
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
3rd Edition
ISBN: 9780137533268
Author: Paula Bruice
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 52P
Propose a mechanism for the following reaction (show all curved arrows):
- a. Which step is the rate-determining step?
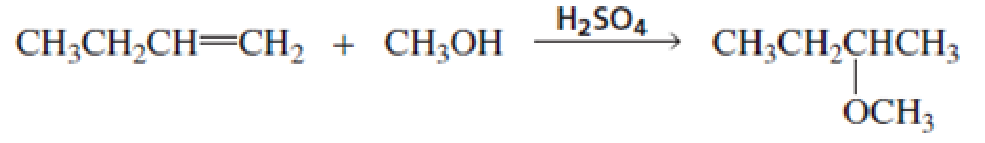
- b. What is the electrophile in the first step?
- c. What is the nucleophile in the first step?
- d. What is the electrophile in the second step?
- e. What is the nucleophile in the second step?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used hand raiting
Diferencie entre semiconductores extrínsecos de tipo n y semiconductores extrínsecos de tipo p.
None
Chapter 6 Solutions
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
Ch. 6.1 - Draw the mechanism for the reaction of cyclohexene...Ch. 6.2 - a. How many bond orbitals are available for...Ch. 6.2 - Prob. 3PCh. 6.2 - Prob. 4PCh. 6.3 - Prob. 5PCh. 6.3 - Prob. 6PCh. 6.3 - Prob. 7PCh. 6.5 - Prob. 9PCh. 6.5 - Prob. 10PCh. 6.5 - a. What is the major product of each of the...
Ch. 6.5 - Prob. 12PCh. 6.6 - What stereoisomers are obtained from each of the...Ch. 6.6 - Prob. 14PCh. 6.8 - Prob. 15PCh. 6.10 - Name the following:Ch. 6.10 - Draw the structure for each of the following: a....Ch. 6.10 - Draw the structures for and name the seven alkynes...Ch. 6.10 - Name the following:Ch. 6.10 - Name the following:Ch. 6.11 - What hybrid orbitals are used to form the...Ch. 6.13 - Prob. 22PCh. 6.14 - Prob. 23PCh. 6.14 - Which alkyne would be the best one to use for the...Ch. 6.14 - Prob. 25PCh. 6.14 - Prob. 26PCh. 6.15 - Describe the alkyne you would start with and the...Ch. 6.15 - What are products of the following reactions?Ch. 6 - Prob. 29PCh. 6 - Prob. 30PCh. 6 - Prob. 31PCh. 6 - Prob. 32PCh. 6 - What is each compounds systematic name?Ch. 6 - Prob. 34PCh. 6 - Prob. 35PCh. 6 - What reagents could be used to carry out the...Ch. 6 - Prob. 37PCh. 6 - Prob. 38PCh. 6 - Prob. 39PCh. 6 - Prob. 40PCh. 6 - Prob. 41PCh. 6 - Prob. 42PCh. 6 - Answer Problem 42 using 2-butyne as the starting...Ch. 6 - What is each compounds systematic name?Ch. 6 - Prob. 45PCh. 6 - Prob. 46PCh. 6 - Prob. 47PCh. 6 - Prob. 48PCh. 6 - Prob. 49PCh. 6 - Prob. 50PCh. 6 - Draw the keto tautomer for each of the following:Ch. 6 - Propose a mechanism for the following reaction...Ch. 6 - Prob. 53PCh. 6 - Prob. 54PCh. 6 - Prob. 55PCh. 6 - Propose a mechanism for the following reaction:Ch. 6 - Prob. 57PCh. 6 - Prob. 58PCh. 6 - Prob. 59PCh. 6 - Prob. 60PCh. 6 - Prob. 61PCh. 6 - Prob. 62P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Shown below is the major resonance structure for a molecule. Draw the second best resonance structure of the molecule. Include all non-zero formal charges. H. H. +N=C H H H Cl: Click and drag to start drawing a structure. : ? g B S olo Ar B Karrow_forwardDon't used hand raitingarrow_forwardS Shown below is the major resonance structure for a molecule. Draw the second best resonance structure of the molecule. Include all non-zero formal charges. H H = HIN: H C. :0 H /\ H H Click and drag to start drawing a structure. ×arrow_forward
- Part II. two unbranched ketone have molecular formulla (C8H100). El-ms showed that both of them have a molecular ion peak at m/2 =128. However ketone (A) has a fragment peak at m/2 = 99 and 72 while ketone (B) snowed a fragment peak at m/2 = 113 and 58. 9) Propose the most plausible structures for both ketones b) Explain how you arrived at your conclusion by drawing the Structures of the distinguishing fragments for each ketone, including their fragmentation mechanisms.arrow_forwardPart V. Draw the structure of compound tecla using the IR spectrum Cobtained from the compound in KBr pellet) and the mass spectrum as shown below. The mass spectrum of compound Tesla showed strong mt peak at 71. TRANSMITTANCE LOD Relative Intensity 100 MS-NW-1539 40 20 80 T 44 55 10 15 20 25 30 35 40 45 50 55 60 65 70 75 m/z D 4000 3000 2000 1500 1000 500 HAVENUMBERI-11arrow_forwardTechnetium is the first element in the periodic chart that does not have any stable isotopes. Technetium-99m is an especially interesting and valuable isotope as it emits a gamma ray with a half life ideally suited for medical tests. It would seem that the decay of technetium should fit the treatment above with the result In(c/c) = -kt. The table below includes data from the two sites: http://dailymed.nlm.nih.gov/dailymed/druginfo.cfm?id=7130 http://wiki.medpedia.com/Clinical: Neutrospec_(Technetium_(99m Tc)_fanolesomab). a. b. C. Graph the fraction (c/c.) on the vertical axis versus the time on the horizontal axis. Also graph In(c/c.) on the vertical axis versus time on the horizontal axis. When half of the original amount of starting material has hours fraction remaining disappeared, c/c = ½ and the equation In(c/c.) = -kt becomes In(0.5) = -kt1/2 where t₁₂ is the half life (the time for half of the material to decay away). Determine the slope of your In(c/c.) vs t graph and…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License