
Biochemistry: Concepts and Connections (2nd Edition)
2nd Edition
ISBN: 9780134641621
Author: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 14P
The steady-state kinetics of an enzyme are studied the absence and presence of an inhibitor (Inhibitor A). The initial rate is given as a function of substrate concentration in the following table:
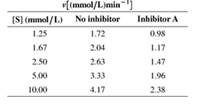
a. What kind of inhibition (competitive, uncompetitive. or mixed) is involved?
b. Determine Vmax and KM the absence and presence of inhibitor.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
draw in the structure of each amino acid (as L-amino acids) using the Fischer projection style. an example has been included. Draw the structure for glycine, alanine, valine, isoleucine, methionine, proline, phenylalanine, tryptophan, serine, threonine, asparagine, glutamine, lysine, arginine, aspartic acid, glutamic acid, histidine, tyrosine, cysteine
draw in the structure of each amino acid (as L-amino acids) using the Fischer projection style. an example has been included
draw in the structure of each amino acid (as L-amino acids) using the Fischer projection style. an example has been included
Chapter 8 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
Ch. 8 - Prob. 1PCh. 8 - The enzyme urease catalyzes the hydrolysis of urea...Ch. 8 - An enzyme contains an active site aspartic acid...Ch. 8 - The folding and unfolding rate constants for a...Ch. 8 - In some reactions, in which a protein molecule is...Ch. 8 - Would you expect an “enzyme” designed to bind to...Ch. 8 - The initial rate for an enzyme-catalyzed reaction...Ch. 8 - a. If the total enzyme concentration in Problem 7...Ch. 8 - Prob. 9PCh. 8 - Prob. 10P
Ch. 8 - The following data describe the catalysis of...Ch. 8 - At 37 oC, the serine protease subtilisin has kcat...Ch. 8 - The accompanying figure shows three...Ch. 8 - The steady-state kinetics of an enzyme are studied...Ch. 8 - The same enzyme as in Problem 14 is studied in the...Ch. 8 - Enalapril is an anti-hypertension “pro-drug"...Ch. 8 - Initial rate data for an enzyme that obeys...Ch. 8 - Prob. 18PCh. 8 - Suggest the effects of each of the following...Ch. 8 - The inhibitory effect of an uncompetitive...Ch. 8 - Prob. 21PCh. 8 - Prob. 22PCh. 8 - Prob. 23PCh. 8 - In kinetics experiments, the hydrolysis of the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw out the following peptide H-R-K-E-D at physiological pH (~7.4). Make sure toreference table 3.1 for pKa values.arrow_forwardThe table provides the standard reduction potential, E', for relevant half-cell reactions. Half-reaction E'° (V) Oxaloacetate² + 2H+ + 2e malate²- -0.166 Pyruvate + 2H+ + 2e → lactate -0.185 Acetaldehyde + 2H+ + 2e¯ →→→ ethanol -0.197 NAD+ + H+ + 2e--> NADH -0.320 NADP+ + H+ + 2e →→ NADPH Acetoacetate + 2H+ + 2e¯ - -0.324 B-hydroxybutyrate -0.346 Which of the reactions listed would proceed in the direction shown, under standard conditions, in the presence of the appropriate enzymes? Malate + NAD+ oxaloacetate + NADH + H+ Malate + pyruvate oxaloacetate + lactate Pyruvate + NADH + H+ lactate + NAD+ Pyruvate + p-hydroxybutyrate lactate + acetoacetate Acetaldehyde + succinate ethanol + fumerate Acetoacetate + NADH + H+ → B-hydroxybutyrate + NAD+arrow_forwardArrange the four structures in order from most reduced to most oxidized. Most reduced R-CH2-CH3 R-CH2-CH₂-OH R-CH,-CHO R-CH₂-COO Most oxidizedarrow_forward
- for each pair of biomolecules, identify the type of reaction (oxidation-reduction, hydrolysis, isomerization, group transfer, or nternal rearrangement) required to convert the first molecule to the second. In each case, indicate the general type of enzyme and cofactor(s) c reactants required, and any other products that would result. R-CH-CH-CH-C-S-COA A(n) A(n) A(n) A(n) Palmitoyl-CoA R-CH-CH=CH-C-S-CoA ° trans-A-Enoyl-CoA reaction converts palmitoyl-CoA to trans-A2-enoyl-CoA. This reaction requires and also produces Coo HN-C-H CH₂ CH₂ CH CH CH, CH, L-Leucine CH, CH, D-Leucine 8/6881 COO HÌNH: reaction converts L-leucine to D-leucine. This reaction is catalyzed by a(n) H-C-OH H-C-OH C=0 HO-C-H HO-C-H H-C-OH H-C-OH H-C-OH CH,OH Glucose H-C-OH CH,OH Fructose OH OH OH CH-C-CH₂ reaction converts glucose to fructose. This reaction is catalyzed by a(n) OH OH OPO I CH-C-CH H Glycerol Glycerol 3-phosphate H reaction converts glycerol to glycerol 3-phosphate. This reaction requires H,N- H,N H…arrow_forwardAfter adding a small amount of ATP labeled with radioactive phosphorus in the terminal position, [7-32P]ATP, to a yeast