
Concept explainers
(a)
Interpretation:
IUPAC name for the parent amine from which the given amine salt is formed has to be given.
Concept Introduction:
IUPAC rules for naming ammonium ions:
There are two rules that has to be followed while naming a positive ion (ammonium ion) and they are,
- In order to name an alkylamine, the ending of the name amine is changed from amine to ammonium ion.
- In order to name an
aromatic amine, the ending of the name “-e” is replaced by “-ium ion”.
- Longest carbon chain has to be identified that is attached to nitrogen atom.
- Suffix “-e” in name of the parent chain
alkane is replaced by “-amine”. - Numbering of the carbon chain is done from the end that is near the nitrogen atom.
- Point of attachment of the nitrogen atom in the carbon chain is indicated by a number before the parent chain name.
- In case if substituents are present, then the identity and location of substituents are appended to the front in the parent chain name.
If the compound contains two amine groups, then the suffix “-e” is replaced by diamine. Tertiary and secondary
Common name for amine is given in a single word. Primary amine is named as alkylamine. Secondary amine is named as alkylalkylamine. Tertiary amine is named as alkylalkylalkylamine.
(a)
Answer to Problem 17.58EP
Name of the parent amine is N-methylethanamine.
Explanation of Solution
Given amine salt is,
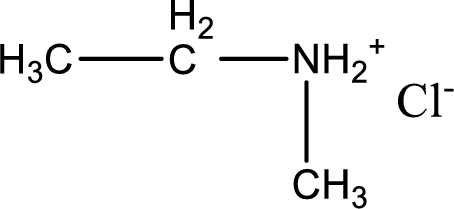
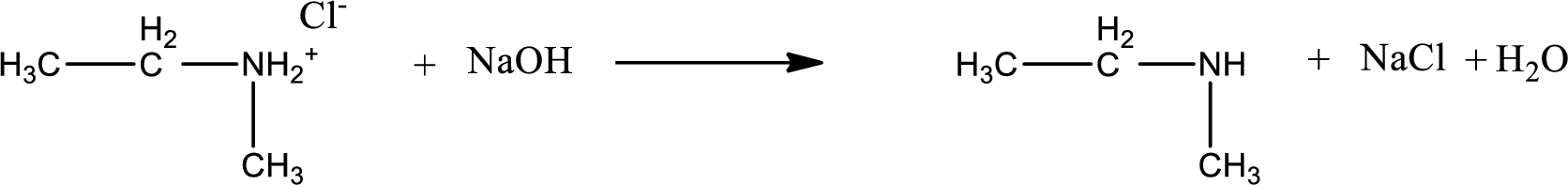
The parent amine can be found by deprotonating the amine salt. This can be accomplished by treating it with a strong base. The complete reaction can be given as,
Structure of the amine is,
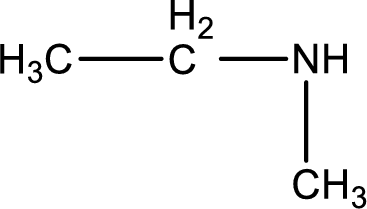
The longest carbon chain attached to the nitrogen atom is found to be containing two carbon atoms. Hence, the parent alkane is ethane. Amine is named by replacing the suffix “-e” in the parent alkane name with “-amine”. This gives the name as ethanamine.
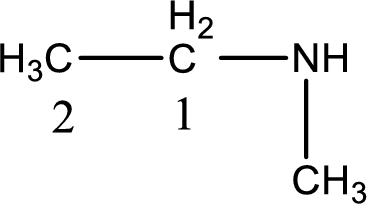
Numbering has to be given from the end that is near to the nitrogen atom. In this case, the nitrogen atom is attached to the carbon atom that is numbered 1. In this case numbering does not make sense as only two carbon atoms are present. Looking for substituent a methyl group is present on the nitrogen atom. This gives the IUPAC name of N-methylethanamine.
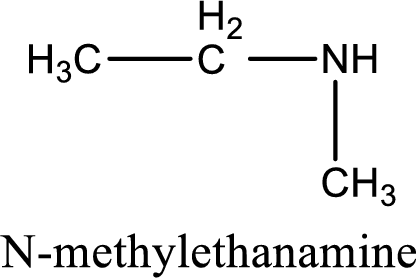
IUPAC name for the parent amine is given.
