
(a)
Interpretation:
Synthesis of the given compound is to be shown.
Concept introduction:
The acetic acid is treated with various reagents to form products that contain different
Answer to Problem 21.85P
The synthesis of the given compound is as follows:
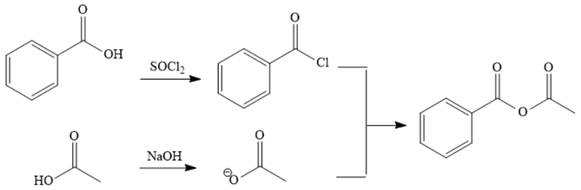
Explanation of Solution
The reaction requires converting a carboxylic acid to an acid anhydride, which represents going up the stability ladder. This is difficult to do directly but can be carried out by first producing an acid chloride. The acid chloride and the salt of acetic acid will make the desired anhydride.
The acetic acid is treated with various reagents to form products that contain different functional groups.
(b)
Interpretation:
The synthesis of the given compound is to be shown.
Concept introduction:
The acetic acid is treated with various reagents to form products that contain different functional groups. The new functional groups formed are anhydride, amine, ether, aldehyde, ketone, etc. When carboxylic acid converted to an acid anhydride, the stability ladder going up. When an anhydride reacted with ethanol to formed the ester, the stability ladder going down.
Answer to Problem 21.85P
The synthesis of the given compound is as follows:
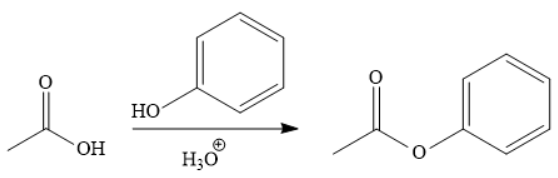
Explanation of Solution
The addition of phenol forms the ester under acidic condition, a Fischer esterification. This is a reversible reaction and can be carried out directly because the reactant and product species are on the same rung of the stability ladder.
The acetic acid is treated with various reagents to form products that contain different functional groups.
(c)
Interpretation:
The synthesis of the given compound is to be shown.
Concept introduction:
The acetic acid is treated with various reagents to form products that contain different functional groups. The new functional groups formed are anhydride, amine, ether, aldehyde, ketone, etc. When carboxylic acid converted to an acid anhydride, the stability ladder going up. When an anhydride reacted with ethanol to formed the ester, the stability ladder going down.
Answer to Problem 21.85P
The synthesis of the given compound is as follows,
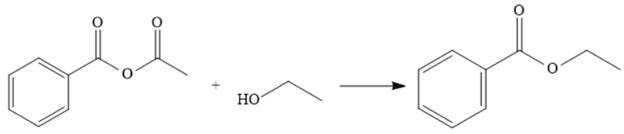
Explanation of Solution
The reaction of the anhydride from (a) with ethanol makes the ester. This represents going down the stability ladder and is done relatively easily.
The acetic acid is treated with various reagents to form products that contain different functional groups.
(d)
Interpretation:
The synthesis of the given compound is to be shown.
Concept introduction:
The acetic acid is treated with various reagents to form products that contain different functional groups. The new functional groups formed are anhydride, amine, ether, aldehyde, ketone, etc. When carboxylic acid converted to an acid anhydride, the stability ladder going up. When an anhydride reacted with ethanol to formed the ester, the stability ladder going down.
Answer to Problem 21.85P
The synthesis of the given compound is as follows:
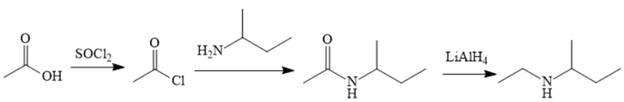
Explanation of Solution
The amine can be produced by reducing the corresponding amide. It is difficult to produce the amide directly from acetic acid because the amine that would be required for the nucleophile would deprotonate the carboxylic acid. The amide can be made relatively easily, however, by first converting acetic acid to acetyl chloride and then treating acetyl chloride with the amine.
The acetic acid is treated with various reagents to form products that contain different functional groups.
(e)
Interpretation:
The synthesis of the given compound is to be shown.
Concept introduction:
The acetic acid is treated with various reagents to form products that contain different functional groups. The new functional groups formed are anhydride, amine, ether, aldehyde, ketone, etc. When carboxylic acid converted to an acid anhydride, the stability ladder going up. When an anhydride reacted with ethanol to formed the ester, the stability ladder going down.
Answer to Problem 21.85P
The synthesis of the given compound is as follows,
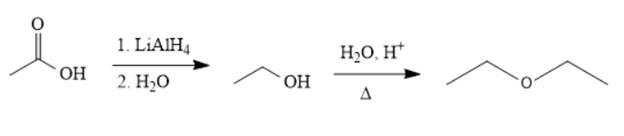
Explanation of Solution
Reducing the acid to an alcohol, followed by dehydration, leads to the ether. Alternatively, ethanol can be deprotonated by
The acetic acid is treated with various reagents to form products that contain different functional groups.
(f)
Interpretation:
The synthesis of the given compound is to be shown.
Concept introduction:
The acetic acid is treated with various reagents to form products that contain different functional groups. The new functional groups formed are anhydride, amine, ether, aldehyde, ketone, etc. When carboxylic acid converted to an acid anhydride, the stability ladder going up. When an anhydride reacted with ethanol to formed the ester, the stability ladder going down.
Answer to Problem 21.85P
The synthesis of the given compound is as follows,
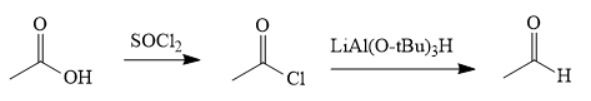
Explanation of Solution
First, acid is treated with
The acetic acid is treated with various reagents to form products which contain different functional groups.
(g)
Interpretation:
The synthesis of the given compound is to be shown.
Concept introduction:
The acetic acid is treated with various reagents to form products that contain different functional groups. The new functional groups formed are anhydride, amine, ether, aldehyde, ketone, etc. When carboxylic acid converted to an acid anhydride, the stability ladder going up. When an anhydride reacted with ethanol to formed the ester, the stability ladder going down.
Answer to Problem 21.85P
The synthesis of the given compound is as follows,
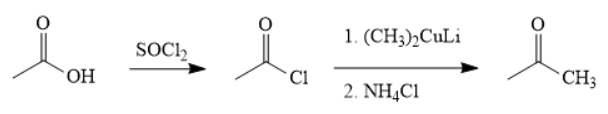
Explanation of Solution
First, acid is treated with
The acetic acid is treated with various reagents to form products which contain different functional groups.
Want to see more full solutions like this?
Chapter 21 Solutions
EBK ORGANIC CHEMISTRY: PRINCIPLES AND M
- Synthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIf possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forwardSynthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Indicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forwardIndicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forward
- Indicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forwardQuestion 3 (4 points), Draw a full arrow-pushing mechanism for the following reaction Please draw all structures clearly. Note that this intramolecular cyclization is analogous to the mechanism for halohydrin formation. COH Br + HBr Brarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





