
Chemistry & Chemical Reactivity
9th Edition
ISBN: 9781133949640
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 22, Problem 63GQ
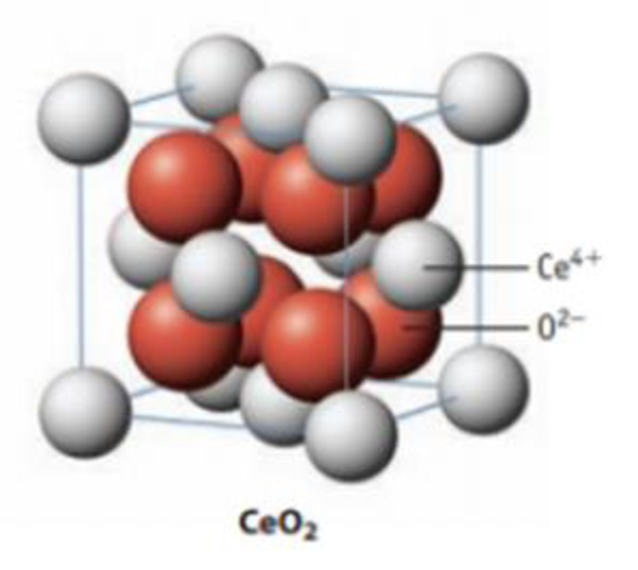
Cerium, as noted in Applying Chemical Principles: 22.3 The Rare Earths, is a relatively abundant lanthanide element that has some important uses. Cerium(IV) oxide. CeO2, is widely used as a polishing agent for glass. Cerium(III) sulfide, Ce2S3, is becoming more widely used as a red pigment to replace cadmium pigments, which are environmentally less desirable.
(a) Give the electron configurations (using the noble gas notation) for Ce, Ce3+, and Ce4+.
(b) Is either Ce3+ or Ce4+ paramagnetic? If so, how many unpaired electrons does each have?
(c) The solid state structure for CeO2 is shown below. Describe the unit cell of the compound. How is this structure related to the formula?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Find a molecular formula for these unknowns
(ME EX2) Prblms 8-11 Can you please explain problems 8 -11 to me in detail, step by step? Thank you so much! If needed color code them for me.
Don't used hand raiting
Chapter 22 Solutions
Chemistry & Chemical Reactivity
Ch. 22.1 - Prob. 1RCCh. 22.2 - Prob. 1QCh. 22.2 - 2. Describe the unit cell of austenite.
Ch. 22.2 - Prob. 3QCh. 22.2 - Prob. 1RCCh. 22.2 - Prob. 2RCCh. 22.3 - (a) What is the formula of a complex ion composed...Ch. 22.3 - (a) Determine the metals oxidation number and...Ch. 22.3 - Name the following coordination compounds. (a)...Ch. 22.3 - Prob. 1RC
Ch. 22.3 - 2. What is the oxidation number of the metal in...Ch. 22.3 - Prob. 3RCCh. 22.3 - Prob. 4RCCh. 22.4 - What types of isomers are possible for the...Ch. 22.4 - Prob. 1RCCh. 22.4 - Prob. 2RCCh. 22.4 - Prob. 3RCCh. 22.4 - Prob. 4RCCh. 22.4 - Prob. 1QCh. 22.5 - Prob. 1CYUCh. 22.5 - Prob. 1RCCh. 22.5 - Prob. 2RCCh. 22.6 - Prob. 1QCh. 22.6 - Prob. 2QCh. 22.6 - Prob. 3QCh. 22.6 - Prob. 4QCh. 22.6 - Prob. 1CYUCh. 22.6 - Prob. 1RCCh. 22.6 - How are the d electrons of Pt distributed in a...Ch. 22.6 - What are the electron configurations for Nd and...Ch. 22.6 - Prob. 3CSCh. 22.6 - Prob. 4CSCh. 22.6 - Prob. 5CSCh. 22 - Prob. 1PSCh. 22 - Prob. 2PSCh. 22 - Identify a cation of a first series transition...Ch. 22 - Match up the isoelectronic ions on the following...Ch. 22 - Prob. 5PSCh. 22 - Prob. 6PSCh. 22 - Which of the following ligands is expected to be...Ch. 22 - One of the following nitrogen compounds or ions is...Ch. 22 - Prob. 9PSCh. 22 - Prob. 10PSCh. 22 - Prob. 11PSCh. 22 - Prob. 12PSCh. 22 - Prob. 13PSCh. 22 - Prob. 14PSCh. 22 - Prob. 15PSCh. 22 - Prob. 16PSCh. 22 - Give the name or formula for each ion or compound,...Ch. 22 - Prob. 18PSCh. 22 - Prob. 19PSCh. 22 - Prob. 20PSCh. 22 - Prob. 21PSCh. 22 - Prob. 22PSCh. 22 - Prob. 23PSCh. 22 - Prob. 24PSCh. 22 - Prob. 25PSCh. 22 - Prob. 26PSCh. 22 - Prob. 27PSCh. 22 - Prob. 28PSCh. 22 - Prob. 29PSCh. 22 - Prob. 30PSCh. 22 - In water, the titanium(III) ion, [Ti(H2O)6]3+, has...Ch. 22 - Prob. 32PSCh. 22 - Prob. 33GQCh. 22 - Prob. 34GQCh. 22 - How many unpaired electrons are expected for...Ch. 22 - Prob. 36GQCh. 22 - Which of the following complex ions is (are)...Ch. 22 - Prob. 38GQCh. 22 - How many geometric isomers are possible for the...Ch. 22 - For a tetrahedral complex of a metal in the first...Ch. 22 - Prob. 41GQCh. 22 - Prob. 42GQCh. 22 - Prob. 43GQCh. 22 - A platinum-containing compound, known as Magnuss...Ch. 22 - Prob. 45GQCh. 22 - Prob. 46GQCh. 22 - Prob. 47GQCh. 22 - How many geometric isomers of the complex ion...Ch. 22 - Prob. 49GQCh. 22 - Prob. 50GQCh. 22 - Prob. 51GQCh. 22 - The square-planar complex Pt(en)Cl2 has chloride...Ch. 22 - The complex [Mn(H2O)6]2+ has