(b)
Interpretation:
IUPAC name for the parent amine from which the given amine salt is formed has to be given.
Concept Introduction:
IUPAC rules for naming ammonium ions:
There are two rules that has to be followed while naming a positive ion (ammonium ion) and they are,
- In order to name an alkylamine, the ending of the name amine is changed from amine to ammonium ion.
- In order to name an aromatic amine, the ending of the name “-e” is replaced by “-ium ion”.
IUPAC nomenclature for amine: There are about five rules to be followed in giving IUPAC name for an amine.
- Longest carbon chain has to be identified that is attached to nitrogen atom.
- Suffix “-e” in name of the parent chain alkane is replaced by “-amine”.
- Numbering of the carbon chain is done from the end that is near the nitrogen atom.
- Point of attachment of the nitrogen atom in the carbon chain is indicated by a number before the parent chain name.
- In case if substituents are present, then the identity and location of substituents are appended to the front in the parent chain name.
If the compound contains two amine groups, then the suffix “-e” is replaced by diamine. Tertiary and secondary amines are named as N-substituted primary amines.
Common name for amine is given in a single word. Primary amine is named as alkylamine. Secondary amine is named as alkylalkylamine. Tertiary amine is named as alkylalkylalkylamine.
(b)
Answer to Problem 17.58EP
Name of the parent amine is 1-butanamine.
Explanation of Solution
Given amine salt is
The parent amine can be found by deprotonating the amine salt. This can be accomplished by treating it with a strong base. The complete reaction can be given as,

Structure of the amine is,
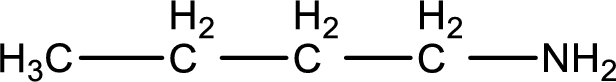
The longest carbon chain attached to the nitrogen atom is found to be containing four carbon atoms. Hence, the parent alkane is butane. Amine is named by replacing the suffix “-e” in the parent alkane name with “-amine”. This gives the name as butanamine.
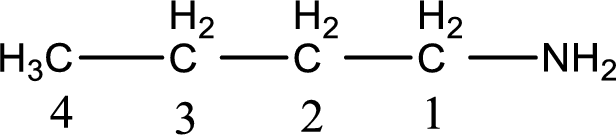
Numbering has to be given from the end that is near to the nitrogen atom. In this case, the nitrogen atom is attached to the carbon atom that is numbered 1. This has to be added to the name in front. This gives the IUPAC name of 1-butanamine.
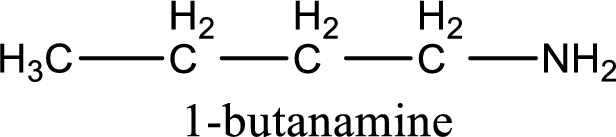
IUPAC name for the parent amine is given.
(c)
Interpretation:
IUPAC name for the parent amine from which the given amine salt is formed has to be given.
Concept Introduction:
IUPAC rules for naming ammonium ions:
There are two rules that has to be followed while naming a positive ion (ammonium ion) and they are,
- In order to name an alkylamine, the ending of the name amine is changed from amine to ammonium ion.
- In order to name an aromatic amine, the ending of the name “-e” is replaced by “-ium ion”.
IUPAC nomenclature for amine: There are about five rules to be followed in giving IUPAC name for an amine.
- Longest carbon chain has to be identified that is attached to nitrogen atom.
- Suffix “-e” in name of the parent chain alkane is replaced by “-amine”.
- Numbering of the carbon chain is done from the end that is near the nitrogen atom.
- Point of attachment of the nitrogen atom in the carbon chain is indicated by a number before the parent chain name.
- In case if substituents are present, then the identity and location of substituents are appended to the front in the parent chain name.
If the compound contains two amine groups, then the suffix “-e” is replaced by diamine. Tertiary and secondary amines are named as N-substituted primary amines.
Common name for amine is given in a single word. Primary amine is named as alkylamine. Secondary amine is named as alkylalkylamine. Tertiary amine is named as alkylalkylalkylamine.
(c)
Answer to Problem 17.58EP
Name of the parent amine is N,N-dimethyl-2-propanamine.
