
Chemistry 2012 Student Edition (hard Cover) Grade 11
12th Edition
ISBN: 9780132525763
Author: Prentice Hall
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Question
Chapter 22.3, Problem 19SP
Interpretation Introduction
Interpretation: Below drawn alkane should be named using the IUPAC system.
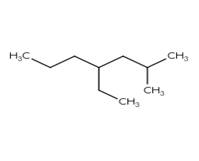
Concept Introduction: Organic compounds containing single-bonded Carbon and Hydrogen atoms are
Formula for Alkane is CnH2n+2
Expert Solution & Answer
Answer to Problem 19SP
IUPAC name is 3-isobutyl hexane.
Explanation of Solution
Given Information: The given compound has a name according to the IUPAC system.
Alkane containing 6 Carbon atoms has n=6.
Substituting n=6 in the molecular formula for Alkane (CnH2n+2), we get C6H14.
Molecule of Hexane with isobutyl group (-CH2-CH-(CH3)2) are replacing Hydrogen atom on Carbon atom 3.
Therefore, the IUPAC name is 3-isobutyl hexane.
Conclusion
IUPAC name is 3-isobutyl hexane.
Chapter 22 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
Ch. 22.1 - Prob. 1SPCh. 22.1 - Prob. 2SPCh. 22.1 - Prob. 3SPCh. 22.1 - Prob. 4SPCh. 22.1 - Prob. 5SPCh. 22.1 - Prob. 6SPCh. 22.1 - Prob. 7LCCh. 22.1 - Prob. 8LCCh. 22.1 - Prob. 9LCCh. 22.1 - Prob. 10LC
Ch. 22.1 - Prob. 11LCCh. 22.1 - Prob. 12LCCh. 22.1 - Prob. 13LCCh. 22.2 - Prob. 14LCCh. 22.2 - Prob. 15LCCh. 22.2 - Prob. 16LCCh. 22.2 - Prob. 17LCCh. 22.2 - Prob. 18LCCh. 22.3 - Prob. 19SPCh. 22.3 - Prob. 20SPCh. 22.3 - Prob. 21LCCh. 22.3 - Prob. 22LCCh. 22.3 - Prob. 23LCCh. 22.3 - Prob. 24LCCh. 22.3 - Prob. 25LCCh. 22.3 - Prob. 26LCCh. 22.3 - Prob. 27LCCh. 22.4 - Prob. 28LCCh. 22.4 - Prob. 29LCCh. 22.4 - Prob. 30LCCh. 22.4 - Prob. 31LCCh. 22.4 - Prob. 32LCCh. 22.5 - Prob. 33LCCh. 22.5 - Prob. 34LCCh. 22.5 - Prob. 35LCCh. 22.5 - Prob. 36LCCh. 22.5 - Prob. 37LCCh. 22.5 - Prob. 38LCCh. 22.5 - Prob. 39LCCh. 22.5 - Prob. 40LCCh. 22 - Prob. 41ACh. 22 - Prob. 42ACh. 22 - Prob. 43ACh. 22 - Prob. 44ACh. 22 - Prob. 45ACh. 22 - Prob. 46ACh. 22 - Prob. 47ACh. 22 - Prob. 48ACh. 22 - Prob. 49ACh. 22 - Prob. 50ACh. 22 - Prob. 51ACh. 22 - Prob. 52ACh. 22 - Prob. 53ACh. 22 - Prob. 54ACh. 22 - Prob. 55ACh. 22 - Prob. 56ACh. 22 - Prob. 57ACh. 22 - Prob. 58ACh. 22 - Prob. 59ACh. 22 - Prob. 60ACh. 22 - Prob. 61ACh. 22 - Prob. 62ACh. 22 - Prob. 63ACh. 22 - Prob. 64ACh. 22 - Prob. 65ACh. 22 - Prob. 66ACh. 22 - Prob. 67ACh. 22 - Prob. 68ACh. 22 - Prob. 69ACh. 22 - Prob. 70ACh. 22 - Prob. 71ACh. 22 - Prob. 72ACh. 22 - Prob. 73ACh. 22 - Prob. 74ACh. 22 - Prob. 75ACh. 22 - Prob. 76ACh. 22 - Prob. 77ACh. 22 - Prob. 78ACh. 22 - Prob. 79ACh. 22 - Prob. 80ACh. 22 - Prob. 81ACh. 22 - Prob. 82ACh. 22 - Prob. 84ACh. 22 - Prob. 85ACh. 22 - Prob. 86ACh. 22 - Prob. 87ACh. 22 - Prob. 89ACh. 22 - Prob. 90ACh. 22 - Prob. 91ACh. 22 - Prob. 92ACh. 22 - Prob. 93ACh. 22 - Prob. 94ACh. 22 - Prob. 95ACh. 22 - Prob. 96ACh. 22 - Prob. 97ACh. 22 - Prob. 98ACh. 22 - Prob. 99ACh. 22 - Prob. 100ACh. 22 - Prob. 101ACh. 22 - Prob. 102ACh. 22 - Prob. 103ACh. 22 - Prob. 104ACh. 22 - Prob. 105ACh. 22 - Prob. 106ACh. 22 - Prob. 107ACh. 22 - Prob. 108ACh. 22 - Prob. 109ACh. 22 - Prob. 110ACh. 22 - Prob. 111ACh. 22 - Prob. 1STPCh. 22 - Prob. 2STPCh. 22 - Prob. 3STPCh. 22 - Prob. 4STPCh. 22 - Prob. 5STPCh. 22 - Prob. 6STPCh. 22 - Prob. 7STPCh. 22 - Prob. 8STPCh. 22 - Prob. 9STPCh. 22 - Prob. 10STPCh. 22 - Prob. 11STPCh. 22 - Prob. 12STPCh. 22 - Prob. 13STPCh. 22 - Prob. 14STPCh. 22 - Prob. 15STPCh. 22 - Prob. 16STP
Knowledge Booster
Similar questions
- Each of the highlighted carbon atoms is connected to hydrogen atoms.arrow_forwardく Complete the reaction in the drawing area below by adding the major products to the right-hand side. If there won't be any products, because nothing will happen under these reaction conditions, check the box under the drawing area instead. Note: if the products contain one or more pairs of enantiomers, don't worry about drawing each enantiomer with dash and wedge bonds. Just draw one molecule to represent each pair of enantiomers, using line bonds at the chiral center. More... No reaction. Explanation Check O + G 1. Na O Me Click and drag to start drawing a structure. 2. H + 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility 000 Ar Parrow_forwardDraw a tetramer of this alternating copolymer.arrow_forward
- H I T H HH H -H C. H- Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above. H- H CIH H H H HHHH H H 0 ·H H– 冊 CH CHI HH C- H- H H- H H A. H H C H H- -H HH H B. H- -H D. H H H H • H -H E. -H H H HICH T HHH F. H-arrow_forwardPolylactic acid (shown below) is a biodegradable polymer used for food packaging. Identify the monomer(s) used in the production of this polymer using a condensation process.arrow_forwardDraw the product of the reaction shown below. Ignore small byproducts that would evaporate pleasearrow_forward
- Poly(ethylene adipate) is a biodegradable polyester (shown below). Identify the type of polymerization process used in the production of this polymer.arrow_forwardPolymers may be composed of thousands of monomers. draw two repeat units(dimer) of the polymer formed in this reaction. assume there are hydrogen atoms on the two ends of the dimer. ignore inorganic byproducts pleasearrow_forwardDraw the product of the reaction shown below. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, Ignore inorganic byproductsarrow_forward
- Draw the product of this reaction please. Ignore inorganic byproductsarrow_forwardOne of the pi molecular orbitals of 1,3-butadiene (CH2=CHCH=CH2) is shown below. Please identify the number of nodal planes perpendicular to the bonding axisarrow_forwardDraw the monomers required to synthesize this condensation polymer please.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY