
(a)
Interpretation:
A complete orbital diagram of Helium with
Concept introduction:
Electronic configuration can be assigned to any elements in the ground when they follow certain rules like the Hund rule, Pauli Exclusion Principle and Aufbau rule. If the
No two electrons in an atom can have the same group of four quantum numbers and this is Pauli Exclusion Principle.
While filling of orbital’s, the electron first enters to each energy level with degenerate energy before the paring of electron begins and this is Hund’s rules.
(a)
Answer to Problem 39A
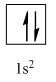
Complete orbital diagram of Helium with
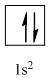
Explanation of Solution
As per the Aufbau rule, electrons are filled in lower energy orbitals that are closer to the nucleus before they are filled in higher energy ones. The order of orbital arranged in their increasing energies is as follows:
So, the complete electronic configuration of Helium with
Therefore, the complete orbital diagram of Helium with
(b)
Interpretation:
A complete orbital diagram of Neon with
Concept introduction:
Electronic configuration can be assigned to any elements in the ground when they follow certain rules like the Hund rule, Pauli Exclusion Principle and Aufbau rule. If the atomic number of an element is
No two electrons in an atom can have the same group of four quantum numbers and this is Pauli Exclusion Principle.
While filling of orbital’s, the electron first enters to each energy level with degenerate energy before the paring of electron begins and this is Hund’s rules.
(b)
Answer to Problem 39A
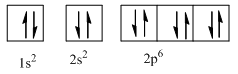
Complete orbital diagram of Neon with
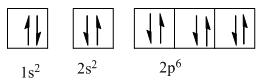
Explanation of Solution
As per Aufbau rule electrons are filled in lower energy orbitals that are closer to the nucleus before they are filled in higher energy ones. The order of orbital arranged in their increasing energies is as follows:
So complete electronic configuration of Neon with
Therefore, the complete orbital diagram of Neon with
(c)
Interpretation:
A complete orbital diagram of krypton with
Concept introduction:
Electronic configuration can be assigned to any elements in the ground when they follow certain rules like the Hund rule, Pauli Exclusion Principle and Aufbau rule. If the atomic number of an element is
No two electrons in an atom can have the same group of four quantum numbers and this is Pauli Exclusion Principle.
While filling of orbital’s, the electron first enters to each energy level with degenerate energy before the paring of electron begins and this is Hund’s rules.
(c)
Answer to Problem 39A
Complete orbital diagram of krypton with

Explanation of Solution
As per the Aufbau rule, electrons are filled in lower energy orbitals that are closer to the nucleus before they are filled in higher energy ones. The order of orbital arranged in their increasing energies is as follows:
So, the complete electronic configuration of krypton with
Therefore, the complete orbital diagram of krypton with

(a)
Interpretation:
A complete orbital diagram of Xenon with
Concept introduction:
Electronic configuration can be assigned to any elements in the ground when they follow certain rules like the Hund rule, Pauli Exclusion Principle and Aufbau rule. If the atomic number of an element is
No two electrons in an atom can have the same group of four quantum numbers and this is Pauli Exclusion Principle.
While filling of orbital’s, the electron first enters to each energy level with degenerate energy before the paring of electron begins and this is Hund’s rules.
(a)
Answer to Problem 39A
Complete orbital diagram of Xenon with
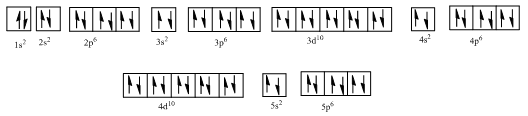
Explanation of Solution
As per the Aufbau rule, electrons are filled in lower energy orbitals that are closer to the nucleus before they are filled in higher energy ones. The order of orbital arranged in their increasing energies is as follows:
So, the complete electronic configuration of Xenon with
Therefore, the complete orbital diagram of Xenon with
Chapter 11 Solutions
World of Chemistry
- These are in the wrong boxes. Why does the one on the left have a lower molar mass than the one on the right?arrow_forwardSYNTHESIS REACTIONS. For the following reactions, synthesize the given products from the given reactants. Multiple reactions/steps will be needed. For the one of the steps (ie reactions) in each synthesis, write out the mechanism for that reaction and draw an energy diagram showing the correct number of hills and valleys for that step's mechanism. CI b. a. Use acetylene (ethyne) and any alkyl halide as your starting materials Br C. d. "OH OH III. OHarrow_forwardCalculate the pH and the pOH of each of the following solutions at 25 °C for which the substances ionize completely: (a) 0.200 M HClarrow_forward
- Calculate the pH and the pOH of each of the following solutions at 25 °C for which the substances ionize completely: (a) 0.000259 M HClO4arrow_forwardWhat is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forwardDetermine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. NaN₃arrow_forward
- A. Draw the structure of each of the following alcohols. Then draw and name the product you would expect to produce by the oxidation of each. a. 4-Methyl-2-heptanol b. 3,4-Dimethyl-1-pentanol c. 4-Ethyl-2-heptanol d. 5,7-Dichloro-3-heptanolarrow_forwardWhat is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forwardCan I please get help with this.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