Explanation of Solution
Given amine salt is,
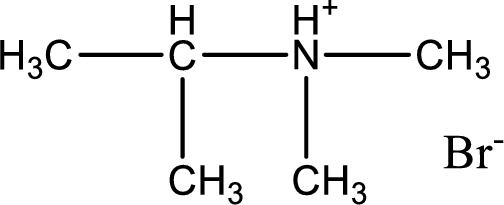
The parent amine can be found by deprotonating the amine salt. This can be accomplished by treating it with a strong base. The complete reaction can be given as,
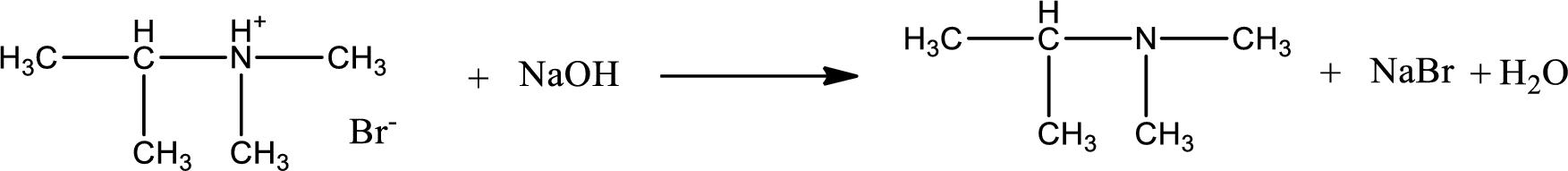
Structure of the amine is,
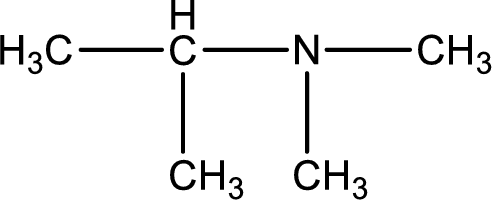
The longest carbon chain attached to the nitrogen atom is found to be containing three carbon atoms. Hence, the parent alkane is propane. Amine is named by replacing the suffix “-e” in the parent alkane name with “-amine”. This gives the name as propanamine.
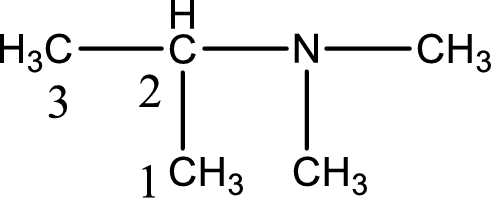
Numbering has to be given from the end that is near to the nitrogen atom. In this case, the nitrogen atom is attached to the carbon atom that is numbered 2. Looking for substituents, two methyl groups are present on the nitrogen atom. This gives the IUPAC name of N,N-dimethyl-2-propanamine.
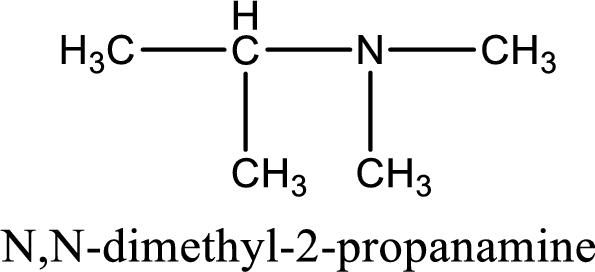
IUPAC name for the parent amine is given.
(d)
Interpretation:
IUPAC name for the parent amine from which the given amine salt is formed has to be given.
Concept Introduction:
IUPAC rules for naming ammonium ions:
There are two rules that has to be followed while naming a positive ion (ammonium ion) and they are,
- In order to name an alkylamine, the ending of the name amine is changed from amine to ammonium ion.
- In order to name an aromatic amine, the ending of the name “-e” is replaced by “-ium ion”.
IUPAC nomenclature for amine: There are about five rules to be followed in giving IUPAC name for an amine.
- Longest carbon chain has to be identified that is attached to nitrogen atom.
- Suffix “-e” in name of the parent chain alkane is replaced by “-amine”.
- Numbering of the carbon chain is done from the end that is near the nitrogen atom.
- Point of attachment of the nitrogen atom in the carbon chain is indicated by a number before the parent chain name.
- In case if substituents are present, then the identity and location of substituents are appended to the front in the parent chain name.
If the compound contains two amine groups, then the suffix “-e” is replaced by diamine. Tertiary and secondary amines are named as N-substituted primary amines.
Common name for amine is given in a single word. Primary amine is named as alkylamine. Secondary amine is named as alkylalkylamine. Tertiary amine is named as alkylalkylalkylamine.
(d)
Answer to Problem 17.58EP
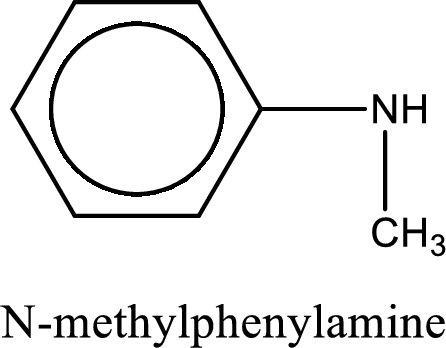
Name of the parent amine is N-methylphenylamine.
Explanation of Solution
Given amine salt is,
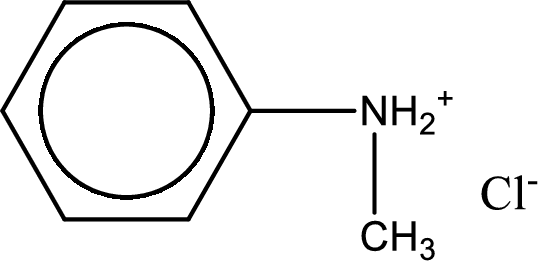
The parent amine can be found by deprotonating the amine salt. This can be accomplished by treating it with a strong base. The complete reaction can be given as,
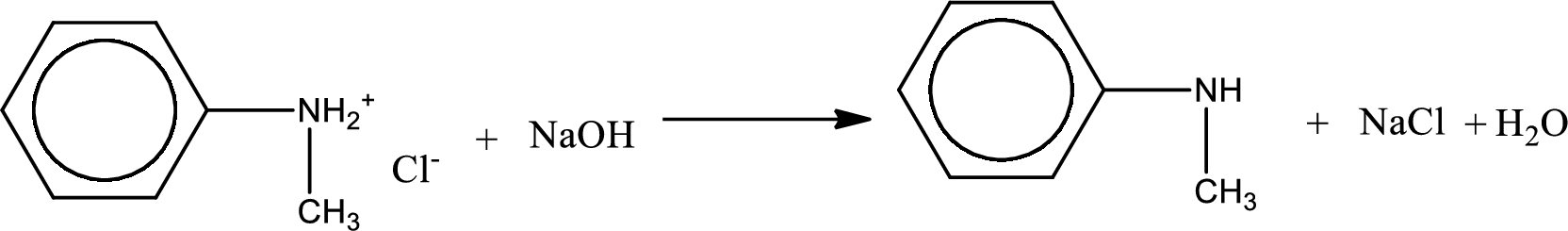
Structure of the amine is,
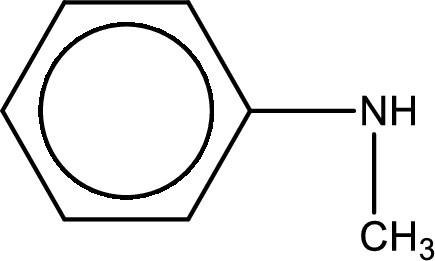
The longest carbon chain attached to the nitrogen atom is found to be containing six carbon cyclic chain. Hence, the parent is benzene. This has to be named as phenylamine.
Looking for substituents, one methyl group is present on the nitrogen atom. This gives the IUPAC name of N-methylphenylamine.
IUPAC name for the parent amine is given.
Want to see more full solutions like this?
Chapter 17 Solutions
General, Organic, and Biological Chemistry Seventh Edition
- If you had an unknown microbe, what steps would you take to determine what type of microbe (e.g., fungi, bacteria, virus) it is? Are there particular characteristics you would search for? Explain.arrow_forwardavorite Contact avorite Contact favorite Contact ୫ Recant Contacts Keypad Messages Pairing ง 107.5 NE Controls Media Apps Radio Nav Phone SCREEN OFF Safari File Edit View History Bookmarks Window Help newconnect.mheducation.com M Sign in... S The Im... QFri May 9 9:23 PM w The Im... My first.... Topic: Mi Kimberl M Yeast F Connection lost! You are not connected to internet Sigh in... Sign in... The Im... S Workin... The Im. INTRODUCTION LABORATORY SIMULATION Tube 1 Fructose) esc - X Tube 2 (Glucose) Tube 3 (Sucrose) Tube 4 (Starch) Tube 5 (Water) CO₂ Bubble Height (mm) How to Measure 92 3 5 6 METHODS RESET #3 W E 80 A S D 9 02 1 2 3 5 2 MY NOTES LAB DATA SHOW LABELS % 5 T M dtv 96 J: ப 27 כ 00 alt A DII FB G H J K PHASE 4: Measure gas bubble Complete the following steps: Select ruler and place next to tube 1. Measure starting height of gas bubble in respirometer 1. Record in Lab Data Repeat measurement for tubes 2-5 by selecting ruler and move next to each tube. Record each in Lab Data…arrow_forwardCh.23 How is Salmonella able to cross from the intestines into the blood? A. it is so small that it can squeeze between intestinal cells B. it secretes a toxin that induces its uptake into intestinal epithelial cells C. it secretes enzymes that create perforations in the intestine D. it can get into the blood only if the bacteria are deposited directly there, that is, through a puncture — Which virus is associated with liver cancer? A. hepatitis A B. hepatitis B C. hepatitis C D. both hepatitis B and C — explain your answer thoroughlyarrow_forward
- Ch.21 What causes patients infected with the yellow fever virus to turn yellow (jaundice)? A. low blood pressure and anemia B. excess leukocytes C. alteration of skin pigments D. liver damage in final stage of disease — What is the advantage for malarial parasites to grow and replicate in red blood cells? A. able to spread quickly B. able to avoid immune detection C. low oxygen environment for growth D. cooler area of the body for growth — Which microbe does not live part of its lifecycle outside humans? A. Toxoplasma gondii B. Cytomegalovirus C. Francisella tularensis D. Plasmodium falciparum — explain your answer thoroughlyarrow_forwardCh.22 Streptococcus pneumoniae has a capsule to protect it from killing by alveolar macrophages, which kill bacteria by… A. cytokines B. antibodies C. complement D. phagocytosis — What fact about the influenza virus allows the dramatic antigenic shift that generates novel strains? A. very large size B. enveloped C. segmented genome D. over 100 genes — explain your answer thoroughlyarrow_forwardWhat is this?arrow_forward
- Molecular Biology A-C components of the question are corresponding to attached image labeled 1. D component of the question is corresponding to attached image labeled 2. For a eukaryotic mRNA, the sequences is as follows where AUGrepresents the start codon, the yellow is the Kozak sequence and (XXX) just represents any codonfor an amino acid (no stop codons here). G-cap and polyA tail are not shown A. How long is the peptide produced?B. What is the function (a sentence) of the UAA highlighted in blue?C. If the sequence highlighted in blue were changed from UAA to UAG, how would that affecttranslation? D. (1) The sequence highlighted in yellow above is moved to a new position indicated below. Howwould that affect translation? (2) How long would be the protein produced from this new mRNA? Thank youarrow_forwardMolecular Biology Question Explain why the cell doesn’t need 61 tRNAs (one for each codon). Please help. Thank youarrow_forwardMolecular Biology You discover a disease causing mutation (indicated by the arrow) that alters splicing of its mRNA. This mutation (a base substitution in the splicing sequence) eliminates a 3’ splice site resulting in the inclusion of the second intron (I2) in the final mRNA. We are going to pretend that this intron is short having only 15 nucleotides (most introns are much longer so this is just to make things simple) with the following sequence shown below in bold. The ( ) indicate the reading frames in the exons; the included intron 2 sequences are in bold. A. Would you expected this change to be harmful? ExplainB. If you were to do gene therapy to fix this problem, briefly explain what type of gene therapy youwould use to correct this. Please help. Thank youarrow_forward
- Molecular Biology Question Please help. Thank you Explain what is meant by the term “defective virus.” Explain how a defective virus is able to replicate.arrow_forwardMolecular Biology Explain why changing the codon GGG to GGA should not be harmful. Please help . Thank youarrow_forwardStage Percent Time in Hours Interphase .60 14.4 Prophase .20 4.8 Metaphase .10 2.4 Anaphase .06 1.44 Telophase .03 .72 Cytukinesis .01 .24 Can you summarize the results in the chart and explain which phases are faster and why the slower ones are slow?arrow_forward
 Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College- Essentials of Pharmacology for Health ProfessionsNursingISBN:9781305441620Author:WOODROWPublisher:Cengage
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning





