
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN: 9781305580343
Author: Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 13, Problem 13.28QP
Kinetics II
You and a friend are working together in order to obtain as much kinetic information as possible about the reaction
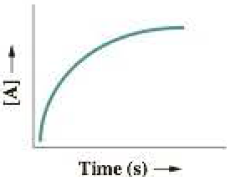
- a After studying the curve of the graph, she declares that the reaction is second order, with a corresponding rate law of Rate = k[A]2. Judging solely on the basis of the information presented in this plot, is she correct in her statement that the reaction must be second order?
Here are some data collected from her experiment:
| Time (s) | [A] |
| 0.0 | 1.0 |
| 1.0 | 0.14 |
| 3.0 | 2.5 × 10−3 |
| 5.0 | 4.5 × 10−5 |
| 7.0 | 8.3 × 10−7 |
- b The half-life of the reaction is 0.35 s. Do these data support the reaction being second order, or is it something else? Try to reach a conclusive answer without graphing the data.
- c What is the rate constant for the reaction?
- d The mechanism for this reaction is found to be a two-step process, with intermediates X and Y. The first step of the reaction is the rate-determining step. Write a possible mechanism for the reaction.
- e You perform additional experiments and find that the rate constant doubles in value when you increase the temperature by 10oC. Your lab partner doesn’t understand why the rate constant changes in this manner. What could you say to your partner to help her understand? Feel free to use figures and pictures as part of your explanation.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Organic bases have lone pairs of electrons that are capable of accepting protons. Lone pair electrons in a neutral or negatively charged species, or pi electron pairs. Explain the latter case (pi electron pairs).
Describe the propyl anion.
Indicate the names of these compounds (if they exist).
0:
HỌC—NH
CH3CH2-CH2
Chapter 13 Solutions
General Chemistry - Standalone book (MindTap Course List)
Ch. 13.1 - For the reaction given in Example 13.1, how is the...Ch. 13.1 - Iodide ion is oxidized by hypochlorite ion in...Ch. 13.1 - Shown here is a plot of the concentration of a...Ch. 13.3 - Prob. 13.3ECh. 13.3 - Prob. 13.2CCCh. 13.3 - Prob. 13.4ECh. 13.3 - Prob. 13.3CCCh. 13.4 - a. What would be the concentration of dinitrogen...Ch. 13.4 - The isomerization of cyclopropane, C3H6, to...Ch. 13.4 - A reaction believed to be either first or second...
Ch. 13.5 - Consider the following potential-energy curves for...Ch. 13.6 - Acetaldehyde, CH3CHO, decomposes when heated....Ch. 13.7 - Prob. 13.8ECh. 13.7 - Prob. 13.9ECh. 13.7 - Prob. 13.10ECh. 13.8 - The iodide-ion-catalyzed decomposition of hydrogen...Ch. 13.8 - Prob. 13.12ECh. 13.8 - Prob. 13.6CCCh. 13 - List the four variables or factors that can affect...Ch. 13 - Define the rate of reaction of HBr in the...Ch. 13 - Give at least two physical properties that might...Ch. 13 - A rate of reaction depends on four variables...Ch. 13 - Prob. 13.5QPCh. 13 - The reaction...Ch. 13 - The rate of a reaction is quadrupled when the...Ch. 13 - Prob. 13.8QPCh. 13 - The reaction A(g)B(g)+C(g) is known to be first...Ch. 13 - Prob. 13.10QPCh. 13 - Prob. 13.11QPCh. 13 - Sketch a potential-energy diagram for the...Ch. 13 - Draw a structural formula for the activated...Ch. 13 - Prob. 13.14QPCh. 13 - Prob. 13.15QPCh. 13 - Prob. 13.16QPCh. 13 - Prob. 13.17QPCh. 13 - Prob. 13.18QPCh. 13 - The dissociation of N2O4 into NO2, N2O4(g)2NO2(g)...Ch. 13 - Prob. 13.20QPCh. 13 - Prob. 13.21QPCh. 13 - Prob. 13.22QPCh. 13 - You are running the reaction 2A+BC+3D. Your lab...Ch. 13 - At a constant temperature, which of the following...Ch. 13 - Consider the reaction E+FG+H, which has the...Ch. 13 - The hypothetical reaction A+B+CD+E has the rate...Ch. 13 - Kinetics I Consider the hypothetical reaction A(g)...Ch. 13 - Kinetics II You and a friend are working together...Ch. 13 - Consider