
Interpretation:
The number of possible isomers for galactose, glucose and fructose should be calculated.
Concept introduction:
The compounds having similar chemical formula but different structures are known as isomers.
The compounds having similar chemical or molecular formula but different connectivity is known as constitutional isomers.
Number of stereoisomers is calculated by below expression:
Where, n = number of chiral carbons
Answer to Problem 10SSC
Number of isomers of glucose = 16
Number of isomers of galactose = 16
Number of isomers of fructose = 8
Explanation of Solution
Glucose consists of single unit of sugar which can’t be hydrolyzed into simpler molecule.
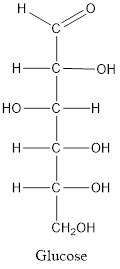
When a carbon atom is linked with different groups is known as chiral carbon and center is known as chiral center. Chiral center is represented by a symbol *.
The structure is drawn as with symbol *:
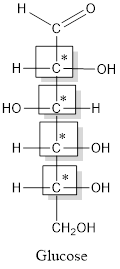
Thus, four chiral centers are present in the above structure as first four carbon atoms are linked with four different groups.
Number of isomers is calculated as:
Here, n = 4 (3 chiral carbon atoms are present)
Number of isomers =
= 16
Therefore, sixteen isomers are present for the structure of glucose.
Galactose consists of single unit of sugar which can’t be hydrolyzed into simpler molecule.
In galactose, the hydroxyl group linked with forth carbon atom of the chain is present on left side whereas in glucose, the hydroxyl group linked with forth carbon atom of the chain is present on right side.
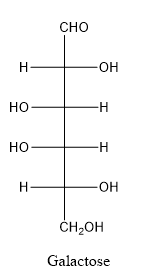
When a carbon atom is linked with different groups is known as chiral carbon and center is known as chiral center. Chiral center is represented by a symbol *.
The structure is drawn as with symbol *:
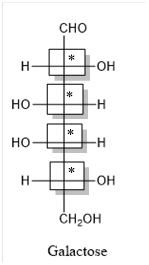
Thus, four chiral centers are present in the above structure as first four carbon atoms are linked with four different groups.
Number of isomers is calculated as:
Here, n = 4 (3 chiral carbon atoms are present)
Number of isomers =
= 16
Therefore, sixteen isomers are present for the structure of galactose.
Fructose consists of single unit of sugar which can’t be hydrolyzed into simpler molecule.
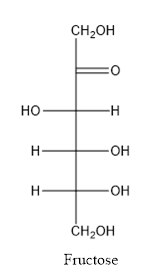
When a carbon atom is linked with different groups is known as chiral carbon and center is known as chiral center. Chiral center is represented by a symbol *.
The structure is drawn as with symbol *:
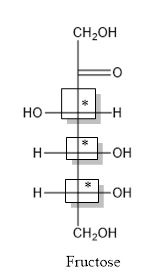
Thus, three chiral centers are present in the above structure as first three carbon atoms are linked with four different groups.
Number of isomers is calculated as:
Here, n = 3 (3 chiral carbon atoms are present)
Number of isomers =
= 8
Therefore, eight isomers are present for the structure of fructose.
Chapter 23 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Campbell Biology: Concepts & Connections (9th Edition)
College Physics: A Strategic Approach (3rd Edition)
Microbiology with Diseases by Body System (5th Edition)
Chemistry: The Central Science (14th Edition)
Campbell Biology in Focus (2nd Edition)
Human Anatomy & Physiology (2nd Edition)
- State the products (formulas) of the reaction of acetophenone with iodine and NaOH.arrow_forwardExplanation Check Draw the skeletal ("line") structure of 5-hydroxy-4-methyl-2-pentanone. Click and drag to start drawing a structure. Х © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Cer ☐ : Carrow_forward1. Using a Model set Build a model for the following compound [CH2BrCI]. 2. Build another model of the mirror image of your first molecule. 3. Place the two models next to each other and take a picture which shows the differences between the two models. 4. Determine the absolute stereochemistry R or S for the two models. 5. Write or type a paragraph to Discuss the stereochemical relationship between the two models of CH2BrCl. You must provide an explanation for your conclusions also provide a description for the colors used to represent each atom in the model's images.arrow_forward
- What parameters are included in the specific rotation calculation of a pure substance based on measurement from a polarimeter? Select one or more: Density of the sample Pathlength of the sample container Enantiomeric excess of the sample Measured rotation of lightarrow_forwardV Determine whether the following molecule is a hemiacetal, acetal, or neither and select the appropriate box below. Also, highlight the hemiacetal or acetal carbon if there is one. Explanation O CH O Ohemiacetal Oacetal Oneither Check A 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Cer 000 Ararrow_forward1. Using Online resources and chemical structures hand draw four different organic compounds (not those already shown in your handout) that are chiral, optically active (a pair of enantiomers will count as one). Pay attention to correct stereochemistry 2. Write or type a short paragraph to Discuss the stereochemical relationship between the four compounds.arrow_forward
- 1. Using a Model set Build a model for the following compound [CHBRIF] 2. Build another model of the mirror image of your first molecule. 3. Place the two models next to each other and take a picture which shows the differences between the two models. 4. Determine the absolute stereochemistry R or S for the two models. 5. Write or type a paragraph to Discuss the stereochemical relationship between the two models of CHBгCIF. You must provide an explanation for your conclusions also provide a description for the colors used to representarrow_forwardThe specific rotation of a sample depends upon measured angle of rotation, the density of the sample, and the pathway length of the light. True Falsearrow_forwardConsider the molecule A,B, C and D shown below, (1 x 4) Br NH2 A OH Br 边 H B C D 1. Assign the R/S configuration to each chiral center and identify by circling all the chiral centers. 2. Draw an image for the enantiomer of each of the compounds A, B, C and D.arrow_forward
- Could you crystallize one enantiomer of mandelic acid from a racemic mixture (using the typical achiral solvents found in our lab) without preparing a diastereomeric salt? Why or why not? No, because both enantiomers have the same solubility in achiral solvents. than the other. ооо Yes, because one enantiomer has a higher melting point No, because both enantiomers are liquids. Yes, because one enantiomer is more crystalline than the other.arrow_forwardIf the literature value of specific rotation for a chiral compound is -53.6°, what is the enantiomeric excess of a compound with a measured specific rotation of -40.5°?arrow_forwardThe process to determine the configuration, starts by placing the lowest priority substituent toward the back. If the substituents pointing forward decrease in priority in a clockwise order, the configuration is S. If the substituents decrease in priority in a counterclockwise order, the configuration is R. True Falsearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





