
Concept explainers
(a)
Interpretation:
The Lewis electron dot structure for
Concept Introduction:
- Lewis structures are diagrams that represent the
chemical bonding of covalently bonded molecules and coordination compounds. - It is also known as Lewis dot structures which represent the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
- The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Lewis structure for any molecule is drawn by using the following steps,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined.
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
(a)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for given molecule can be determined by first drawing the skeletal structure. Then, the total number of valence electrons for all atoms present in the molecule is determined.
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed considering each atom contains eight electrons in its valence shell.
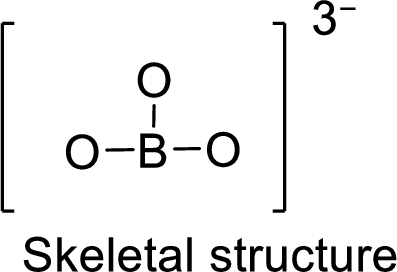 .
.
Total number of valence electrons is given below:
Total number of electrons in bonds present is given below:
The eighteen electrons remaining will be distributing in such a way that each atom should have 8 electrons in its outer shell.
Therefore, the Lewis structure is given below:
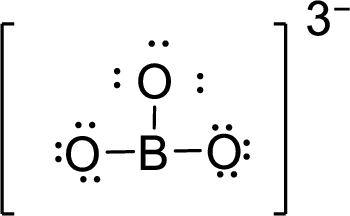 .
.
The molecular geometry will be triangular planar because of the presence of three bond pairs around the central atom.
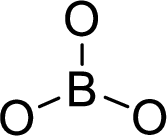 .
.
There will be three electron regions in the molecule and hence the electron-region geometry will also be triangular planar.
(b)
Interpretation:
The Lewis electron dot structure for
Concept Introduction:
Refer to (a).
(b)
Explanation of Solution
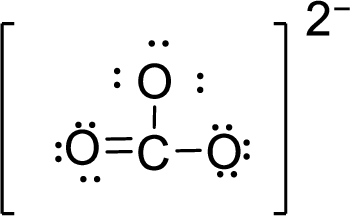
Given molecule is
The Lewis electron dot structure for given molecule can be determined by first drawing the skeletal structure. Then, the total number of valence electrons for all atoms present in the molecule is determined.
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed considering each atom contains eight electrons in its valence shell.
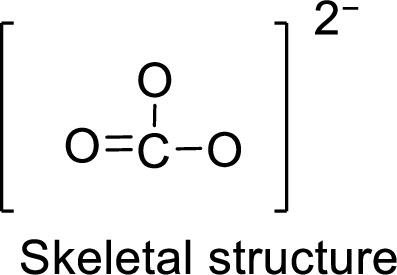 .
.
Total number of valence electrons is given below:
Total number of electrons in bonds present is given below:
The sixteen electrons remaining will be distributing in such a way that each atom should have 8 electrons in its outer shell.
Therefore, the Lewis structure is given below:
 .
.
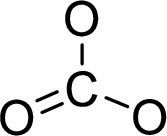
The molecular geometry will be triangular planar because of the presence of three bond pairs around the central atom.
 .
.
There will be three electron regions in the molecule and hence the electron-region geometry will also be triangular planar.
(c)
Interpretation:
The Lewis electron dot structure for
Concept Introduction:
Refer to (a).
(c)
Explanation of Solution
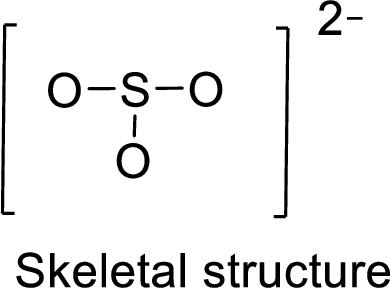
Given molecule is
The Lewis electron dot structure for given molecule can be determined by first drawing the skeletal structure. Then, the total number of valence electrons for all atoms present in the molecule is determined.
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed considering each atom contains eight electrons in its valence shell.
 .
.
Total number of valence electrons is given below:
Total number of electrons in bonds present is given below:
The twenty electrons remaining will be distributing in such a way that each atom should have 8 electrons in its outer shell.
Therefore, the Lewis structure is given below:
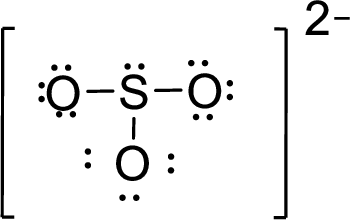
The molecular geometry will be triangular pyramidal because of the presence of three bond pairs and one lone pair around the central atom.
