thus the compound has two hydrogen atoms (14-12) less than the parent and hence has one unsaturation unit perhaps a C=O.
The IR absorption at 1715 cm-1 and 13CNMR peak at 214.0 δ indicate that the compound is a ketone. The 13CNMR further indicates that the carbonyl group is flanked by a secondary and tertiary carbon indicated by the two downfield signals at 33.5 δ and 40.6 δ. The remaining three carbons has to be primary, of which one is attached as methyl(8.0 δ) to the secondary carbon and two others are attached as two methyl groups (18.5δ) on the tertiary carbon. Hence the compound is 2-methyl-3-pentanone.
A structure for the molecule which meets the spectral descriptions given is

b)

Interpretation:
A structure for the molecule which meets the descriptions given is to be assigned.
C5H10O; IR: 1730 cm-1; 13CNMR: 22.6 δ (10), 23.6 δ (30), 52.8 δ (20), 202.4 δ (30).
Concept introduction:
Aldehydes and ketones show a strong absorption band in IR from 1660-1770 cm-1. Aldehydes show two characteristic C-H absorptions between 2700-2760cm-1 and 2800-2860 cm-1. Saturated aldehydes absorb near 1730 cm-1 while aromatic aldehydes and α, β- unsaturated aldehydes absorb near 1705 cm-1. Saturated ketones and cyclohexanones absorb near 1715 cm-1 while aromatic ketones and α, β- unsaturated ketones absorb near 1685-1690 cm-1. Cyclopentanones absorb around 1750 cm-1.
In 13CNMR, carbons in double bond which are sp2 hybridized absorb from 110 to 220 δ. Saturated aldehydes and ketones usually absorb in the region from 200 to 215 δ While aromatic and unsaturated carbonyl compounds absorb in the 190 to 200 δ region. The primary alkyl carbon absorbs in the range 10-15 δ, a secondary alkyl radical in the range 16-25 δ while a tertiary alkyl in the range 25-35 δ. The signals will be observed at downfield if these alkyl groups are attached to electron withdrawing groups.
To assign:
A structure for the molecule which meets the descriptions given.
C5H10O; IR: 1730 cm-1; 13CNMR: 22.6 δ (10), 23.6 δ (30), 52.8 δ (20), 202.4 δ (30).
Answer to Problem 69AP
A structure for the molecule which meets the spectral descriptions given is

Explanation of Solution
The molecular formula of the compound is C6H12O. Ignoring oxygen the molecular formula of the parent compound becomes C6H14. Thus the compound has two hydrogen atoms less than the parent and hence has one unsaturation unit, a carbonyl group.
The IR absorption at 1730 cm-1 and 13CNMR peak at 202.4 δ indicate that the compound is a saturated aldehyde. The 13CNMR further indicates that the carbonyl group is attached to a secondary carbon indicated by the downfield signal at 52.8 δ. Of the remaining three carbons one has to be tertiary(23.6 δ) which is attached to two methyl groups(22.6 δ). Hence the compound is 2-methylbutanal.
A structure for the molecule which meets the spectral descriptions given is

c)
Interpretation:
A structure for the molecule which meets the descriptions given is to be assigned.
C6H8O; IR: 1680 cm-1; 13CNMR: 22.9 δ (20), 25.8 δ (20), 38.2 δ (20), 129.8 δ (30) 150.6 δ (30), 198.7 δ (40).
Concept introduction:
Aldehydes and ketones show a strong absorption band in IR from 1660-1770 cm-1. Aldehydes show two characteristic C-H absorptions between 2700-2760cm-1 and 2800-2860 cm-1. Saturated aldehydes absorb near 1730 cm-1 while aromatic aldehydes and α, β- unsaturated aldehydes absorb near 1705 cm-1. Saturated ketones and cyclohexanones absorb near 1715 cm-1 while aromatic ketones and α, β- unsaturated ketones absorb near 1685-1690 cm-1. Cyclopentanones absorb around 1750 cm-1.
In 13CNMR, carbons in double bond which are sp2 hybridized absorb from 110 to 220 δ. Saturated aldehydes and ketones usually absorb in the region from 200 to 215 δ While aromatic and unsaturated carbonyl compounds absorb in the 190 to 200 δ region. The primary alkyl carbon absorbs in the range 10-15 δ, a secondary alkyl radical in the range 16-25 δ while a tertiary alkyl in the range 25-35 δ.
To assign:
A structure for the molecule which meets the descriptions given.
C6H8O; IR: 1680 cm-1; 13CNMR: 22.9 δ (20), 25.8 δ (20), 38.2 δ (20), 129.8 δ (30) 150.6 δ (30), 198.7 δ (40).
Answer to Problem 69AP
A structure for the molecule which meets the spectral descriptions given is
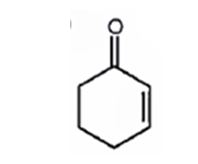
Explanation of Solution
The molecular formula of the compound is C6H8O. Ignoring oxygen the molecular formula of the parent compound becomes C6H14. Thus the compound has six hydrogen atoms (14-8= 6) less than the parent and hence has three unsaturation units.
The IR absorption at 1680 cm-1 and 13CNMR peak at 198.7 δ indicate that the compound is an α, β- unsaturatedketone. The compound has three unsaturated units. α, β- unsaturatedketone accounts for two unsaturation units. Hence the compound contains a six membered ring. The signals at 129.8 δ (30) 150.6 δ (30) can be attributed to the carbons in double bond. The other three signals at 22.9 δ (20), 25.8 δ (20), 38.2 δ (20) can be attributed to three methylene groups. The signal at 38.2 δ (20) can be assigned to methylene carbon attached to carbonyl carbon, that at 25.8 δ (20) to the methylene carbon carbon attached to C4 while that at 22.9 δ (20) is due to methylene carbon C5.
A structure for the molecule which meets the spectral descriptions given is

Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry
- [Review Topics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. The IUPAC name is Submit Answer Retry Entire Group 9 more group attempts remainingarrow_forwardPlease draw.arrow_forwardA chromatogram with ideal Gaussian bands has tR = 9.0 minutes and w1/2 = 2.0 minutes. Find the number of theoretical plates that are present, and calculate the height of each theoretical plate if the column is 10 centimeters long.arrow_forward
- An open tubular column has an inner diameter of 207 micrometers, and the thickness of the stationary phase on the inner wall is 0.50 micrometers. Unretained solute passes through in 63 seconds and a particular solute emerges at 433 seconds. Find the distribution constant for this solute and find the fraction of time spent in the stationary phase.arrow_forwardConsider a chromatography column in which Vs= Vm/5. Find the retention factor if Kd= 3 and Kd= 30.arrow_forwardTo improve chromatographic separation, you must: Increase the number of theoretical plates on the column. Increase the height of theoretical plates on the column. Increase both the number and height of theoretical plates on the column. Increasing the flow rate of the mobile phase would Increase longitudinal diffusion Increase broadening due to mass transfer Increase broadening due to multiple paths You can improve the separation of components in gas chromatography by: Rasing the temperature of the injection port Rasing the temperature of the column isothermally Rasing the temperature of the column using temperature programming In GC, separation between two different solutes occurs because the solutes have different solubilities in the mobile phase the solutes volatilize at different rates in the injector the solutes spend different amounts of time in the stationary phasearrow_forward
- please draw and example of the following: Show the base pair connection(hydrogen bond) in DNA and RNAarrow_forwardNaming and drawing secondary Write the systematic (IUPAC) name for each of the following organic molecules: CH3 Z structure CH3 CH2 CH2 N-CH3 CH3-CH2-CH2-CH-CH3 NH CH3-CH-CH2-CH2-CH2-CH2-CH2-CH3 Explanation Check ☐ name ☐ 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C Garrow_forwardC This question shows how molecular orbital (MO) theory can be used to understand the chemical properties of elemental oxygen O₂ and its anionic derivative superoxide Oz. a) Draw the MO energy diagram for both O2 and O2. Clearly label your diagram with atomic orbital names and molecular orbital symmetry labels and include electrons. Draw the Lewis structure of O2. How does the MO description of O2 differ from the Lewis structure, and how does this difference relate to the high reactivity and magnetic properties of oxygen? ) Use the MO diagram in (a) to explain the difference in bond length and bond energy between superoxide ion (Oz, 135 pm, 360 kJ/mol) and oxygen (O2, 120.8 pm, 494 kJ/mol).arrow_forward
- Please drawarrow_forward-Page: 8 nsition metal ions have high-spin aqua complexes except one: [Co(HO)₁]". What is the d-configuration, oxidation state of the metal in [Co(H:O))"? Name and draw the geometry of [Co(H2O)]? b) Draw energy diagrams showing the splitting of the five d orbitals of Co for the two possible electron configurations of [Co(H2O)]: Knowing that A = 16 750 cm and Пl. = 21 000 cm, calculate the configuration energy (.e., balance or ligand-field stabilization energy and pairing energy) for both low spin and high spin configurations of [Co(H2O)]. Which configuration seems more stable at this point of the analysis? (Note that 349.76 cm = 1 kJ/mol) Exchange energy (IT) was not taken into account in part (d), but it plays a role. Assuming exchange an occur within t29 and within eg (but not between tz, and ea), how many exchanges are possible in the low in configuration vs in the high spin configuration? What can you say about the importance of exchange energy 07arrow_forwardDraw everything please on a piece of paper explaining each steparrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

