
a)
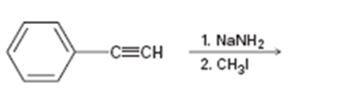
Interpretation:
The product of the two step process shown is to be given and complete electron-pushing mechanism for its formation also is to be provided.
Concept introduction:
The terminal
To propose:
The product of the two step process shown and to give a complete electron-pushing mechanism for its formation.
b)
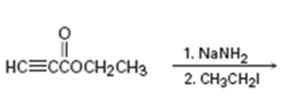
Interpretation:
The product of the two step process shown is to be given and complete electron-pushing mechanism for its formation also is to be provided.
Concept introduction:
The terminal alkynes being acidic form sodium alkynides when treated with bases like sodium amide. The alkynides when treated with alkyl halides yield the higher alkynes as the product. The alkynide ion being nucleophilic attacks the positively polarized carbon of C-X bond in alkyl halides displaces the halogen to yield the higher alkyne as the product.
To propose:
The product of the two step process shown and to give a complete electron-pushing mechanism for its formation.
c)
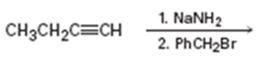
Interpretation:
The product of the two step process shown is to be given and complete electron-pushing mechanism for its formation also is to be provided.
Concept introduction:
The terminal alkynes being acidic form sodium alkynide when treated with bases like sodium amide. The alkynides when treated with alkyl halides yield the higher alkynes as the product. The alkynide ion being nucleophilic attacks the positively polarized carbon of C-X bond in alkyl halides displaces the halogen to yield the higher alkyne as the product.
To propose:
The product of the two step process shown and to give a complete electron-pushing mechanism for its formation.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
- Justify the polymerization of vanadates VO43-, as a function of concentration and pH.arrow_forwardWhat is the preparation of 500 mL of 100mM MOPS buffer (pH=7.5) starting with 1 M MOPS and 1 M NaOH? How would I calculate the math?arrow_forwardIndicate the correct option.a) Isopolianions are formed around metallic atoms in a low oxidation state.b) Non-metals such as N, S, C, Cl, ... give rise to polyacids (oxygenated).c) Both are incorrect.arrow_forward
- 14. Which one of the compounds below is the major organic product obtained from the following series of reactions? Br OH OH CH3O™ Na+ H*, H₂O SN2 HO OH A B C D 0 Earrow_forwardWavelength (nm) I'm not sure what equation I can come up with other than the one generated with my graph. Can you please show me the calculations that were used to find this equation? Give an equation that relates energy to wavelength. Explain how you arrived at your equation. Wavelength Energy (kJ/mol) (nm) 350 341.8 420 284.8 470 254.5 530 225.7 580 206.3 620 192.9 700 170.9 750 159.5 Energy vs. Wavelength (Graph 1) 400 350 y=-0.4367x+470.82 300 250 200 150 100 50 O 0 100 200 300 400 500 600 700 800 Energy (kJ/mol)arrow_forward5. Draw molecular orbital diagrams for superoxide (O2¯), and peroxide (O2²-). A good starting point would be MO diagram for O2 given in your textbook. Then: a) calculate bond orders in superoxide and in peroxide; indicate which species would have a stronger oxygen-oxygen bond; b) indicate which species would be a radical. (4 points)arrow_forward
- 16. Which one of the compunds below is the final product of the reaction sequence shown here? عملاء .OH Br. (CH3)2CH-C=C H+,H,O 2 mol H2, Pt A OH B OH D OH E OH C OHarrow_forwardIndicate whether any of the two options is correct.a) The most common coordination structure for isopolianions is the prismb) Heteropolianions incorporate alkaline cations into their structuresarrow_forwardPlease correct answer and don't use hand ratingarrow_forward
- Wavelength (nm) I'm not sure what equation I can come up with other than the one generated with my graph. Can you please show me the calculations that were used to find this equation? Give an equation that relates energy to wavelength. Explain how you arrived at your equation. Wavelength Energy (kJ/mol) (nm) 350 341.8 420 284.8 470 254.5 530 225.7 580 206.3 620 192.9 700 170.9 750 159.5 Energy vs. Wavelength (Graph 1) 400 350 y=-0.4367x+470.82 300 250 200 150 100 50 O 0 100 200 300 400 500 600 700 800 Energy (kJ/mol)arrow_forward6. For the following molecules: draw Lewis dot-structures; use VSEPR method to determine geometries of the following molecules/ions. Are the central atoms in these molecules/ions considered of normal valency, or are they hypervalent? (please read paragraph 2.6) a) BrF3 (6 points) b) BrF4 c) IF₂ 4arrow_forwardNonearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

