
Concept explainers
(a)
Interpretation:
IUPAC name for the given compound has to be assigned.
Concept Introduction:
For naming an acid anhydride, it can be structurally viewed in a way that contains two carbonyl groups that is joined by a single oxygen atom. This can also be said as two acyl group joined by a single oxygen atom.
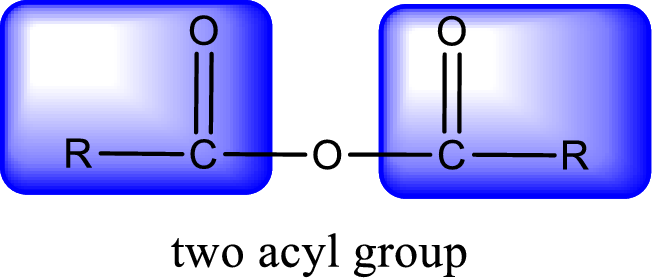
Rules to obtain IUPAC name and common name for an acid anhydride:
- IUPAC name and common name for Symmetric anhydride is obtained by replacing the acid present in the name of parent
carboxylic acid with the word anhydride. - IUPAC name and common name for mixed anhydride is obtained by using the names of the parent carboxylic acids arranged in alphabetical order that is followed by the word anhydride.
(b)
Interpretation:
IUPAC name for the given compound has to be assigned.
Concept Introduction:
For naming an acid chloride, it can be structurally viewed in a way that contains one acyl group with a chlorine atom bonded to the carbonyl group
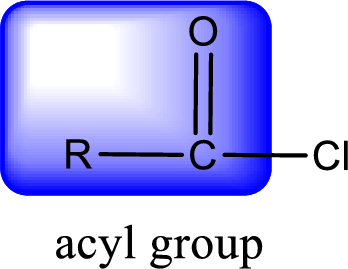
Rules to obtain IUPAC name and common name for an acid chloride:
- IUPAC name for acid chloride can be obtained from the parent carboxylic acid name. In the parent carboxylic acid name, the ending “-oic acid” is replaced by “-oyl chloride”.
- Common name for acid chloride can be obtained from the parent carboxylic acid name. In the parent carboxylic acid name, the ending “-ic acid” is replaced by “-yl chloride”.
(c)
Interpretation:
IUPAC name for the given compound has to be assigned.
Concept Introduction:
For naming an acid chloride, it can be structurally viewed in a way that contains one acyl group with a chlorine atom bonded to the carbonyl group
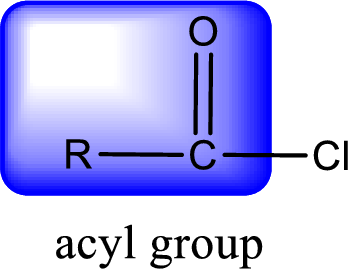
Rules to obtain IUPAC name and common name for an acid chloride:
- IUPAC name for acid chloride can be obtained from the parent carboxylic acid name. In the parent carboxylic acid name, the ending “-oic acid” is replaced by “-oyl chloride”.
- Common name for acid chloride can be obtained from the parent carboxylic acid name. In the parent carboxylic acid name, the ending “-ic acid” is replaced by “-yl chloride”.
(d)
Interpretation:
IUPAC name for the given compound has to be assigned.
Concept Introduction:
For naming an acid anhydride, it can be structurally viewed in a way that contains two carbonyl groups that is joined by a single oxygen atom. This can also be said as two acyl group joined by a single oxygen atom.
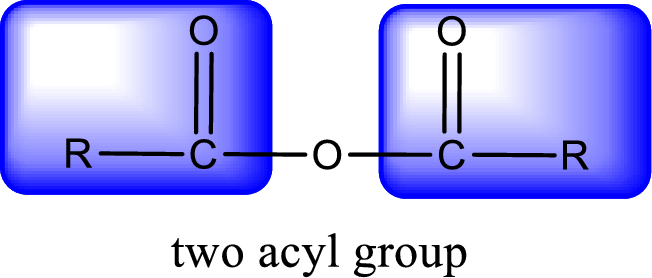
Rules to obtain IUPAC name and common name for an acid anhydride:
- IUPAC name and common name for Symmetric anhydride is obtained by replacing the acid present in the name of parent carboxylic acid with the word anhydride.
- IUPAC name and common name for mixed anhydride is obtained by using the names of the parent carboxylic acids arranged in alphabetical order that is followed by the word anhydride.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
EBK ORGANIC AND BIOLOGICAL CHEMISTRY
- What would you expect to be the major product obtained from the following reaction? Please explain what is happening here. Provide a detailed explanation and a drawing showing how the reaction occurs. The correct answer to this question is V.arrow_forwardPlease answer the question for the reactions, thank youarrow_forwardWhat is the product of the following reaction? Please include a detailed explanation of what is happening in this question. Include a drawing showing how the reagent is reacting with the catalyst to produce the correct product. The correct answer is IV.arrow_forward
- Please complete the reactions, thank youarrow_forwardConsider the synthesis. What is compound Y? Please explain what is happening in this question. Provide a detailed explanation and a drawing to show how the compound Y creates the product. The correct answer is D.arrow_forwardWhat would be the major product of the following reaction? Please include a detailed explanation of what is happening in this question. Include steps and a drawing to show this reaction proceeds and how the final product is formed. The correct answer is B. I put answer D and I don't really understand what is going on in the question.arrow_forward
- What is the product of the following reaction? Please explain what is happening in this question. Provide a detailed explanation and a drawing showing how the reagent is reacting with the catalysts to product the correct product. The correct answer is B.arrow_forwardWhat is the missing intermediate 1 and the final product 2. Please include a detailed explanation explaining the steps of malonic ester synthesis. Please include drawings of the intermediate and how it occurs and how the final product is former.arrow_forwardWhat would be the reagents and conditions above and below the arrow that will complete the proposed acetoacetic ester synthesis? If it cannot be done efficiently, then I will choose that answer. There could be 2 or 4 reagents involved. Please provide a detailed explanation and drawings showing how it would proceed with the correct reagents.arrow_forward
- For benzene, the ∆H° of vaporization is 30.72 kJ/mol and the ∆S° of vaporization is 86.97 J/mol・K. At 1.00 atm and 228.0 K, what is the ∆G° of vaporization for benzene, in kJ/mol?arrow_forwardThe reaction Q(g) + R(g) → Z(l) is shown to be exothermic. Which of the following is true concerning the reaction. it is spontaneous only at High T, it is spontaneous at low T it is nonspontaneous at all T it is spontanrous at all T. it is non spontaneous only at low T.arrow_forwardThe reaction Q(g) + R(g) → Z(l) is shown to be exothermic. Which of the following is true concerning the reactionarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning


