
(a)
Interpretation:
The compound that has higher solubility in water among the given pair has to be identified.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
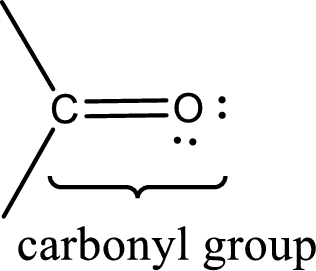
Aldehydes contain a carbonyl group that contains a hydrogen atom and a carbon atom bonded to it. Aldehyde that has one and two carbon atoms are gas at room temperature. The physical state of aldehyde that contains three carbon atoms to eleven carbon atoms that are not branched is liquid at room temperature. Aldehydes that contain more than eleven carbon atoms are solid at room temperature.
Ketones contain a carbonyl group that contains two carbon atoms bonded to it. For a compound to be ketone, a minimum of three carbon atom is required. Ketones that contain three carbon atoms to eight carbon atoms which have the carbonyl group at the second carbon atom are liquid at room temperature.
Solubility of aldehydes and ketones depend upon the length of the carbon chain. Those contain less than six carbon atoms are soluble in both water and organic solvents. Aldehydes and ketones that contain six or more carbon atoms are not soluble in water but soluble in organic solvents only. Solubility of smaller ketones and aldehydes are result of the hydrogen bond formation between the lone pairs of oxygen atom and the hydrogen atom in the water molecule.
(b)
Interpretation:
The compound that has higher solubility in water among the given pair has to be identified.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
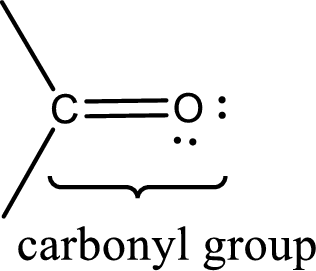
Aldehydes contain a carbonyl group that contains a hydrogen atom and a carbon atom bonded to it. Aldehyde that has one and two carbon atoms are gas at room temperature. The physical state of aldehyde that contains three carbon atoms to eleven carbon atoms that are not branched is liquid at room temperature. Aldehydes that contain more than eleven carbon atoms are solid at room temperature.
Ketones contain a carbonyl group that contains two carbon atoms bonded to it. For a compound to be ketone, a minimum of three carbon atom is required. Ketones that contain three carbon atoms to eight carbon atoms which have the carbonyl group at the second carbon atom are liquid at room temperature.
Solubility of aldehydes and ketones depend upon the length of the carbon chain. Those contain less than six carbon atoms are soluble in both water and organic solvents. Aldehydes and ketones that contain six or more carbon atoms are not soluble in water but soluble in organic solvents only. Solubility of smaller ketones and aldehydes are result of the hydrogen bond formation between the lone pairs of oxygen atom and the hydrogen atom in the water molecule.
Trending nowThis is a popular solution!

Chapter 15 Solutions
GENERAL,ORGANIC,+BIO.CHEM.-MINDTAP
- How does the square root mean square velocity of gas molecules vary with temperature? Illustrate this relationship by plotting the square root mean square velocity of N2 molecules as a function of temperature from T=100 K to T=300 K.arrow_forwardDraw product B, indicating what type of reaction occurs. F3C CF3 NH2 Me O .N. + B OMearrow_forwardBenzimidazole E. State its formula. sState the differences in the formula with other benzimidazoles.arrow_forward
- Draw product A, indicating what type of reaction occurs. F3C CN CF3 K2CO3, DMSO, H₂O2 Aarrow_forward19) Which metal is most commonly used in galvanization to protect steel structures from oxidation? Lead a. b. Tin C. Nickel d. Zinc 20) The following molecule is an example of a: R₁ R2- -N-R3 a. Secondary amine b. Secondary amide c. Tertiary amine d. Tertiary amidearrow_forwardpls helparrow_forward
- pls helparrow_forward35) Complete the following equation by drawing the line the structure of the products that are formed. Please note that in some cases more than one product is possible. You must draw all possible products to recive full marks! a. ethanol + 2-propanol + H2SO4 → b. OH conc. H2SO4 CH2 H3C CH + K2Cr2O7 C. d. H3C A pressure CH3 + H2 CH Pt catalystarrow_forward21) The rate of reaction depends upon: a. the concentration and nature of reactants b. the temperature of the reaction C. whether or not a catalyst was used d. all of the above 22) A Maxwell-Boltzmann curve shows the distribution of molecular energies in a reaction system. When the temperature in this system is increased, the peak is a. higher and further to the right. b. higher and further to the left. c. lower and further to the right. d. lower and further to the left. 23) Which of the following correctly describes the reaction represented by the reaction below? CaCO3 (s) + energy → CaO (s) + CO2 (g) a. It is exothermic and the potential energy is greater in the reactants than the products. b. c. It is exothermic and the potential energy is greater in the products than the reactants. It is endothermic and the potential energy is greater in the products than the reactants. d. It is endothermic and the potential energy is equal for the products and reactants.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





