
(a)
Interpretation: The four structures having same physical properties like boiling point, melting point, density etc needs to be determined.
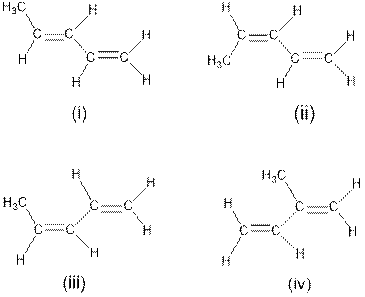
Concept Introduction : Cis-trans isomerism is also known as Configurational or geometrical isomerism. The prefixes cis and trans are Latin words in which ‘‘cis’’ represents ‘‘this side of’’ and ‘‘trans’’ represents ‘‘the other side’’. This type of isomerism occurs in unsaturated compounds (i.e. double bonds) or cyclic compounds depending on the presence of substituted groups in a molecule.
(b)
Interpretation: The structure from the given four structures which are trans isomers needs to be determined.
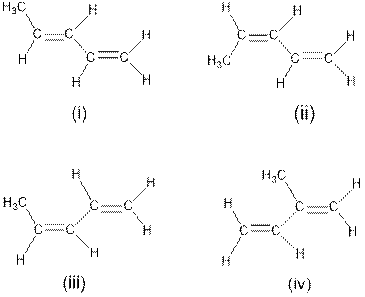
Concept Introduction : Cis-trans isomerism is also known as Configurational or geometrical isomerism. The prefixes cis and trans are Latin words in which ‘‘cis’’ represents ‘‘this side of’’ and ‘‘trans’’ represents ‘‘the other side’’. This type of isomerism occurs in unsaturated compounds (i.e. double bonds) or cyclic compounds depending on the presence of substituted groups in a molecule.
(c)
Interpretation: The structures which do not possess cis-trans isomerism needs to be determined.
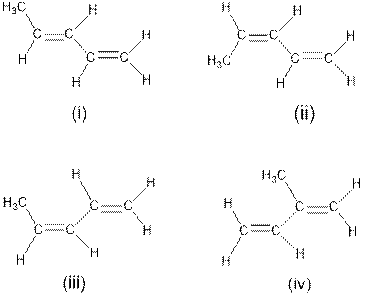
Concept Introduction : Cis-trans isomerism is also known as configurational or geometrical isomerism. The prefixes cis and trans are Latin words in which ‘‘cis’’ represents ‘‘this side of’’ and ‘‘trans’’ represents ‘‘the other side’’. This type of isomerism occurs in unsaturated compounds (i.e. double bonds) or cyclic compounds depending on the presence of substituted groups in a molecule.
Trending nowThis is a popular solution!

Chapter 21 Solutions
Chemical Principles
- Synthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIf possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forwardSynthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Indicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forwardIndicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forward
- Indicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forwardQuestion 3 (4 points), Draw a full arrow-pushing mechanism for the following reaction Please draw all structures clearly. Note that this intramolecular cyclization is analogous to the mechanism for halohydrin formation. COH Br + HBr Brarrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