the reaction 3A2B+C. a One rate...Ch. 13 - Given the reaction 2A+BC+3D, can you write the...Ch. 13 - The reaction 2A(g)A2(g) is being run in each of...Ch. 13 - Prob. 13.32QPCh. 13 - You perform some experiments for the reaction AB+C...Ch. 13 - A friend of yours runs a reaction and generates...Ch. 13 - Prob. 13.35QPCh. 13 - You carry out the following reaction by...Ch. 13 - Prob. 13.37QPCh. 13 - The chemical reaction AB+C has a rate constant...Ch. 13 - Relate the rate of decomposition of NH4NO2 to the...Ch. 13 - For the reaction of hydrogen with iodine...Ch. 13 - To obtain the rate of the reaction...Ch. 13 - To obtain the rate of the reaction...Ch. 13 - Ammonium nitrite, NH4NO2, decomposes in solution,...Ch. 13 - Iron(III) chloride is reduced by tin(II) chloride....Ch. 13 - Azomethane, CH3NNCH3, decomposes according to the...Ch. 13 - Nitrogen dioxide, NO2, decomposes upon heating to...Ch. 13 - Hydrogen sulfide is oxidized by chlorine in...Ch. 13 - For the reaction of nitrogen monoxide, NO, with...Ch. 13 - Prob. 13.49QPCh. 13 - Prob. 13.50QPCh. 13 - In experiments on the decomposition of azomethane....Ch. 13 - Ethylene oxide. C2H4O, decomposes when heated to...Ch. 13 - Nitrogen monoxide NO, reacts with hydrogen to give...Ch. 13 - In a kinetic study of the reaction...Ch. 13 - Chlorine dioxide, ClO2, is a reddish-yellow gas...Ch. 13 - Iodide ion is oxidized to hypoiodite ion, IO, by...Ch. 13 - Sulfuryl chloride, SO2Cl2, decomposes when heated....Ch. 13 - Cyclopropane, C3H6, is converted to its isomer...Ch. 13 - A reaction of the form aA Products is second-order...Ch. 13 - A reaction of the form aA Products is second order...Ch. 13 - Ethyl chloride, CH3CH2Cl, used to produce...Ch. 13 - Cyclobutane, C4H8, consisting of molecules in...Ch. 13 - Methyl isocyanide, CH3NC, isomerizes, when heated,...Ch. 13 - Dinitrogen pentoxide, N2O5, decomposes when heated...Ch. 13 - In the presence of excess thiocyanate ion, SCN,...Ch. 13 - In the presence of excess thiocyanate ion, SCN,...Ch. 13 - A reaction of the form aA Products is second order...Ch. 13 - A reaction of the form aA Products is second order...Ch. 13 - In the presence of excess thiocyanate ion, SCN,...Ch. 13 - In the presence of excess thiocyanate ion, SCN,...Ch. 13 - It is found that a gas undergoes a zero-order...Ch. 13 - The reaction AB+C is found to be zero order. If it...Ch. 13 - Chlorine dioxide oxidizes iodide ion in aqueous...Ch. 13 - Methyl acetate, CH3COOCH3, reacts in basic...Ch. 13 - Sketch a potential-energy diagram for the reaction...Ch. 13 - Sketch a potential-energy diagram for the...Ch. 13 - In a series of experiments on the decomposition of...Ch. 13 - The reaction 2NOCl(g)2NO(g)+Cl2(g) has...Ch. 13 - The rate of a particular reaction increases by a...Ch. 13 - The rate of a particular reaction quadruples when...Ch. 13 - The following values of the rate constant were...Ch. 13 - The following values of the rate constant were...Ch. 13 - Nitrogen monoxide, NO, is believed to react with...Ch. 13 - The decomposition of ozone is believed to occur in...Ch. 13 - Identify the molecularity of each of the following...Ch. 13 - Prob. 13.86QPCh. 13 - Write a rate equation, showing the dependence of...Ch. 13 - Prob. 13.88QPCh. 13 - The isomerization of cyclopropane, C3H6, is...Ch. 13 - The thermal decomposition of nitryl chloride,...Ch. 13 - The reaction H2(g)+I2(g)2HI(g) may occur by the...Ch. 13 - Ozone decomposes to oxygen gas. 2O3(g)3O2(g) A...Ch. 13 - The following is a possible mechanism for a...Ch. 13 - Consider the following mechanism for a reaction in...Ch. 13 - A study of the decomposition of azomethane,...Ch. 13 - Nitrogen dioxide decomposes when heated....Ch. 13 - Prob. 13.97QPCh. 13 - Prob. 13.98QPCh. 13 - Methyl acetate reacts in acidic solution....Ch. 13 - Benzene diazonium chloride, C6H5NNCl, decomposes...Ch. 13 - What is the half-life of methyl acetate hydrolysis...Ch. 13 - What is