
Concept explainers
Answer the following questions about aldosterone, a compound that helps to control the absorption of Na+and Cl- ions in the kidneys and, as a result, affects water retention.
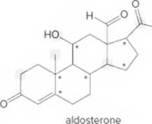
- Identify the
functional groups. - Draw in all lone pairs on O atoms.
- How many C’s does aldosterone contain?
- How many H’s are present at each C labeled with an asterisk (*)?
- Give the shape around each atom labeled in gray.
- Label all polar bonds.
(a)
Interpretation:
The functional group in aldosterone should be midentified.
Concept Introduction:
Functional groups are the atoms or group of atoms that gives chemical properties to an organic compound and also chemical reactivity centre. Examples of functional groups are aldehyde, ketone, alcohol, double bond.
Answer to Problem 83P
In aldosterone, functional groups present are alcohol, aldehyde, ketone and alkene.
Explanation of Solution
Functional groups are the atoms or group of atoms that gives chemical properties to an organic compound and also chemical reactivity centre. Structures of different functional groups are as follows:
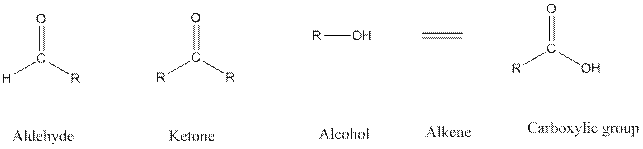
R represents any alkyl chain.
In functional group ketone carbonyl carbon is bonded to two alkyl groups, in aldehyde carbonyl carbon is attached to one alkyl group and one hydrogen atom, in alcohol functional group an −OH is bonded to an alkyl group.
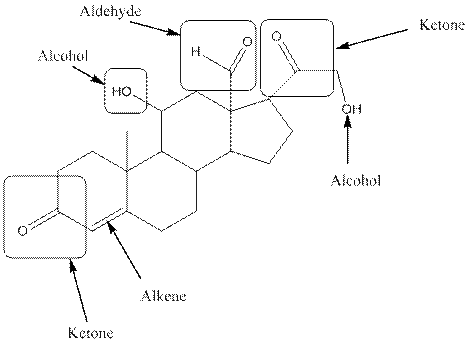
The compound aldosterone consists of two alcohol frunctional group, two ketone functional groups and one aldehyde functional group and one alkene. So, four types of functional group, that is, alcohol, aldehyde, ketone and alkene.
(b)
Interpretation:
All the lone pairs on oxygen atom of aldosterone should be drawn.
Concept Introduction:
Oxygen atom has six valence electrons. The electrons which are used in bond formation are called bonding electrons. The electrons that remain on an atom after bond formation are called lone pair electrons.
Answer to Problem 83P
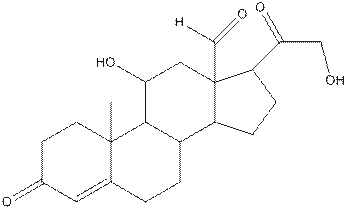
The structure of aldosterone having all lone pairs on oxygen atom is,
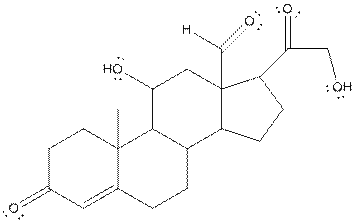
Explanation of Solution
The structure of aldosterone is as follows:
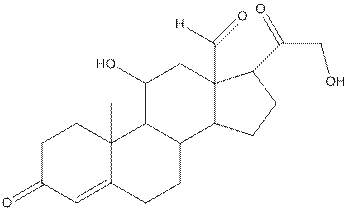
In the compound, three double bonded oxygen atoms present. Oxygen has six valence electrons. Two electrons in all double bonded oxygen atoms are used for formation of double bond. Remaining four electrons present as two lone pair. Also the compound contains three single bonded oxygen atoms. One electron is used for making O-H bond and one electron is used for making O-C bond. Remaining four electrons present as two lone pairs on each single bonded oxygen atom. Hence, the structure having all lone pairs on oxygen atom is,
(c)
Interpretation:
The number of carbon atoms in the structure of aldosterone should be determined.
Concept Introduction:
In skeletal structure the terminals represent methyl
Answer to Problem 83P
Twenty one carbon atoms present in the aldosterone.
Explanation of Solution
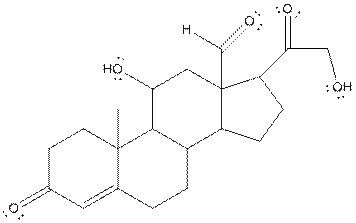
The structure of aldosterone is given as follows:
In skeletal structure the terminals represent methyl
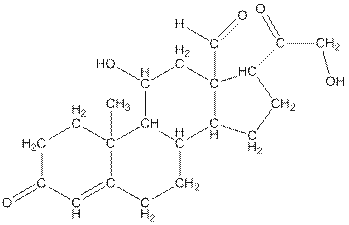
Hence, twenty one carbon atoms present in the compound.
(d)
Interpretation:
The number of hydrogen atoms present in the * carbon atoms in aldosterone should be determined.
Concept Introduction:
In skeletal structure the terminals represent methyl
Answer to Problem 83P
The * carbon atoms have total five hydrogen atoms.
Explanation of Solution
In skeletal structure the terminals represent methyl
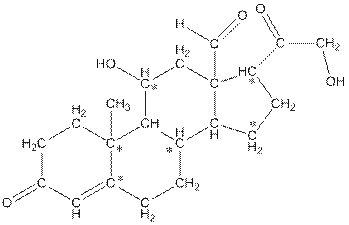
Hence, the * carbon atoms have total five hydrogen atoms.
(e)
Interpretation:
The shape around each indicated carbon atom in aldosterone should be determined.
Concept Introduction:
The following table should be used while determining the shape around an atom.
| Number of groups | Number of atoms | Number of lone pairs | Shape | Bond angle |
| 2 | 2 | 0 | Linear | |
| 3 | 3 | 0 | Trigonal planar | |
| 4 | 4 | 0 | Tetrahedral | |
| 4 | 3 | 1 | Trigonal pyramidal | |
| 4 | 2 | 2 | Bent |
Answer to Problem 83P
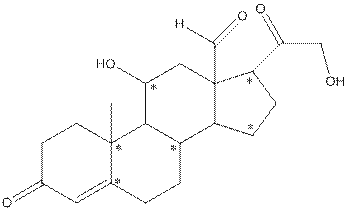
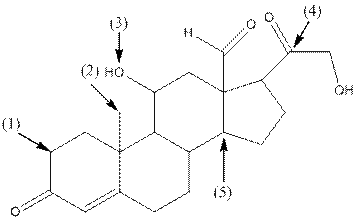
- Shape around carbon (1) is tetrahedral, shape around carbon (2) is tetrahedral, shape around oxygen (3) is bent, shape around carbon (4) is trigonal planar and shape around carbon (5) is tetrahedral.
Explanation of Solution
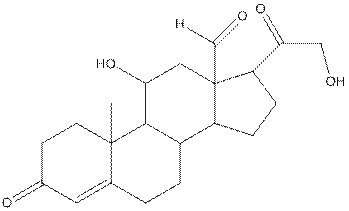
The complete structure of aldosterone is as follows:
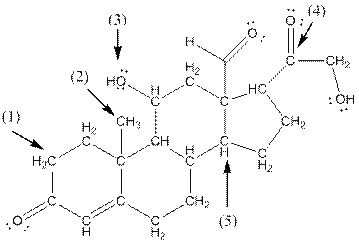
- Carbon (1) has four groups ( two hydrogen and two carbon) surrounding it. So, shape around carbon (1) is tetrahedral.
- Carbon (2) has four groups (three hydrogen and one carbon) surrounding it. So, shape around carbon (2) is tetrahedral.
- Oxygen (3) has two groups (one carbon and one hydrogen) and two lone pairs surrounding it. So, shape around oxygen (3) is bent.
- Carbon (4) has three groups (two carbon and one oxygen) surrounding it. So, shape around carbon (4) is trigonal planar.
- Carbon (5) has four groups (three carbon and one hydrogen) surrounding it. So, shape around carbon (5) is tetrahedral.
(f)
Interpretation:
All the polar bonds in aldosterone should be labeled.
Concept Introduction:
The unequal sharing of valence electrons in a bond is called polar bond. Polar bond result when the bond formed between two atoms in which one atom is more electronegative than the other one. One example of polar bond is
Structure of HCl is as follows:

In
Answer to Problem 83P
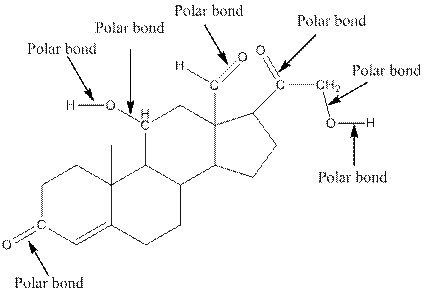
The structure of aldosterone with all polar bonds is as follows:
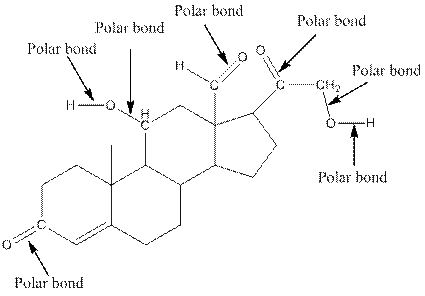
Explanation of Solution
In organic compound, most of the polar bonds formed between carbon and heteroatoms like oxygen, nitrogen, sulphur etc. To identify the polar bonds in the compound aldosterone, find the bonds between carbon and oxygen or oxygen and hydrogen because oxygen is more electronegative than carbon and oxygen is more electronegative than carbon. In aldostrone, five polar bonds are formed between carbon and oxygen (oxygen is more electronegative than carbon) and two polar bonds present between oxygen and hydrogen (oxygen is more electronegative than hydrogen). Thus, the polar bonds are:
Want to see more full solutions like this?
Chapter 11 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- 6 Which of the following are likely to be significant resonance structures of a resonance hybrid? Draw another resonance structure for each of the compounds you select as being a resonance form. (A Br: Br: A B C D Earrow_forwardWrite the systematic (IUPAC) name for the following organic molecules. Note for advanced students: you do not need to include any E or Z prefixes in your names. Br structure Br Br Oweuarrow_forwardConservation of mass was discussed in the background. Describe how conservation of mass (actual, not theoretical) could be checked in the experiment performed.arrow_forward
- What impact would adding twice as much Na2CO3 than required for stoichiometric quantities have on the quantity of product produced? Initial results attachedarrow_forwardGiven that a theoretical yield for isolating Calcium Carbonate in this experiment would be 100%. From that information and based on the results you obtained in this experiment, describe your success in the recovery of calcium carbonate and suggest two possible sources of error that would have caused you to not obtain 100% yield. Results are attached form experimentarrow_forward5) Calculate the flux of oxygen between the ocean and the atmosphere(2 pts), given that: (from Box 5.1, pg. 88 of your text): Temp = 18°C Salinity = 35 ppt Density = 1025 kg/m3 Oxygen concentration measured in bulk water = 263.84 mmol/m3 Wind speed = 7.4 m/s Oxygen is observed to be about 10% initially supersaturated What is flux if the temperature is 10°C ? (2 pts) (Hint: use the same density in your calculations). Why do your calculated values make sense (or not) based on what you know about the relationship between gas solubility and temperature (1 pt)?arrow_forward
- The following 'H NMR spectrum was taken with a 750 MHz spectrometer: 1.0 0.5 0.0 10.0 9.0 8.0 7.0 6.0 5.0 4.0 3.0 ' 2.0 1.0 0.0 (ppm) What is the difference Av in the frequency of RF ac Δν ac radiation absorbed by the a and c protons? (Note: it's not equal to the difference in chemical shifts.) Round your answer to 2 significant digits, and be sure it has an appropriate unit symbol. = O O a will shift left, c will shift right. O a will shift right, c will shift left. a and c will both shift left, with more space between them. Suppose a new spectrum is taken with a 500 MHz spectrometer. What will be true about this new spectrum? O a and c will both shift left, with less space between them. O a and c will both shift right, with more space between them. O a and c will both shift right, with less space between them. Which protons have the largest energy gap between spin up and spin down states? O None of the above. ○ a Ob Explanation Check C Ar B 2025 McGraw Hill LLC. All Rights Reserved.…arrow_forwardWhat mass of Na2CO3 must you add to 125g of water to prepare 0.200 m Na2CO3? Calculate mole fraction of Na2CO3, mass percent, and molarity of the resulting solution. MM (g/mol): Na2CO3 105.99; water 18.02. Final solution density is 1.04 g/mL.arrow_forward(ME EX2) Prblms Can you please explain problems to me in detail, step by step? Thank you so much! If needed color code them for me.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

