
Concept explainers
(a)
Interpretation:
The citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid intermediate butyrate has to be determined.
Concept introduction:
Lipogenesis is the process employed for the synthesis of fatty acid. The starting precursor for the synthesis is acetyl CoA. The enzyme employed for the process is fatty acid synthase. It is a multienzyme complex that ties the reaction responsible for the synthesis of fatty acid. This process is the reverse of the degradation of fatty acid.
The Citric acid cycle is a series of biochemical reactions that use acetyl CoA (produced by oxidation of pyruvate) to produce carbon dioxide, NADH and FADH2 in a series of
(a)
Answer to Problem 14.105EP
The citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid butyrate intermediate is succinate.
Explanation of Solution
Intermediates involved in the lipogenesis are derivative of C4 molecule butyric acid. Butyric acid is a monocarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the lipogenesis is a C4 derivative of monocarboxylic acid. The structure of butyric acid is,
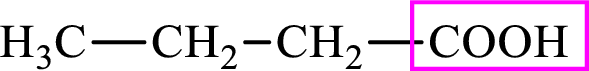
Succinic acid is a dicarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the citric acid cycle is a
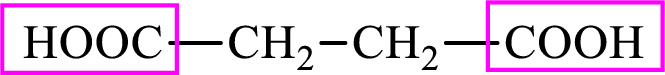
The structure of butyrate is,
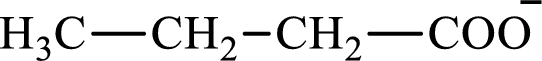
The structure of succinate is,
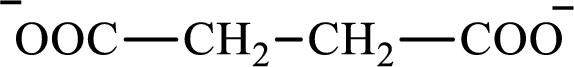
Butyrate and succinate are saturated acids with four carbon atoms in each molecule. Butyrate is a monoacid that is formed as an intermediate in lipogenesis while succinate is a diacid that is formed as an intermediate in the citric acid cycle. Therefore, the citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid butyrate intermediate is succinate.
(b)
Interpretation:
The citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid intermediate acetoacetate has to be determined.
Concept introduction:
Lipogenesis is the process employed for the synthesis of fatty acid. The starting precursor for the synthesis is acetyl CoA. The enzyme employed for the process is fatty acid synthase. It is a multienzyme complex that ties the reaction responsible for the synthesis of fatty acid. This process is the reverse of the degradation of fatty acid.
The Citric acid cycle is a series of biochemical reactions that use acetyl CoA (produced by oxidation of pyruvate) to produce carbon dioxide, NADH and FADH2 in a series of redox reactions.
Intermediates involved in the lipogenesis are derivatives of C4 molecule butyric acid. Butyric acid is a monocarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the lipogenesis is a C4 derivative of monocarboxylic acid. The structure of butyric acid is,
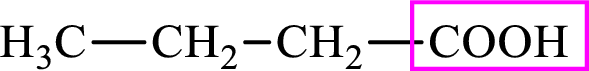
Succinic acid is a dicarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the citric acid cycle is a C4 derivative of dicarboxylic acid. The structure of succinic acid is,
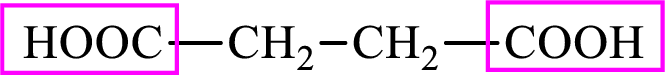
(b)
Answer to Problem 14.105EP
The citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid acetoacetate intermediate is oxaloacetate.
Explanation of Solution
Acetoacetate is the intermediate in the lipogenesis. The structure of acetoacetate is,
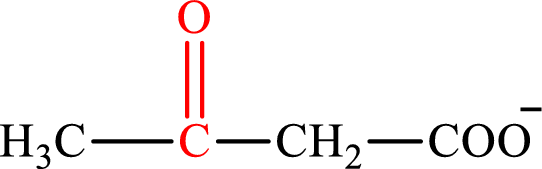
Oxaloacetate is the intermediate in the citric acid cycle. The structure of oxaloacetate is,
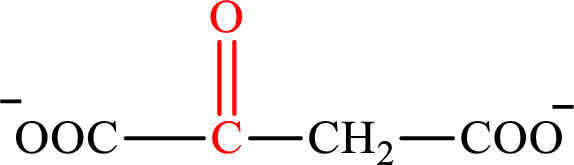
Acetoacetate and oxaloacetate are the keto derivatives of saturated acid with four carbon atoms in each molecule. Acetoacetate is a keto derivative of monocarboxylic acid while oxaloacetate is a keto derivative of dicarboxylic acid. Therefore, the citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid acetoacetate intermediate is oxaloacetate.
(c)
Interpretation:
The citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid intermediate β-hydroxybutyrate has to be determined.
Concept introduction:
Lipogenesis is the process employed for the synthesis of fatty acid. The starting precursor for the synthesis is acetyl CoA. The enzyme employed for the process is fatty acid synthase. It is a multienzyme complex that ties the reaction responsible for the synthesis of fatty acid. This process is the reverse of the degradation of fatty acid.
The Citric acid cycle is a series of biochemical reactions that use acetyl CoA (produced by oxidation of pyruvate) to produce carbon dioxide, NADH and FADH2 in a series of redox reactions.
Intermediates involved in the lipogenesis are derivatives of C4 molecule butyric acid. Butyric acid is a monocarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the lipogenesis is a C4 derivative of monocarboxylic acid. The structure of butyric acid is,
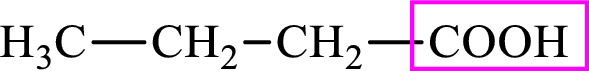
Succinic acid is a dicarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the citric acid cycle is a C4 derivative of dicarboxylic acid. The structure of succinic acid is,
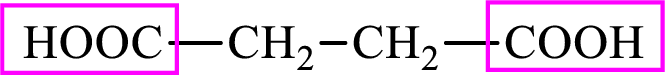
(c)
Answer to Problem 14.105EP
The citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid β-hydroxybutyrate intermediate is malate.
Explanation of Solution
β-Hydroxybutyrate is the intermediate in the lipogenesis. The structure of β-hydroxybutyrate is,
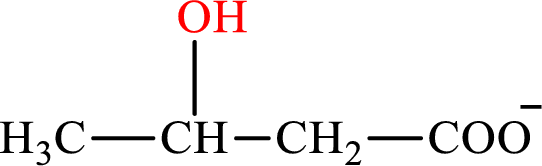
Malate is the intermediate in the citric acid cycle. The structure of Malate is,
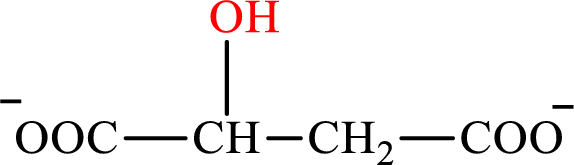
β-Hydroxybutyrate and malate are the hydroxy derivatives of saturated acid with four carbon atoms in each molecule. β-Hydroxybutyrate is a hydroxy derivative of monocarboxylic acid while malate is a hydroxy derivative of dicarboxylic acid. Therefore, the citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid β-hydroxybutyrate intermediate is malate.
(d)
Interpretation:
The citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid intermediate crotonate has to be determined.
Concept introduction:
Lipogenesis is the process employed for the synthesis of fatty acid. The starting precursor for the synthesis is acetyl CoA. The enzyme employed for the process is fatty acid synthase. It is a multienzyme complex that ties the reaction responsible for the synthesis of fatty acid. This process is the reverse of the degradation of fatty acid.
The Citric acid cycle is a series of biochemical reactions that use acetyl CoA (produced by oxidation of pyruvate) to produce carbon dioxide, NADH and FADH2 in a series of redox reactions.
Intermediates involved in the lipogenesis are derivatives of C4 molecule butyric acid. Butyric acid is a monocarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the lipogenesis is a C4 derivative of monocarboxylic acid. The structure of butyric acid is,
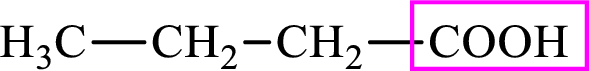
Succinic acid is a dicarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the citric acid cycle is a C4 derivative of dicarboxylic acid. The structure of succinic acid is,
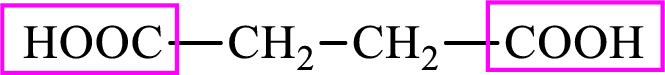
(d)
Answer to Problem 14.105EP
The citric acid cycle diacid intermediate counterpart for lipogenesis C4-ACP monoacid crotonate intermediate is fumarate.
