
(a)
Interpretation:
Given compound has to be classified as an amine, an amide, or both amine and amide.
Concept Introduction:
Organic compounds are the important basis of life. They include gasoline, coal, dyes, and clothing fibers etc. The compounds that are obtained from living organisms are termed as organic compounds and those obtained from the earth are known as inorganic compounds. Organic compounds are found in earth also apart from living organisms. All the organic compounds contain the element carbon. Urea was synthesized in the laboratory which is an organic compound.
Organic compounds contain heteroatom also. Some of them are nitrogen, sulfur, oxygen etc. Nitrogen containing organic compounds are of two important types and they are
Amines are organic compounds that contain a nitrogen atom that is bonded to a hydrocarbon chain.
One of the
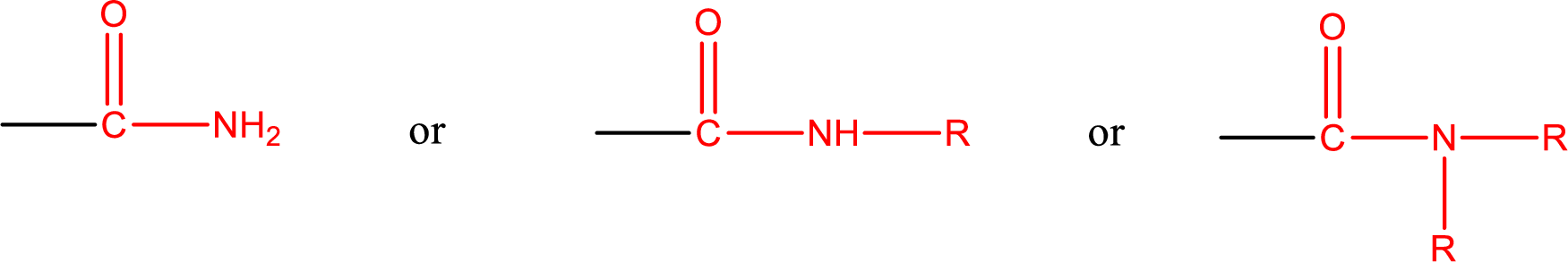
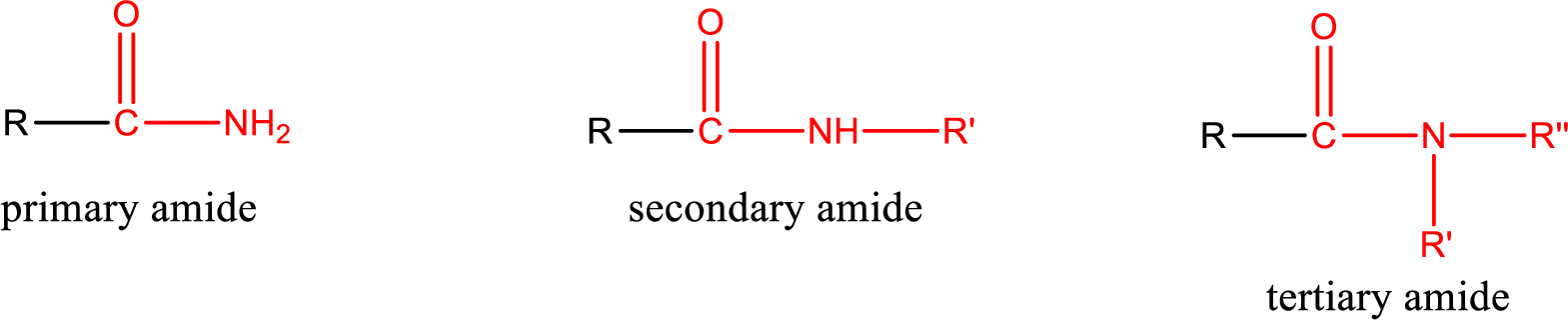
Amides are also classified as primary, secondary, and tertiary amide.
