
a) Styrene (PhCH=CH2)
Interpretation:
How to prepare styrene from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols undergo dehydration when treated with POCl3 in pyridine to yield
To state:
How to prepare styrene from 2-phenylethanol.
Answer to Problem 47AP
Styrene can be prepared by treating 2-phenylethanol with POCl3 in pyridine.
Explanation of Solution
2-Phenylethanol when treated with POCl3 in pyridine eliminates a molecule of water from the side chain to yield styrene.
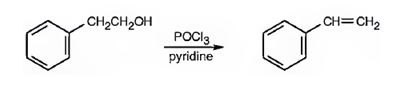
Styrene can be prepared by treating 2-phenylethanol with POCl3 in pyridine.
b) Phenylacetaldehyde (PhCH2CHO)
Interpretation:
How to prepare phenylacetaldehyde from 2-phenylethanol is to be stated.
Concept introduction:
Dess-Martin periodinate in dichloromethane oxidizes 10alcohols to
To state:
How to prepare phenylacetaldehyde from 2-phenylethanol
Answer to Problem 47AP
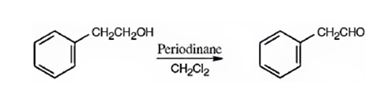
Phenylacetaldehyde can be prepared by oxidizing 2-phenylethanol with Dess-Martin periodinate in dichloromethane.
Explanation of Solution
2-phenylethanol is a 10 alcohol. It gets oxidized to phenylacetaldehyde when treated with Dess-Martin periodinate in dichloromethane.
Phenylacetaldehyde can be prepared by oxidizing 2-phenylethanol with Dess-Martin periodinate in dichloromethane.

c) Phenylacetic acid (PhCH2COOH)
Interpretation:
How to prepare phenylacetic acid from 2-phenylethanol is to be stated.
Concept introduction:
CrO3 in acidic solutions oxidize 10 alcohols directly into acids and 20 alcohols to ketones. It does not oxidize 30 alcohols.
To state:
How to prepare phenylacetic acid from 2-phenylethanol.
Answer to Problem 47AP
Phenylacetic acid can be prepared by oxidizing 2-phenylethanol with CrO3 in acidic solutions.
Explanation of Solution
2-phenylethanol is a 10 alcohol. It gets oxidized to phenylacetic acid when treated with CrO3 in acidic solutions.
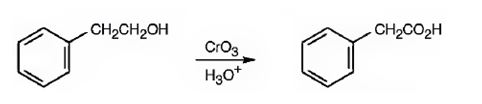
Phenylacetic acid can be prepared by oxidizing 2-phenylethanol with CrO3 in acidic solutions.
d) Benzoic acid
Interpretation:
How to prepare benzoic acid from 2-phenylethanol is to be stated.
Concept introduction:
KMnO4 in acidic solutions oxidize
To state:
How to prepare benzoic acid from 2-phenylethanol.
Answer to Problem 47AP
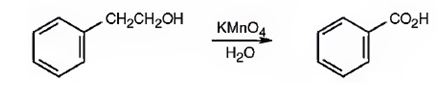
Benzoic acid can be prepared by oxidizing 2-phenylethanol with KMnO4 in acidic solutions.
Explanation of Solution
2-phenylethanol is a 10 alcohol. It gets oxidized to benzoic acid when treated with KMnO4 in acidic solutions.
Benzoic acid can be prepared by oxidizing 2-phenylethanol with KMnO4 in acidic solutions.
e) Ethylbenzene
Interpretation:
How to prepare ethylbenzene from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols undergo dehydration when treated with POCl3 in pyridine to yield an alkene. The alkene upon reduction gives the
To state:
How to prepare ethylbenzene from 2-phenylethanol.
Answer to Problem 47AP
Ethylbenzene can be prepared from 2-phenylethanol by following the steps shown below.
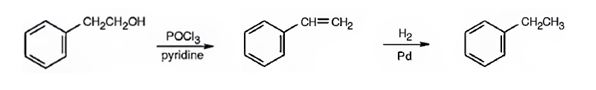
Explanation of Solution
2-Phenylethanol when treated with POCl3 in pyridine eliminates a molecule of water from the side chain to yield styrene. When treated with H2/Pd, the double bond in the side chain gets reduced to yield ethyl benzene.
Ethylbenzene can be prepared from 2-phenylethanol by following the steps shown below.

f) benzaldehyde
Interpretation:
How to prepare benzaldehyde from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols undergo dehydration when treated with POCl3 in pyridine to yield an alkene. The alkene upon ozonolyzis followed by reduction will yield the aldehyde required.
To state:
How to prepare benzaldehyde from 2-phenylethanol.
Answer to Problem 47AP
Benzaldehyde can be prepared from 2-phenylethanol by following the steps shown below.
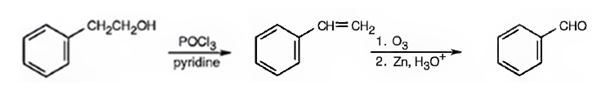
Explanation of Solution
2-Phenylethanol when treated with POCl3 in pyridine eliminates a molecule of water from the side chain to yield styrene. When styrene is subjected to ozonolysis followed by reduction, the double bond in side chain gets cleaved resulting in the formation of benzaldehyde.
Benzaldehyde can be prepared from 2-phenylethanol by following the steps shown below.

g) 1-phenylethanol
Interpretation:
How to prepare 1-phenylethanol from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols undergo dehydration when treated with POCl3 in pyridine to yield an alkene. The alkene adds a molecule of water following oxymercuration-demercuration process. The addition will take place following Markovnikov regiochemistry.
To state:
How to prepare 1-phenylethanol from 2-phenylethanol.
Answer to Problem 47AP
1- Phenylethanol can be prepared from 2-phenylethanol by following the steps shown below.
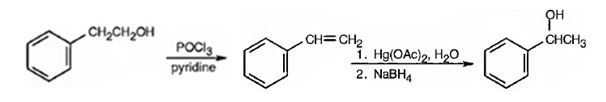
Explanation of Solution
2-Phenylethanol, when treated with POCl3 in pyridine eliminates a molecule of water from the side chain to yield styrene. When styrene is subjected oxymercuration-demercuration processes, a molecule of water is added, following Markovnikov regiochemistry, to the double bond. The –OH adds on to the more alkyl substituted carbon and H to the less alkyl substituted carbon in double bond to yield 1-phenylethanol.
1-Phenylethanol can be prepared from 2-phenylethanol by following the steps shown below.

h) 1-Bromo-2-phenylethane
Interpretation:
How to prepare 1-bromo-2-phenylethane from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols yield the corresponding alkyl bromides when treated with PBr3.
To state:
How to prepare 1-bromo-2-phenylethane from 2-phenylethanol is to be stated.
Answer to Problem 47AP
1-Bromo-2-phenylethane can be prepared from 2-phenylethanol by following the steps shown below.
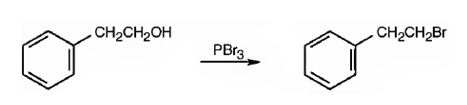
Explanation of Solution
When 2-phenylethanol is treated with PBr3, a bimolecular nucleophilic substitution of –OH by Br takes place to yield 1-bromo-2-phenylethane.
1-Bromo-2-phenylethane can be prepared from 2-phenylethanol by following the steps shown below.

Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- Calculating the pH at equivalence of a titration Try Again Your answer is incorrect. 0/5 a A chemist titrates 70.0 mL of a 0.7089 M hydrocyanic acid (HCN) solution with 0.4574M KOH solution at 25 °C. Calculate the pH at equivalence. The pK of hydrocyanic acid is 9.21. Round your answer to 2 decimal places. Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of KOH solution added. pH = 11.43] G 00. 18 Ar B•arrow_forwardBiological Macromolecules Naming and drawing the products of aldose oxidation and reduction aw a Fischer projection of the molecule that would produce L-ribonic acid if it were subjected to mildly oxidizing reaction conditions. Click and drag to start drawing a structure. X AP ‡ 1/5 Naor Explanation Check McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibilarrow_forward● Biological Macromolecules Identifying the parts of a disaccharide Take a look at this molecule, and then answer the questions in the table below it. CH2OH O H H H OH OH OH H H CH2OH H O OH H OH H H H H OH Is this a reducing sugar? Does this molecule contain a glycosidic bond? If you said this molecule does contain a glycosidic bond, write the symbol describing it. If you said this molecule does contain a glycosidic bond, write the common names (including anomer and enantiomer labels) of the molecules that would be released if that bond were hydrolyzed. If there's more than one molecule, separate each name with a comma. Explanation Check O yes X O no ○ yes O no Uarrow_forward
- The aim of the lab is to measure the sodium content from tomato sauce using the Mohr titration method. There are two groups being: Regular Tomato sauce & Salt Reduced tomato sauce QUESTION: State how you would prepare both Regular & Salt reduced tomato sauce samples for chemical analysis using the Mohr titration methodarrow_forwardUsing the conditions of spontaneity to deduce the signs of AH and AS Use the observations about each chemical reaction in the table below to decide the sign (positive or negative) of the reaction enthalpy AH and reaction entropy AS. Note: if you have not been given enough information to decide a sign, select the "unknown" option. reaction observations conclusions A The reverse of this reaction is always spontaneous but proceeds faster at temperatures above -48. °C. ΔΗ is (pick one) ✓ AS is (pick one) B This reaction is spontaneous except below 114. °C but proceeds at a slower rate below 135. °C. ΔΗ is (pick one) AS is (pick one) ΔΗ is C This reaction is exothermic and proceeds faster at temperatures above -43. °C. (pick one) AS is (pick one) v Х 5 ? 18 Ararrow_forwardion. A student proposes the following Lewis structure for the perchlorate (CIO) io : :0: : Cl : - - : :0: ك Assign a formal charge to each atom in the student's Lewis structure. atom central O formal charge ☐ top O ☐ right O ☐ bottom O ☐ Cl ☐arrow_forward
- Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure Yes. Is the proposed Lewis structure reasonable? Cl- : 2: :Z: :Z: N—N : 0: C C1: O CO No, it has the wrong number of valence electrons. The correct number is: ☐ No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* ☐ Yes. No, it has the wrong number of valence electrons. The correct number is: ☐ No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* | Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* | If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many times as necessary. For example, if two oxygen atoms don't satisfy the octet rule, enter "0,0". ☑arrow_forwardUse the observations about each chemical reaction in the table below to decide the sign (positive or negative) of the reaction enthalpy AH and reaction entropy AS. Note: if you have not been given enough information to decide a sign, select the "unknown" option. reaction observations conclusions ΔΗ is (pick one) A This reaction is faster above 103. °C than below. AS is (pick one) ΔΗ is (pick one) B This reaction is spontaneous only above -9. °C. AS is (pick one) ΔΗ is (pick one) C The reverse of this reaction is always spontaneous. AS is (pick one) 18 Ararrow_forwardUse the observations about each chemical reaction in the table below to decide the sign (positive or negative) of the reaction enthalpy AH and reaction entropy AS. Note: if you have not been given enough information to decide a sign, select the "unknown" option. reaction observations conclusions A The reverse of this reaction is always spontaneous but proceeds slower at temperatures below 41. °C. ΔΗ is (pick one) AS is (pick one) ΔΗ is (pick one) B This reaction is spontaneous except above 94. °C. AS is (pick one) This reaction is always spontaneous, but ΔΗ is (pick one) C proceeds slower at temperatures below −14. °C. AS is (pick one) Х 00. 18 Ar 무ㅎ B 1 1arrow_forward
- Draw the product of the reaction shown below. Ignore inorganic byproducts. + H CH3CH2OH HCI Drawingarrow_forwardplease explain this in simple termsarrow_forwardK Most Reactive Na (3 pts) Can the metal activity series (shown on the right) or a standard reduction potential table explain why potassium metal can be prepared from the reaction of molten KCI and Na metal but sodium metal is not prepared from the reaction of molten NaCl and K metal? Show how (not). Ca Mg Al с Zn Fe Sn Pb H Cu Ag Au Least Reactivearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