five unpaired...Ch. 22 - Experiments show that K4[Cr(CN)6] is paramagnetic...Ch. 22 - Give a systematic name or the formula for the...Ch. 22 - When CrCI3 dissolves in water, three different...Ch. 22 - Prob. 57GQCh. 22 - The glycinate ion, H2NCH2CO2, formed by...Ch. 22 - Prob. 59GQCh. 22 - Nickel and palladium both form complexes of the...Ch. 22 - The transition metals form a class of compounds...Ch. 22 - Cerium, as noted in Applying Chemical Principles:...Ch. 22 - Prob. 64GQCh. 22 - Two different coordination compounds containing...Ch. 22 - Prob. 71SCQCh. 22 - Prob. 69SCQCh. 22 - Prob. 70SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The following 'H NMR spectrum was taken with a 750 MHz spectrometer: 1.0 0.5 0.0 10.0 9.0 8.0 7.0 6.0 5.0 4.0 3.0 ' 2.0 1.0 0.0 (ppm) What is the difference Av in the frequency of RF ac Δν ac radiation absorbed by the a and c protons? (Note: it's not equal to the difference in chemical shifts.) Round your answer to 2 significant digits, and be sure it has an appropriate unit symbol. = O O a will shift left, c will shift right. O a will shift right, c will shift left. a and c will both shift left, with more space between them. Suppose a new spectrum is taken with a 500 MHz spectrometer. What will be true about this new spectrum? O a and c will both shift left, with less space between them. O a and c will both shift right, with more space between them. O a and c will both shift right, with less space between them. Which protons have the largest energy gap between spin up and spin down states? O None of the above. ○ a Ob Explanation Check C Ar B 2025 McGraw Hill LLC. All Rights Reserved.…arrow_forwardWhat mass of Na2CO3 must you add to 125g of water to prepare 0.200 m Na2CO3? Calculate mole fraction of Na2CO3, mass percent, and molarity of the resulting solution. MM (g/mol): Na2CO3 105.99; water 18.02. Final solution density is 1.04 g/mL.arrow_forward(ME EX2) Prblms Can you please explain problems to me in detail, step by step? Thank you so much! If needed color code them for me.arrow_forward
- Experiment #8 Electrical conductivity & Electrolytes. Conductivity of solutions FLINN Scientific Scale RED LED Green LED LED Conductivity 0 OFF OFF 1 Dim OFF 2 medium OFF 3 Bright Dim Low or Nowe Low Medium High 4 Very Bright Medium nd very high AA Δ Δ Δ Δ Δ Δ Δ Δ Δ Δ Δ SE=Strong Electrolyte, FE = Fair Electrolyte CWE = Weak Electrolyte, NE= Noni Electrolyte, #Solutions 1 0.1 M NaCl 2/1x 102 M NaCl, 3/1X103 M Nall Can Prediction M Observed Conductivity Very bright red Bright red Dim red you help me understand how I'm supposed to find the predictions of the following solutions? I know this is an Ionic compound and that the more ions in a solution means it is able to carry a charge, right? AAAA Darrow_forward(SE EX 2) Prblsm 4-7: Can you please explain problems 4-7 and color code if needed for me. (step by step) detail explanationsarrow_forward(SE EX 2) Problems 8-11, can you please explain them to me in detail and color-code anything if necessary?arrow_forward
- (ME EX2) Problems 15-16 Could you please explain problems 15 through 16 to me in detail, step by step? Thank you so much! If necessary, please color-code them for me.arrow_forward1.)show any electrophilic aromatic substitution, identify the electriphile, nucleophile and transition statearrow_forward(SE EX 2) Problems 15-16, can you please explain them to me in detail and color-code anything if necessary?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lanthanoids and its Position in Periodic Table - D and F Block Elements - Chemistry Class 12; Author: Ekeeda;https://www.youtube.com/watch?v=ZM04kRxm6tY;License: Standard YouTube License, CC-BY