
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
8th Edition
ISBN: 9780134015187
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 21, Problem 21.87GP
Interpretation Introduction
Interpretation:
The enzyme that gets inhibited by fluoroacetate should be identified.
Concept Introduction:
Enzyme: The proteins that helps
Citric Acid Cycle: It involves 8 series reaction which converts acetyl group present in acetyl coenzyme A into 2
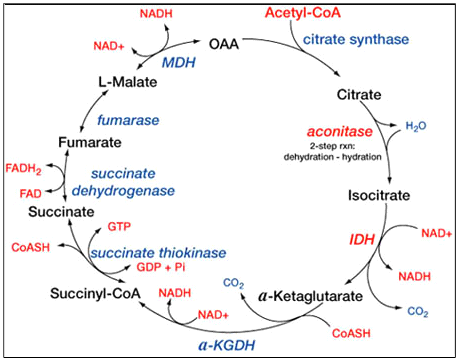
Figure 1
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please explain the process of how an axon degenerates in the central nervous system following injury and how it affects the neuron/cell body, as well as presynaptic and postsynaptic neurons. Explain processes such as chromatolysis and how neurotrophin signaling works.
Please help determine the Relative Response Ratio of my GC-MS laboratory:
Laboratory: Alcohol Content in Hand Sanditizers
Internal Standard: Butanol
Standards of Alcohols: Methanol, Ethanol, Isopropyl, n-Propanol, Butanol
Recorded Retention Times: 0.645, 0.692, 0.737, 0.853, 0.977
Formula: [ (Aanalyte / Canalyte) / (AIS / CIS) ]
Please help determine the Relative Response Ratio of my GC-MS laboratory:
Laboratory: Alcohol Content in Hand Sanditizers
Internal Standard: Butanol
Standards of Alcohols: Methanol, Ethanol, Isopropyl, n-Propanol, Butanol
Recorded Retention Times: 0.645, 0.692, 0.737, 0.853, 0.977
Formula: [ (Aanalyte / Canalyte) / (AIS / CIS) ]
Chapter 21 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Ch. 21.1 - In a cell, glucose can be oxidized via metabolic...Ch. 21.1 - Prob. 21.2KCPCh. 21.1 - Prob. 21.1CIAPCh. 21.1 - Prob. 21.2CIAPCh. 21.1 - Prob. 21.3CIAPCh. 21.1 - Prob. 21.4CIAPCh. 21.3 - Prob. 21.3PCh. 21.4 - Prob. 21.4PCh. 21.4 - Prob. 21.5PCh. 21.4 - Prob. 21.5CIAP
Ch. 21.4 - How does a cell disarm each of the ROS in CIA...Ch. 21.5 - One of the steps in lipid metabolism is the...Ch. 21.5 - Prob. 21.7PCh. 21.5 - Prob. 21.7CIAPCh. 21.5 - Prob. 21.8CIAPCh. 21.5 - Prob. 21.9CIAPCh. 21.5 - Prob. 21.10CIAPCh. 21.5 - Prob. 21.8PCh. 21.6 - Prob. 21.9PCh. 21.6 - Prob. 21.10PCh. 21.7 - Prob. 21.11PCh. 21.7 - Prob. 21.13PCh. 21.7 - Identify the participants in the citric acid cycle...Ch. 21.7 - Prob. 21.15PCh. 21.7 - Prob. 21.16KCPCh. 21.8 - Within the mitochondrion, is the pH higher in the...Ch. 21.8 - Prob. 21.18PCh. 21.8 - Prob. 21.19KCPCh. 21.8 - Prob. 21.11CIAPCh. 21.8 - Prob. 21.12CIAPCh. 21.8 - Prob. 21.13CIAPCh. 21 - Prob. 21.20UKCCh. 21 - Each of these reactions is involved in one of the...Ch. 21 - Prob. 21.22UKCCh. 21 - Prob. 21.23UKCCh. 21 - The reaction that follows is catalyzed by...Ch. 21 - Prob. 21.25UKCCh. 21 - The electron-transport chain uses several...Ch. 21 - Prob. 21.27APCh. 21 - What is the difference between an endergonic...Ch. 21 - Prob. 21.29APCh. 21 - Prob. 21.30APCh. 21 - Prob. 21.31APCh. 21 - Prob. 21.32APCh. 21 - Which of the following organisms are prokaryotes,...Ch. 21 - Label each of the following as a characteristic of...Ch. 21 - Prob. 21.35APCh. 21 - Prob. 21.36APCh. 21 - Describe in general terms the structural makeup of...Ch. 21 - Prob. 21.38APCh. 21 - Prob. 21.39APCh. 21 - Prob. 21.40APCh. 21 - Prob. 21.41APCh. 21 - Prob. 21.42APCh. 21 - Prob. 21.43APCh. 21 - Prob. 21.44APCh. 21 - What does it mean when we say that two reactions...Ch. 21 - Prob. 21.46APCh. 21 - Prob. 21.47APCh. 21 - Prob. 21.48APCh. 21 - Prob. 21.49APCh. 21 - Prob. 21.50APCh. 21 - Prob. 21.51APCh. 21 - Prob. 21.52APCh. 21 - Prob. 21.53APCh. 21 - Prob. 21.54APCh. 21 - Prob. 21.55APCh. 21 - Prob. 21.56APCh. 21 - Prob. 21.57APCh. 21 - Prob. 21.58APCh. 21 - Prob. 21.59APCh. 21 - Prob. 21.60APCh. 21 - Prob. 21.61APCh. 21 - Prob. 21.62APCh. 21 - Prob. 21.63APCh. 21 - Prob. 21.64APCh. 21 - Prob. 21.65APCh. 21 - Prob. 21.66APCh. 21 - Prob. 21.67APCh. 21 - Prob. 21.68APCh. 21 - What does the term oxidative phosphorylation mean?...Ch. 21 - Prob. 21.70APCh. 21 - Prob. 21.71APCh. 21 - Prob. 21.72APCh. 21 - Prob. 21.73APCh. 21 - Prob. 21.74APCh. 21 - Prob. 21.75APCh. 21 - Prob. 21.76APCh. 21 - Prob. 21.77CPCh. 21 - Fumarate produced in step 6 of the citric acid...Ch. 21 - With what class of enzymes are the coenzymes NAD+...Ch. 21 - Prob. 21.80CPCh. 21 - Prob. 21.81CPCh. 21 - Prob. 21.82CPCh. 21 - The mitochondrion pumps H+ from the matrix into...Ch. 21 - Prob. 21.84CPCh. 21 - Prob. 21.85CPCh. 21 - Prob. 21.86CPCh. 21 - Prob. 21.87GPCh. 21 - Prob. 21.88GPCh. 21 - Prob. 21.89GP