Explanation of Solution
Crotonate is the intermediate in the lipogenesis. The structure of crotonate is,
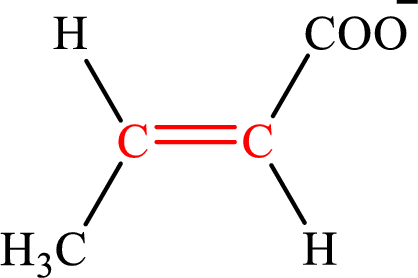
Fumarate is the intermediate in the citric acid cycle. The structure of fumarate is,
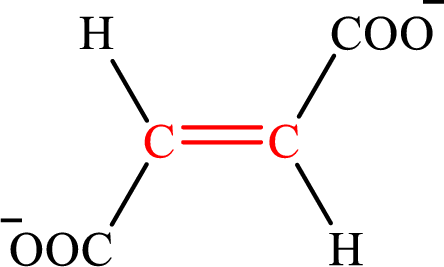
Crotonate and fumarate are the unsaturated derivatives of a
Want to see more full solutions like this?
Chapter 14 Solutions
Organic And Biological Chemistry
- Can anyone help me solve this step by step. Thank you in advaarrow_forwardPlease draw the mechanism for this Friedel-crafts acylation reaction using arrowsarrow_forwardDraw the Fischer projection of D-fructose. Click and drag to start drawing a structure. Skip Part Check AP 14 tv SC F1 F2 80 F3 a F4 ! 2 # 3 CF F5 75 Ax MacBook Air 894 $ 5olo % Λ 6 > W F6 K F7 &arrow_forward
- Consider this step in a radical reaction: Y What type of step is this? Check all that apply. Draw the products of the step on the right-hand side of the drawing area below. If more than one set of products is possible, draw any set. Also, draw the mechanism arrows on the left-hand side of the drawing area to show how this happens. ionization propagation initialization passivation none of the abovearrow_forward22.16 The following groups are ortho-para directors. (a) -C=CH₂ H (d) -Br (b) -NH2 (c) -OCHS Draw a contributing structure for the resonance-stabilized cation formed during elec- trophilic aromatic substitution that shows the role of each group in stabilizing the intermediate by further delocalizing its positive charge. 22.17 Predict the major product or products from treatment of each compound with Cl₁/FeCl₂- OH (b) NO2 CHO 22.18 How do you account for the fact that phenyl acetate is less reactive toward electro- philic aromatic substitution than anisole? Phenyl acetate Anisole CH (d)arrow_forwardShow how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid.arrow_forward
- Help me solve this problem. Thank you in advance.arrow_forward22.7 Predict the monoalkylated products of the following reactions with benzene. (a) AlCl3 Ya (b) AlCl3 (c) H3PO4 (d) 22.8 Think-Pair-Share AICI3 The reaction below is a common electrophilic aromatic substitution. SO3 H₂SO4 SO₂H (a) Draw the reaction mechanism for this reaction using HSO,+ as the electrophile. (b) Sketch the reaction coordinate diagram, where the product is lower in energy than the starting reactant. (c) Which step in the reaction mechanism is highest in energy? Explain. (d) Which of the following reaction conditions could be used in an electrophilic aro- matic substitution with benzene to provide substituted phenyl derivatives? (i) AICI3 HNO3 H₂SO4 K2Cr2O7 (iii) H₂SO4 (iv) H₂PO₁arrow_forwardIs an acid-base reaction the only type of reaction that would cause leavening products to rise?arrow_forward
- Help me understand this! Thank you in advance.arrow_forward22.22 For each compound, indicate which group on the ring is more strongly activating and then draw a structural formula of the major product formed by nitration of the compound. Br CHO (a) CH3 (b) (c) CHO CH3 SO₂H (d) ☑ OCHS NO₂ (e) (f) CO₂H NHCOCH3 NHCOCH, (h) CHS 22.23 The following molecules each contain two aromatic rings. (b) 000-100- H3C (a) (c) Which ring in each undergoes electrophilic aromatic substitution more readily? Draw the major product formed on nitration.arrow_forwardV Consider this step in a radical reaction: Br: ? What type of step is this? Check all that apply. Draw the products of the step on the right-hand side of the drawing area below. If more than one set of products is possible, draw any set. Also, draw the mechanism arrows on the left-hand side of the drawing area to show how this happens. ⚫ionization termination initialization neutralization none of the abc Explanation Check 80 Ο F3 F1 F2 2 F4 01 % do5 $ 94 #3 X 5 C MacBook Air 25 F5 F6 66 ©2025 ˇ F7 29 & 7 8arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning




