
Concept explainers
(a)
Interpretation:
The keto-enol tautomer from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into
The formed enol and ketone are called keto-enol tautomer.
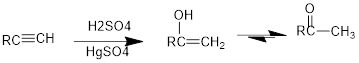
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
(b)
Interpretation:
The keto-enol tautomers from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into ketone. For terminal alkynes, there is a need of catalyst which is mercury.
The formed enol and ketone are called keto-enol tautomers.
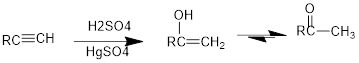
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
(c)
Interpretation:
The keto-enol tautomers from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into ketone. For terminal alkynes, there is a need of catalyst which is mercury.
The formed enol and ketone are called keto-enol tautomers.
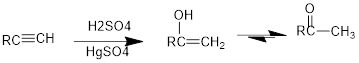
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
(d)
Interpretation:
The keto-enol tautomers from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into ketone. For terminal alkynes, there is a need of catalyst which is mercury.
The formed enol and ketone are called keto-enol tautomers.
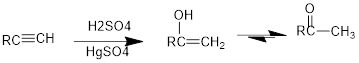
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
(e)
Interpretation:
The keto-enol tautomers from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into ketone. For terminal alkynes, there is a need of catalyst which is mercury.
The formed enol and ketone are called keto-enol tautomers.
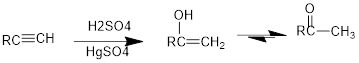
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
