
Concept explainers
(a)
Interpretation:
The given radical is to be classified as
Concept introduction:
A free radical is an atom or ion with unpaired electron. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.1P
The given free radical is classified as
Explanation of Solution
The given species is,
![]()
Figure 1
The given free radical is attached to two carbon atoms. Thus, it is classified as
The given free radical is classified as
(b)
Interpretation:
The given radical is to be classified as
Concept introduction:
A free radical is an atom or ion with unpaired electron. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.1P
The given free radical is classified as
Explanation of Solution
The given species is,
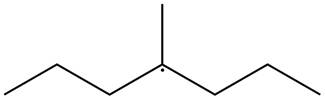
Figure 2
The given free radical is attached to three carbon atoms. Thus, it is classified as
The given free radical is classified as
(c)
Interpretation:
The given radical is to be classified as
Concept introduction:
A free radical is an atom or ion with unpaired electrons. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.1P
The given free radical is classified as
Explanation of Solution
The given species is,
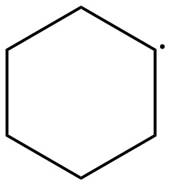
Figure 3
The given free radical is attached to two carbon atoms. Thus, it is classified as
The given free radical is classified as
(d)
Interpretation:
The given radical is to be classified as
Concept introduction:
A free radical is an atom or ion with unpaired electrons. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.1P
The given free radical is classified as
Explanation of Solution
The given species is,
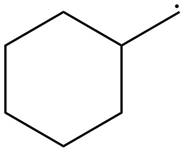
Figure 4
The given free radical is attached to one carbon atom. Thus, it is classified as
The given free radical is classified as
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry-Package(Custom)
- Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. N₂H₅ClO₄arrow_forwardPlease help me with identifying these.arrow_forwardCan I please get help with this?arrow_forward
- Can I please get help with identifying these?arrow_forward4. Calculate the pH of a 0.10 M acetic acid (CH3COOH) solution if the Ka of acetic acid = 1.8 x 10-5arrow_forwardDraw the Zaitsev product of the dehydration of this alcohol. + I X 5 OH ざ~ TSOH Click and drag to start drawing a structure.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