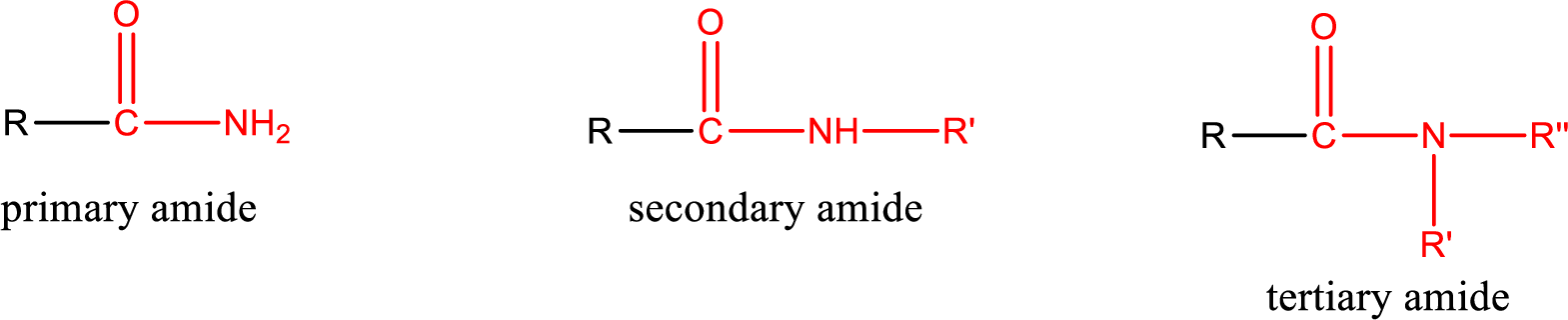
Primary amide is the one that has a nitrogen atom that is bonded to two hydrogen atoms. Primary amides are also known as unsubstituted amides.
Secondary amide is the one that has a nitrogen atom that is bonded to one hydrogen atom and one alkyl (or aryl) group. Secondary amides are also known as monosubstituted amides.
Tertiary amide is the one that has a nitrogen atom that is bonded to two alkyl (or aryl) groups. Tertiary amides are also known as disubstituted amides.
Apart from linear amides, there are also cyclic amides. They are formed by intramolecular condensation. Cyclic amides are also known as lactams.
(b)
Interpretation:
Given compound has to be classified as an amine, an amide, or both amine and amide.
Concept Introduction:
Organic compounds are the important basis of life. They include gasoline, coal, dyes, and clothing fibers etc. The compounds that are obtained from living organisms are termed as organic compounds and those obtained from the earth are known as inorganic compounds. Organic compounds are found in earth also apart from living organisms. All the organic compounds contain the element carbon. Urea was synthesized in the laboratory which is an organic compound.
Organic compounds contain heteroatom also. Some of them are nitrogen, sulfur, oxygen etc. Nitrogen containing organic compounds are of two important types and they are amines, amides.
Amines are organic compounds that contain a nitrogen atom that is bonded to a hydrocarbon chain.
One of the carboxylic acid derivatives is amide. In this the carboxyl
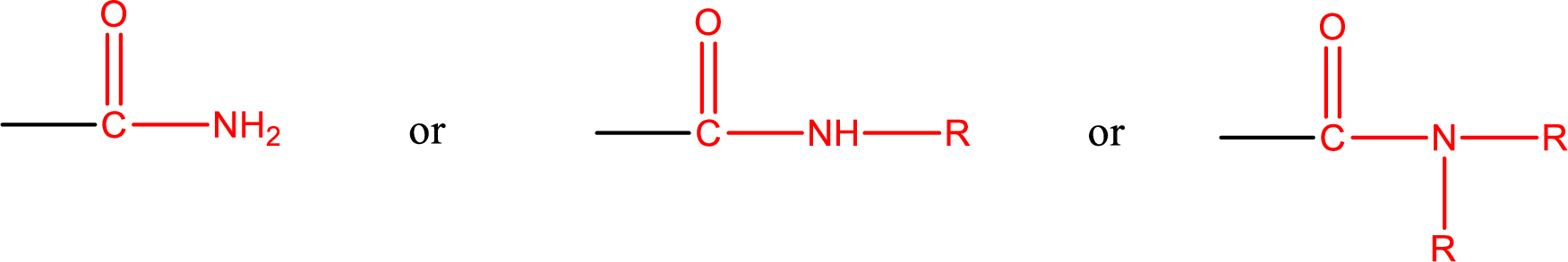
Amides are also classified as primary, secondary, and tertiary amide.
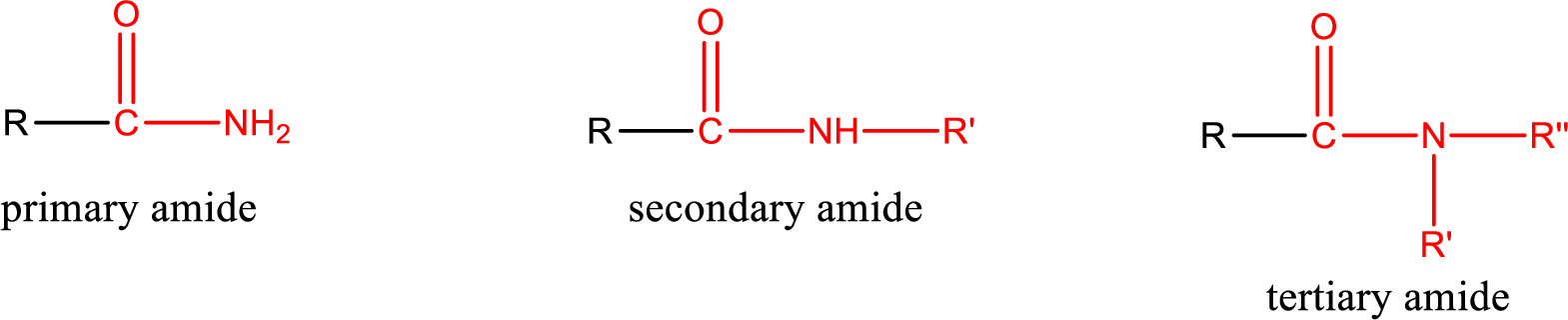
Primary amide is the one that has a nitrogen atom that is bonded to two hydrogen atoms. Primary amides are also known as unsubstituted amides.
Secondary amide is the one that has a nitrogen atom that is bonded to one hydrogen atom and one alkyl (or aryl) group. Secondary amides are also known as monosubstituted amides.
Tertiary amide is the one that has a nitrogen atom that is bonded to two alkyl (or aryl) groups. Tertiary amides are also known as disubstituted amides.
Apart from linear amides, there are also cyclic amides. They are formed by intramolecular condensation. Cyclic amides are also known as lactams.
(c)
Interpretation:
Given compound has to be classified as an amine, an amide, or both amine and amide.
Concept Introduction:
Organic compounds are the important basis of life. They include gasoline, coal, dyes, and clothing fibers etc. The compounds that are obtained from living organisms are termed as organic compounds and those obtained from the earth are known as inorganic compounds. Organic compounds are found in earth also apart from living organisms. All the organic compounds contain the element carbon. Urea was synthesized in the laboratory which is an organic compound.
Organic compounds contain heteroatom also. Some of them are nitrogen, sulfur, oxygen etc. Nitrogen containing organic compounds are of two important types and they are amines, amides.
Amines are organic compounds that contain a nitrogen atom that is bonded to a hydrocarbon chain.
One of the carboxylic acid derivatives is amide. In this the carboxyl
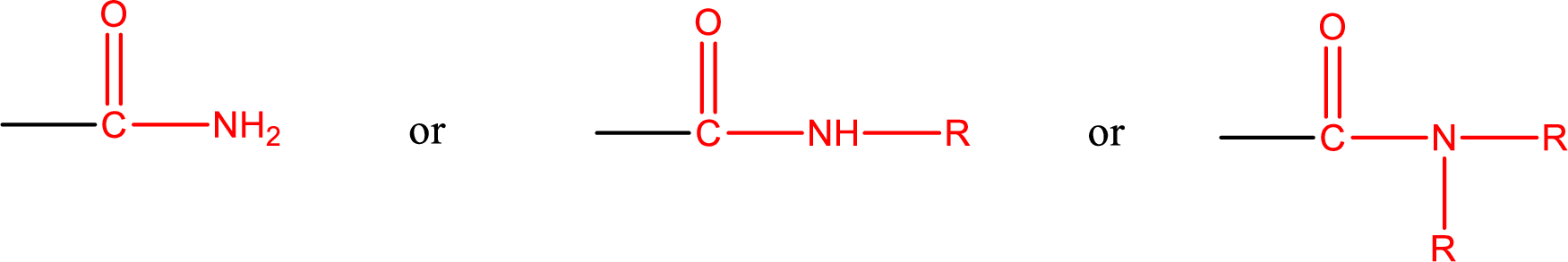
Amides are also classified as primary, secondary, and tertiary amide.
Primary amide is the one that has a nitrogen atom that is bonded to two hydrogen atoms. Primary amides are also known as unsubstituted amides.
Secondary amide is the one that has a nitrogen atom that is bonded to one hydrogen atom and one alkyl (or aryl) group. Secondary amides are also known as monosubstituted amides.
Tertiary amide is the one that has a nitrogen atom that is bonded to two alkyl (or aryl) groups. Tertiary amides are also known as disubstituted amides.
Apart from linear amides, there are also cyclic amides. They are formed by intramolecular condensation. Cyclic amides are also known as lactams.
(d)
Interpretation:
Given compound has to be classified as an amine, an amide, or both amine and amide.
Concept Introduction:
Organic compounds are the important basis of life. They include gasoline, coal, dyes, and clothing fibers etc. The compounds that are obtained from living organisms are termed as organic compounds and those obtained from the earth are known as inorganic compounds. Organic compounds are found in earth also apart from living organisms. All the organic compounds contain the element carbon. Urea was synthesized in the laboratory which is an organic compound.
Organic compounds contain heteroatom also. Some of them are nitrogen, sulfur, oxygen etc. Nitrogen containing organic compounds are of two important types and they are amines, amides.
Amines are organic compounds that contain a nitrogen atom that is bonded to a hydrocarbon chain.
One of the carboxylic acid derivatives is amide. In this the carboxyl
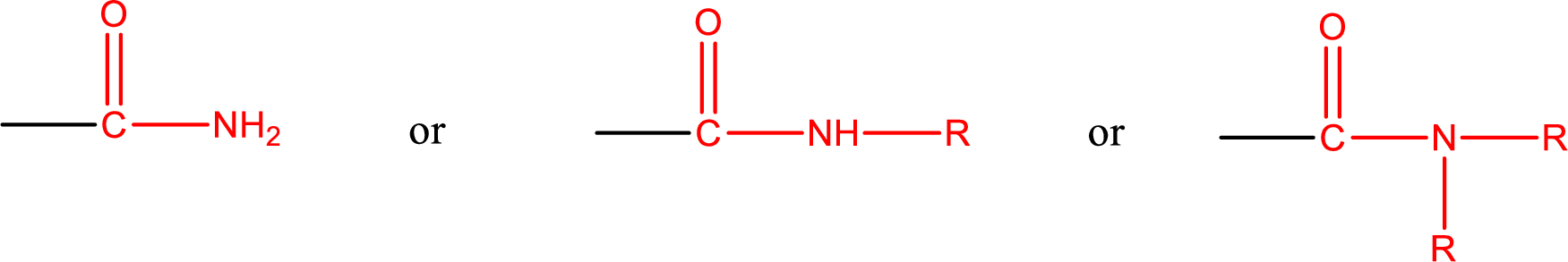
Amides are also classified as primary, secondary, and tertiary amide.
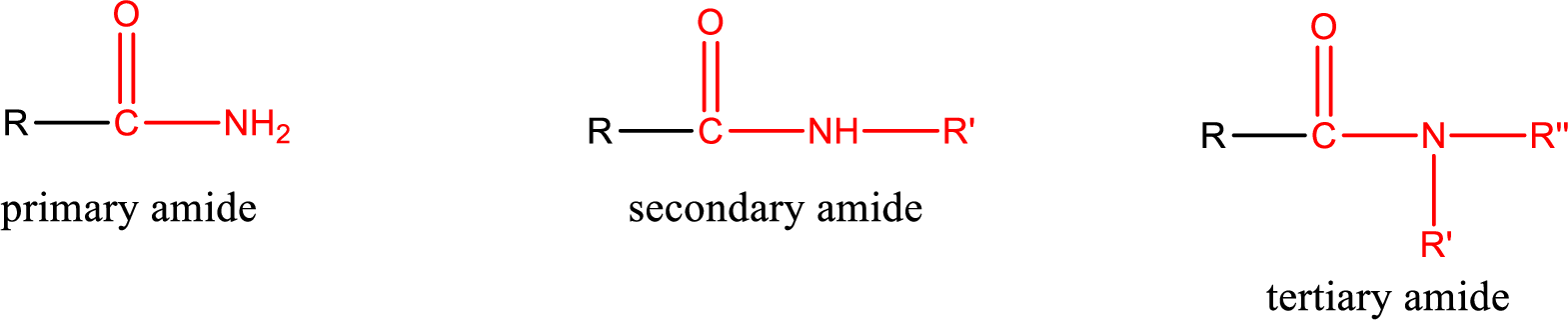
Primary amide is the one that has a nitrogen atom that is bonded to two hydrogen atoms. Primary amides are also known as unsubstituted amides.
Secondary amide is the one that has a nitrogen atom that is bonded to one hydrogen atom and one alkyl (or aryl) group. Secondary amides are also known as monosubstituted amides.
Tertiary amide is the one that has a nitrogen atom that is bonded to two alkyl (or aryl) groups. Tertiary amides are also known as disubstituted amides.
Apart from linear amides, there are also cyclic amides. They are formed by intramolecular condensation. Cyclic amides are also known as lactams.
Trending nowThis is a popular solution!

Chapter 17 Solutions
General, Organic, and Biological Chemistry
- Indicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forwardSynthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIf possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forward
- Synthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forward
- Indicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,