the half-life of benzene diazonium...Ch. 13 - A compound decomposes by a first-order reaction....Ch. 13 - A compound decomposes by a first-order reaction....Ch. 13 - Butadiene can undergo the following reaction to...Ch. 13 - Prob. 13.106QPCh. 13 - Prob. 13.107QPCh. 13 - A second-order decomposition reaction run at 550oC...Ch. 13 - Prob. 13.109QPCh. 13 - Prob. 13.110QPCh. 13 - Prob. 13.111QPCh. 13 - Prob. 13.112QPCh. 13 - The decomposition of nitrogen dioxide,...Ch. 13 - Prob. 13.114QPCh. 13 - Prob. 13.115QPCh. 13 - Prob. 13.116QPCh. 13 - Nitryl bromide, NO2Br, decomposes into nitrogen...Ch. 13 - Tertiary butyl chloride reacts in basic solution...Ch. 13 - Urea, (NH2)2CO, can be prepared by heating...Ch. 13 - Prob. 13.120QPCh. 13 - A study of the gas-phase oxidation of nitrogen...Ch. 13 - The reaction of water with CH3Cl in acetone as a...Ch. 13 - The reaction of thioacelamidc with water is shown...Ch. 13 - Prob. 13.124QPCh. 13 - Prob. 13.125QPCh. 13 - Prob. 13.126QPCh. 13 - Prob. 13.127QPCh. 13 - Prob. 13.128QPCh. 13 - Prob. 13.129QPCh. 13 - Prob. 13.130QPCh. 13 - The rate constant for a certain reaction is 1.4 ...Ch. 13 - The decomposition of hydrogen peroxide is a first...Ch. 13 - Prob. 13.133QPCh. 13 - What is the rate law for the following gas-phase...Ch. 13 - A possible mechanism for a gas-phase reaction is...Ch. 13 - Say you run the following elementary, termolecular...Ch. 13 - Prob. 13.137QPCh. 13 - For the decomposition of one mole of nitrosyl...Ch. 13 - Given the following mechanism for a chemical...Ch. 13 - The following data were collected for the reaction...Ch. 13 - A hypothetical reaction has the two-step mechanism...Ch. 13 - Prob. 13.142QPCh. 13 - Prob. 13.143QPCh. 13 - Prob. 13.144QPCh. 13 - Dinitrogen pentoxide decomposes according to the...Ch. 13 - Prob. 13.146QPCh. 13 - Dinitrogen pentoxide, N2O5, undergoes first-order...Ch. 13 - Prob. 13.148QPCh. 13 - Hydrogen peroxide in aqueous solution decomposes...Ch. 13 - Nitrogen dioxide reacts with carbon monoxide by...Ch. 13 - Nitrogen monoxide reacts with oxygen to give...Ch. 13 - Nitrogen monoxide reacts with hydrogen as follows:...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- N Classify each of the following molecules as aromatic, antiaromatic, or nonaromatic. NH O aromatic O antiaromatic O nonaromatic O aromatic O antiaromatic O nonaromatic O aromatic O antiaromatic O nonaromatic Garrow_forwardThe conjugate base of alkanes is called alkides. Correct?.arrow_forwardName these organic compounds: structure Br name CH3 CH3 ☐ ☐arrow_forward
- HH H-C H -C-H HH Draw the Skeletal Structures & H Name the molecules HH H H H H-C-C-C-C-C-C-H HHH HHH H H HHHHHHH H-C-C-C-C-C-C-C-C-C-H HHHHH H H H Harrow_forwarddont provide AI solution .... otherwise i will give you dislikearrow_forwardName these organic compounds: structure name CH3 CH3 ☐ F F CH3 ☐ O Explanation Check 2025 McGraw Hill LLC. All Rights Reserved. Terms ofarrow_forward
- Classify each of the following molecules as aromatic, antiaromatic, or nonaromatic. ZI NH Explanation Check O aromatic O antiaromatic O nonaromatic O aromatic O antiaromatic H O nonaromatic O aromatic O antiaromatic O nonaromatic ×arrow_forwardPart I. Draw the stepwise reaction mechanism of each product (a, b, c, d, e, f) HO HO OH НОН,С HO OH Sucrose HO CH₂OH H N N HO -H H -OH KMnO4, Heat H OH CH₂OH (d) Phenyl Osatriazole OH НОН,С HO HO + Glacial HOAC HO- HO CH₂OH OH HO Fructose (a) Glucose OH (b) H₂N HN (c) CuSO4-5H2O, ethanol H N N N HO ·H H OH H OH N CH₂OH OH (f) Phenyl Osazone H (e) Carboxy phenyl osatriazole Figure 2.1. Reaction Scheme for the Total Synthesis of Fine Chemicalsarrow_forwardWhich molecule is the most stable? Please explain.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Kinetics: Initial Rates and Integrated Rate Laws; Author: Professor Dave Explains;https://www.youtube.com/watch?v=wYqQCojggyM;License: Standard YouTube License, CC-BY