Knowledge Booster
Similar questions
- please draw it for me and tell me where i need to modify the structurearrow_forwardPlease help determine the standard curve for my Kinase Activity in Excel Spreadsheet. Link: https://mnscu-my.sharepoint.com/personal/vi2163ss_go_minnstate_edu/_layouts/15/Doc.aspx?sourcedoc=%7B958f5aee-aabd-45d7-9f7e-380002892ee0%7D&action=default&slrid=9b178ea1-b025-8000-6e3f-1cbfb0aaef90&originalPath=aHR0cHM6Ly9tbnNjdS1teS5zaGFyZXBvaW50LmNvbS86eDovZy9wZXJzb25hbC92aTIxNjNzc19nb19taW5uc3RhdGVfZWR1L0VlNWFqNVc5cXRkRm4zNDRBQUtKTHVBQldtcEtWSUdNVmtJMkoxQzl3dmtPVlE_cnRpbWU9eEE2X291ZHIzVWc&CID=e2126631-9922-4cc5-b5d3-54c7007a756f&_SRM=0:G:93 Determine the amount of VRK1 is present 1. Average the data and calculate the mean absorbance for each concentration/dilution (Please over look for Corrections) 2. Blank Correction à Subtract 0 ug/mL blank absorbance from all readings (Please over look for Corrections) 3. Plot the Standard Curve (Please over look for Corrections) 4. Convert VRK1 concentration from ug/mL to g/L 5. Use the molar mass of VRK1 to convert to M and uM…arrow_forwardMacmillan Learning Cholesterol synthesis begins with the formation of mevalonate from acetyl CoA. This process activates mevalonate and converts it to isopentenyl pyrophosphate. Identify the atoms in mevalonate and isopentenyl pyrophosphate that will be labeled from acetyl CoA labeled with 14C in the carbonyl carbon. Place 14C atoms and C atoms to denote which carbon atoms are labeled and which are not labeled. H₂C COA 14C-labeled acetyl-CoA HHH [c] H H OH 014C - OH H HH H Mevalonate CH3 H H 14C H Η H H Incorrect Answer of o -P-O-P-0- Isopentenyl pyrophosphate с Answer Bank 14Carrow_forward
- Draw the reaction between sphingosine and arachidonic acid. Draw out the full structures.arrow_forwardDraw both cis and trans oleic acid. Explain why cis-oleic acid has a melting point of 13.4°C and trans-oleic acid has a melting point of 44.5°C.arrow_forwardDraw the full structure of the mixed triacylglycerol formed by the reaction of glycerol and the fatty acids arachidic, lauric and trans-palmitoleic. Draw the line structure.arrow_forward
- Draw out the structure for lycopene and label each isoprene unit. "Where is lycopene found in nature and what health benefits does it provide?arrow_forwardWhat does it mean to be an essential fatty acid? What are the essential fatty acids?arrow_forwardCompare and contrast primary and secondary active transport mechanisms in terms of energy utilisation and efficiency. Provide examples of each and discuss their physiological significance in maintaining ionic balance and nutrient uptake. Rubric Understanding the key concepts (clearly and accurately explains primary and secondary active transport mechanisms, showing a deep understanding of their roles) Energy utilisation analysis ( thoroughly compares energy utilisation in primary and secondary transport with specific and relevant examples Efficiency discussion Use of examples (provides relevant and accurate examples (e.g sodium potassium pump, SGLT1) with clear links to physiological significance. Clarity and structure (presents ideas logically and cohesively with clear organisation and smooth transition between sections)arrow_forward
- 9. Which one of the compounds below is the major organic product obtained from the following reaction sequence, starting with ethyl acetoacetate? 요요. 1. NaOCH2CH3 CH3CH2OH 1. NaOH, H₂O 2. H3O+ 3. A OCH2CH3 2. ethyl acetoacetate ii A 3. H3O+ OH B C D Earrow_forward7. Only one of the following ketones cannot be made via an acetoacetic ester synthesis. Which one is it? Ph کہ A B C D Earrow_forward2. Which one is the major organic product obtained from the following reaction sequence? HO A OH 1. NaOEt, EtOH 1. LiAlH4 EtO OEt 2. H3O+ 2. H3O+ OH B OH OH C -OH HO -OH OH D E .CO₂Etarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning


Principles Of Radiographic Imaging: An Art And A ...
Health & Nutrition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Cengage Learning

Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

