
a)
Interpretation:
The structural formula of 2, 3- dimethylheptane should be stated.
Concept Introduction:
The systematic structure determination of organic compound is given by IUPAC. The name of the organic compound is determined from the structure that is by analyzing the position of groups substituted on it.
a)
Answer to Problem 63A
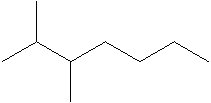
The structural formula of the compound 2, 3- dimethylheptane is:
Explanation of Solution
In the structure; the parent atom contains seven carbon atom.so, the word root is heptane.
At 2 and 3 position there are two methyl groups which is named as dimethyl.
Therefore; the structure of the given molecule is

(b)
Interpretation:
The structural formula of 2, 2-dimethyl-3-chloro-1-octanol should be stated.
Concept Introduction:
The systematic structure determination of organic compound is given by IUPAC. The name of the organic compound is determined from the structure that is by analyzing the position of groups substituted on it.
(b)
Answer to Problem 63A
The structural formula of the compound 2, 2-dimethyl-3-chloro-1-octanol is
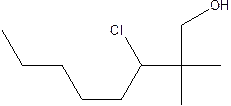
Explanation of Solution
In the structure; the parent chain contains eight atoms so; the word root is octane.
At position 2 there are 2 methyl groups are present which is named as dimethyl, at 3rd position chlorine is present which is named as chloroand at position 1, the OH group is present which is names as ‘ol’
Therefore, the structure of the given molecule is
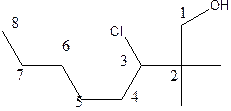
C)
Interpretation:
The structural formula of 2-chloro-1-hexene should be stated.
Concept Introduction:
The systematic structure determination of organic compound is given by IUPAC. The name of the organic compound is determined from the structure that is by analyzing the position of groups substituted on it.
C)
Answer to Problem 63A
The structural formula ofthe compound 2-chloro-1-hexene is
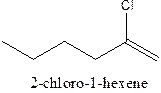
Explanation of Solution
In the structure; the parent chain contains the six carbon atoms and the word root is hexane.
At position 1, there is
Therefore; the structure of the given molecule is
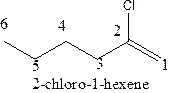
d)
Interpretation:
The structural formula of 1- chloro-2-hexene should be stated.
Concept Introduction:
The systematic structure determination of organic compound is given by IUPAC. The name of the organic compound is determined from the structure that is by analyzing the position of groups substituted on it.
d)
Answer to Problem 63A
The structural formula ofthe compound 1- chloro-2-hexene is
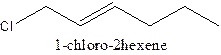
Explanation of Solution
In the structure; the parent chain contains the six carbon atoms and the word root is hexane.
At position 1, there is chlorine group which is named as chloroand at position 2, alkene group is present.which is names as ‘ene’.
Therefore, the structure of the given molecule is
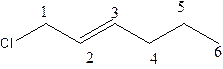
e)
Interpretation:
The structural formula of 2- methylphenol should be stated.
Concept Introduction:
The systematic structure determination of organic compound is given by IUPAC. The name of the organic compound is determined from the structure that is by analyzing the position of groups substituted on it.
e)
Answer to Problem 63A
The structural formula of the compound 2- methylphenol is
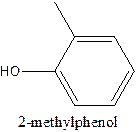
Explanation of Solution
In the structure, the parent ring contains six carbon atom which is named as benzene ring. At position 1, the methyl group present and at 2nd position there is OH group present which is named as ‘ol’.
Therefore; the structure of the given molecule is
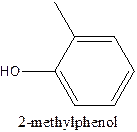
Chapter 20 Solutions
World of Chemistry
- Add substituents to draw the conformer below (sighting down the indicated bond), then rotate the back carbon to provide the anti staggered conformer. + H3C H Ph H Problem 25 of 30 Drawing Atoms, Bonds and Rings Charges Tap a node to see suggestions H H H Undo Rasat Remove Done Finish update Rotate Submitarrow_forwardwhat temperature does a 50% (mole fraction) of ammonia/water liquid mixture boil at 1 atmarrow_forward1) Suppose 0.1 kg ice at 0°C (273K) is in 0.5kg water at 20°C (293K). What is the change in entropy of the ice as it melts at 0°? To produce the original "water gas" mixture, carbon (in a combustible form known as coke) is reacted with steam: 131.4 kJ + H20(g) + C(s) → CO(g) + H2(g) From this information and the equations in the previous problem, calculate the enthalpy for the combustion or carbon to form carbon dioxide. kindly show me how to solve both parts of the same long problem. Thanksarrow_forward
- we were assigned to dilute 900ppm in to 18ppm by using only 250ml vol flask. firstly we did calc and convert 900ppm to 0.9 ppm to dilute in 1 liter. to begin the experiment we took 0,225g of kmno4 and dissolved in to 250 vol flask. then further we took 10 ml sample sol and dissolved in to 100 ml vol flask and put it in to a spectrometer and got value of 0.145A . upon further calc we got v2 as 50ml . need to find DF, % error (expval and accptVal), molarity, molality. please write the whole report. thank you The format, tables, introduction, procedure and observation, result, calculations, discussion and conclusionarrow_forwardQ5. Predict the organic product(s) for the following transformations. If no reaction will take place (or the reaction is not synthetically useful), write "N.R.". Determine what type of transition state is present for each reaction (think Hammond Postulate). I Br₂ CH3 F2, light CH3 Heat CH3 F₂ Heat Br2, light 12, light CH3 Cl2, light Noarrow_forwardNonearrow_forward
- In the phase diagram of steel (two components Fe and C), region A is the gamma austenite solid and region B contains the gamma solid and liquid. Indicate the degrees of freedom that the fields A and B have,arrow_forwardFor a condensed binary system in equilibrium at constant pressure, indicate the maximum number of phases that can exist.arrow_forwardPart V. Label ad match the carbons in compounds Jane and Diane w/ the corresponding peak no. in the Spectra (Note: use the given peak no. To label the carbons, other peak no are intentionally omitted) 7 4 2 -0.13 -0.12 -0.11 -0.10 -0.08 8 CI Jane 1 -0.09 5 210 200 190 180 170 160 150 140 130 120 110 100 -8 90 f1 (ppm) 11 8 172.4 172.0 f1 (ppr HO CI NH Diane 7 3 11 80 80 -80 -R 70 60 60 2 5 -8 50 40 8. 170 160 150 140 130 120 110 100 90 -0 80 70 20 f1 (ppm) 15 30 -20 20 -60 60 -0.07 -0.06 -0.05 -0.04 -0.03 -0.02 -0.01 -0.00 -0.01 10 -0.17 16 15 56 16 -0.16 -0.15 -0.14 -0.13 -0.12 -0.11 -0.10 -0.09 -0.08 -0.07 -0.06 -0.05 -0.04 17.8 17.6 17.4 17.2 17.0 f1 (ppm) -0.03 -0.02 550 106 40 30 20 20 -0.01 -0.00 F-0.01 10 0arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





