
(a)
Interpretation:
The element for the following orbital diagram has to be identified. Also, whether this orbital diagram is the ground state or an excited state of the atom has to be determined. If this orbital diagram is an excited state then the ground state orbital diagram has to be drawn.
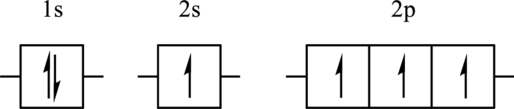
Concept Introduction:
The electronic configuration is defined as the distribution of electrons in various atomic orbitals of the atom. The electrons that are present in an outermost orbital are known as valence electrons whereas those present in the orbitals with lower quantum numbers are called core electrons. Electrons are filled in orbitals in accordance with three rules: Aufbau principle, Hund’s rule, and Pauli’s exclusion principle. Aufbau principle states that electrons are filled in the orbitals from lower to higher energy level as follows:
Hund’s rule states that initially each orbital is singly occupied and then pairing occurs and Pauli’s exclusion principle states that the spin of two electrons in one orbital is always different.
If one or more electron is in higher allowed orbitals of the atom then this is known as excited state of the atom. Excited-state of an atom can be shown by its orbital diagram. The orbital diagram is the way to show the distribution of electrons in an atom.
(b)
Interpretation:
The element for the following orbital diagram has to be identified. Also, whether this orbital diagram is the ground state or an excited state of the atom has to be determined. If this orbital diagram is an excited state then ground state orbital diagram has to be drawn.
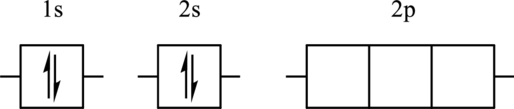
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The element for the following orbital diagram has to be identified. Also, whether this orbital diagram is the ground state or an excited state of the atom has to be determined. If this orbital diagram is an excited state then the ground state orbital diagram has to be drawn.
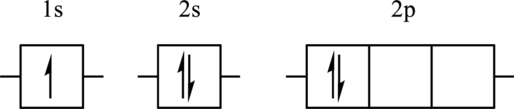
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The element for the following orbital diagram has to be identified. Also, whether this orbital diagram is the ground state or an excited state of the atom has to be determined. If this orbital diagram is an excited state then ground state orbital diagram has to be drawn.
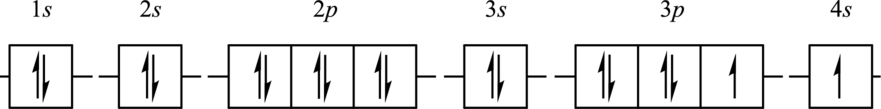
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Chemistry: Principles and Practice
- What is the name of the following compound? SiMe3arrow_forwardK Draw the starting structure that would lead to the major product shown under the provided conditions. Drawing 1. NaNH2 2. PhCH2Br 4 57°F Sunny Q Searcharrow_forward7 Draw the starting alkyl bromide that would produce this alkyne under these conditions. F Drawing 1. NaNH2, A 2. H3O+ £ 4 Temps to rise Tomorrow Q Search H2arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





