
Concept explainers
(a)
Interpretation:
Sigma and pi bond in
Concept Introduction:
Sigma (σ) bonds are the bonds in which shared hybrid orbital’s electron density are concentrated along the internuclear axis.
Pi (π) bonds are the bonds in which shared unhybridized orbital’s (p, d, etc) electron density are concentrated in above and below of the plane of the molecule.
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
Geometry of different types of molecule with respect to the hybridizations are mentioned below,
(a)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
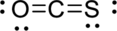 .
.
Sigma bond and pi bonds in the molecule are marked below:
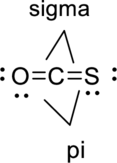
(b)
Interpretation:
Sigma and pi bond in
Concept Introduction:
Refer to (a).
(b)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
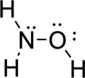 .
.
Sigma bond and pi bonds in the molecule are marked below:
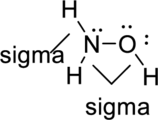
(c)
Interpretation:
Sigma and pi bond in
Concept Introduction:
Refer to (a).
(c)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
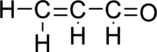 .
.
Sigma bond and pi bonds in the molecule are marked below:
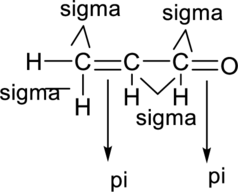
(d)
Interpretation:
Sigma and pi bond in
Concept Introduction:
Refer to (a).
(d)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for
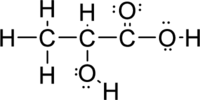
Sigma bond and pi bonds in the molecule are marked below:
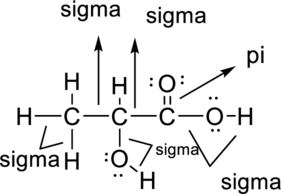
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry: The Molecular Science
- One liter of N2(g) at 2.1 bar and two liters of Ar(g) at 3.4 bar are mixed in a 4.0 liter flask to form an ideal gas mixture. Calculate the value of the final pressure of the mixture if the initial and final temperature of the gases are the same. Repeat this calculation if the initial temperature of the N2(g) and Ar(g) are 304 K and 402 K, respectively, and the final temperature of the mixture is 377 K.arrow_forward10 5 4. These four 'H NMR spectra were recorded from different isomers with molecular formula CsH,CIO. They all contain a carbonyl group. Determine the structure of the different isomers. 0 10 5 0 10 5 10 9 8 7 6 5 4 3. 1 0 9 10 10 66 9 0 10 9 10 5 1 8 7 6 5 3 2 -a 8 7 6 5 1 10 9 8 7 6 5 22 2 1 0 3 2 16 1 0 3 2 1 2 6 0arrow_forwardUse the expression below to ⚫ calculate its value and report it to the proper number of significant digits (you may need to round your answer). ⚫ calculate the % error (or % relative error or % inherent error) ⚫ calculate the absolute error. (20.54±0.02 × 0.254±0.003) / (3.21±0.05) = Value: % Error: Absolute error: ± | % (only 1 significant digit) (only 1 significant digit)arrow_forward
- In each case (more ductile, more brittle, more tough or resistant), indicate which parameter has a larger value. parameter Elastic limit Tensile strength more ductile Strain at break Strength Elastic modulus more fragile more tough or resistantarrow_forwardNonearrow_forwardWhat functional groups are present in this IRarrow_forward
- In each case (more ductile, more brittle, more tough or resistant), indicate which parameter has a larger value. parameter Elastic limit Tensile strength more ductile Strain at break Strength Elastic modulus more fragile more tough or resistantarrow_forward4) A typical bottle of pop holds carbon dioxide at a pressure of 5 atm. What is the concentration of carbon dioxide in th solution? 5) A stream flowing over rocks and such is exposed to the atmosphere and well aerated. What would be the nitrogen concentration in the water at 25°C? (Air pressure is 1.000 bar.)arrow_forwardUse the expression below to ⚫ calculate its value and report it to the proper number of significant digits (you may need to round your answer). ⚫ calculate the % error (or % relative error or % inherent error) ⚫ calculate the absolute error. (30.078±0.003) - (20.174±0.001) + (9.813±0.005) = Value: % Error: absolute error: ± % (only 1 significant digit) (only 1 significant digit)arrow_forward
- Don't used Ai solution and don't used hand raitingarrow_forwardCircle the letter next to the most appropriate response. 1) Which is likely to be the least soluble with water? a) hexane b) acetone c) trichloromethane d) trinitro-toluene 2) Which is likely to be the most soluble in 3,4-dimethyloctane? a) hexane b) acetone c) trichloromethane d) trinitro-toluene 3) When ammonium nitrate is dissolved in water, the solution: a) gets warmer. b) gets colder. c) stays the same temperature. d) is none of the above because potassium nitrate is insoluble.arrow_forwardNonearrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

