
Concept explainers
Interpretation:
The order to name of a hydrocarbon with several substituentshave to be described.
Concept Introduction:
Alkanes can be classified as-
- Straight-chain alkanes
- Branched-chain alkanes
Numbering of the parent chain is necessary to find the position of substituents in the longest chain of a hydrocarbon.
Answer to Problem 15A
When a substituent is attached to a carbon chain, numbering of the carbon chain will start from that end which is closest to the substituent. When several substituents are present in the long chain hydrocarbon, the order is arranged by the name of the substituents in alphabetical order. As for example, the following compound contains three substituents such as chloro, ethyl and methyl. So, the substituent (-Cl) will be placed first in the order and methyl substituent will be the last one.
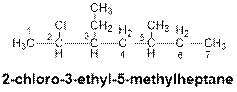
Explanation of Solution
When a substituent is attached to a carbon chain, numbering of the carbon chain will start from that end which is closest to the substituent. When several substituents are present in the long chain hydrocarbon, the order is arranged by the name of the substituents in alphabetical order. As for example the following compound contains three substituents such as chloro, ethyl and methyl. So, the substituent (-Cl) will be placed first in the order and methyl substituent will be the last one.
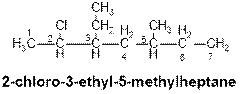
Chapter 20 Solutions
World of Chemistry
- The chemical formula of "benzimidazole E" is C7H6N2. Draw it.arrow_forwardBriefly comment (without formulas) on the steps of the aldol condensation mechanism in acidic and basic media.arrow_forwardThe following intermediates are to proceed by acetoacetic ester synthesis. What are intermediates 1 and 2 plus the final product 3? Please include a detailed explanation and drawings of the intermediates and how they occurred.arrow_forward
- What are the missing intermediates 1, 2, and 3? Please include a detailed explanation explaining the steps of malonic ester synthesis. Please include drawings of the intermediates and how they occur.arrow_forwardWhat is the reactant that makes the following product of the reaction? Please provide a detailed explanation and a drawing to show how the reaction proceeds.arrow_forwardDraw the products formed when each ester is hydrolyzed with water and sulfuric acid.arrow_forward
- Draw the complete structural formula from each condensed structure. include all hydrogen atoms.arrow_forwardDraw the complete structural formula from each condensed structure. Include all hydrogen atoms.arrow_forwardIndicate how H2O2 intervenes in the synthesis of K4[Co2(C2O4)4(OH)2]. Write the reactions.arrow_forward
- Explain how, based on physical gas adsorption isotherms, we can determine whether multi-walled C nanotubes are open at their ends. Explain this.arrow_forwardcan somone answer pleasearrow_forwardConstruct a molecular orbital energy-level diagram for BeH2. Sketch the MO pictures (schematic representation) for the HOMO and LUMO of BeH2 [Orbital Potential Energies, H (1s): -13.6 eV; Be (2s): -9.3 eV, Be (2p): -6.0 eV]arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