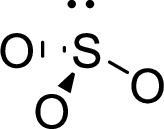 .
.
There will be four electron regions in the molecule and hence the electron-region geometry will be tetrahedral.
(d)
Interpretation:
The Lewis electron dot structure for
Concept Introduction:
Refer to (a).
(d)
Explanation of Solution
Given molecule is
The Lewis electron dot structure for given molecule can be determined by first drawing the skeletal structure. Then, the total number of valence electrons for all atoms present in the molecule is determined.
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed considering each atom contains eight electrons in its valence shell.
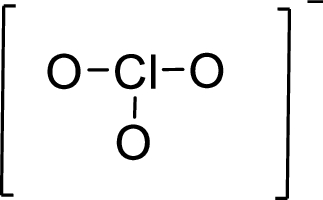 .
.
Total number of valence electrons is given below:
Total number of electrons in bonds present is given below:
The twenty electrons remaining will be distributing in such a way that each atom should have 8 electrons in its outer shell.
Therefore, the Lewis structure is given below:
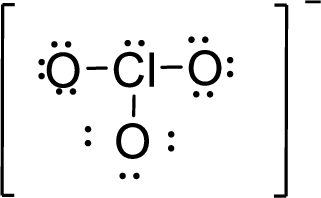 .
.
The molecular geometry will be triangular pyramidal because of the presence of three bond pairs and one lone pair around the central atom.
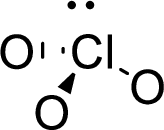 .
.
There will be four electron regions in the molecule and hence the electron-region geometry will be tetrahedral.
Comparing all the molecules, the central atom is oxygen and three bonds are attached to it. Thus, the similarity is that the number of bonds and type of atoms attached around the central atom is the same. But, there are differences in the geometry of the molecules because of the differences in lone pair of electrons around the central atom.
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry: The Molecular Science
- If we have two compounds: acetone (CH₃COCH₃) and acetic acid (CH₃COOH), applying heat to them produces an aldol condensation of the two compounds. If this is correct, draw the formula for the final product.arrow_forwardIf we have two compounds: acetone (CH3COCH3) and acetic acid (CH3COOH); if we apply heat (A), what product(s) are obtained?arrow_forwardQUESTION: Fill out the answers to the empty green boxes attached in the image. *Ensure you all incorporate all 27 values (per column)*arrow_forward
- You need to make a buffer by dissolving benzoic acid and sodium benzoate in water. What is the mass of benzoic acid that you would weigh out, in mg, to create 50 mL of a buffer at pH = 4.7 that will change pH no more than 0.10 units with the addition of 0.001 moles of acid or base? Enter just the answer without the units (mg) - just the number will do!arrow_forwardDraw the formula for 3-isopropylcyclopentane-1-carbonyl chloride.arrow_forwardQUESTION: Fill out the answers to the empty green boxes attached in the image. *Ensure you all incorporate all 27 values (per column)*arrow_forward
- Give the organic products: (benzyne) Br ? CH3 + K* :NH, liq NH3 HINT: Two products are formed. Each is a substituted aniline; they are isomers of each other. NH2 II I H₂N. CH3 CH3 III Select one: ○ A. I and II ○ B. I and III O C. I and IV O D. II and III O E. III and IV H₂N CH3 IV CH₂-NH2arrow_forwardPredict the major products of this organic reaction: HBr (1 equiv) cold ? Some important notes: • Draw the major product, or products, of this reaction in the drawing area below. • You can draw the products in any arrangement you like. • Pay careful attention to the reaction conditions, and only include the major products. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. • Note that there is only 1 equivalent of HBr reactant, so you need not consider the case of multiple additions. Erase something Explanation Check 2025 McGraw Hill LLC. All Rights Reserved. Terarrow_forwardQ14. Fill this chart: (please refer to ppt notes/browser to answer these questions) What alcohol is also called wood alcohol? What is the common name of ethanol? Draw the structure of phenol and thiophene? Are bigger chain alcohol like heptanol and octanol are soluble or insoluble in water and explain it ? Are ethers soluble or insoluble in water? What suffix and prefix are used for alcohol while naming alcohol and ether? What the process called when we add water to any alkene to make alcohol? Q16. Draw the diagram of following aromatic compound (practice from previous module) Aniline Phenol Benzoic acid Methyl benzoate Q17. a. Write the oxidation reactions for the 2 propanol. b. Write the oxidation reaction of the ethanol.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