extract, a researcher finds about half of the 32P activity in P; within a few minutes, but the concentration of ATP remains unchanged. She then carries out the same experiment using ATP labeled with 32P in the central position, [ẞ-³2P]ATP, but the 32P does not appear in P; within such a short time. Which statements explain these results? Yeast cells reincorporate P; released from [ß-³2P]ATP into ATP more quickly than P¡ released from [y-³2P]ATP. Only the terminal (y) phosphorous atom acts as an electrophilic target for nucleophilic attack. The terminal (y) phosphoryl group undergoes a more rapid turnover than the central (B) phosphate group. Yeast cells maintain ATP levels by regulating the synthesis and breakdown of ATP. Correct Answerarrow_forwardCompare the structure of the nucleoside triphosphate CTP with the structure of ATP. NH₂ 0- 0- 0- ·P—O—P—O—P—O—CH₂ H H H H OH OH Cytidine triphosphate (CTP) Consider the reaction: ATP + CDP ADP + CTP NH 0- 0- 0- ¯0— P—O— P—O—P-O-CH₂ H Η о H H OH OH Adenosine triphosphate (ATP) NH₂ Now predict the approximate K'eq for this reaction. Now predict the approximate AG for this reaction. Narrow_forward
- The standard free energy, AGO, of hydrolysis of inorganic polyphosphate, polyP, is about −20 kJ/mol for each P; released. In a cell, it takes about 50 kJ/mol of energy to synthesize ATP from ADP and Pi. ○ P O Inorganic polyphosphate (polyP) Is it feasible for a cell to use polyP to synthesize ATP from ADP? Why or why not? No. The reaction is unidirectional and always proceeds in the direction of polyP synthesis from ATP. Yes. If [ADP] and [polyP] are kept high, and [ATP] is kept low, the actual free-energy change would be negative. No. The synthesis of ATP from ADP and P; has a large positive G'o compared to polyP hydrolysis. Yes. The hydrolysis of polyP has a sufficiently negative AG to overcome the positive AGO of ATP synthesis. Correct Answerarrow_forwardIn the glycolytic pathway, a six-carbon sugar (fructose 1,6-bisphosphate) is cleaved to form two three-carbon sugars, which undergo further metabolism. In this pathway, an isomerization of glucose 6-phosphate to fructose 6-phosphate (as shown in the diagram) occurs two steps before the cleavage reaction. The intervening step is phosphorylation of fructose 6-phosphate to fructose 1,6-bisphosphate. H H | H-C-OH H-C-OH C=0 HO-C-H HO-C-H phosphohexose isomerase H-C-OH H-C-OH H-C-OH H-C-OH CH₂OPO CH₂OPO Glucose 6-phosphate Fructose 6-phosphate What does the isomerization step accomplish from a chemical perspective? Isomerization alters the molecular formula of the compound, allowing for subsequent phosphorylation. Isomerization moves the carbonyl group, setting up a cleavage between the central carbons. Isomerization causes the gain of electrons, allowing for the eventual release of NADH. Isomerization reactions cause the direct production of energy in the form of ATP.arrow_forwardFrom data in the table, calculate the AG value for the reactions. Reaction AG' (kJ/mol) Phosphocreatine + H₂O →>> creatine + P -43.0 ADP + Pi → ATP + H₂O +30.5 Fructose +P → fructose 6-phosphate + H₂O +15.9 Phosphocreatine + ADP creatine + ATP AG'O ATP + fructose → ADP + fructose 6-phosphate AG'° kJ/mol kJ/molarrow_forward
- Macmillan Learning The phosphorylation of glucose to glucose 6-phosphate is the initial step in the catabolism of glucose. The direct phosphorylation of glucose by P, is described by the equation Glucose + P ← glucose 6-phosphate + H₂O AG = 13.8 kJ/mol Coupling ATP hydrolysis to glucose phosphorylation makes thermodynamic sense, but consider how the coupling might take place. Given that coupling requires a common intermediate, one conceivable mechanism is to use ATP hydrolysis to raise the intracellular concentration of Pi. The increase in P; concentration would drive the unfavorable phosphorylation of glucose by Pi- Is increasing the P; concentration a reasonable way to couple ATP hydrolysis and glucose phosphorylation? No. The phosphate salts of divalent cations would be present in excess and precipitate out. Yes. Increasing the concentration of P; would decrease K'eq and shift equilibrium to the right. Yes. The extra ATP hydrolysis would provide enough free energy to drive the…arrow_forwardThe phosphorylation of glucose to glucose 6-phosphate is the initial step in the catabolism of glucose. The direct phosphorylation of glucose by P, is described by the equation Glucose + P → glucose 6-phosphate + H₂O AG' = 13.8 kJ/mol In principle, at least, one way to increase the concentration of glucose 6-phosphate (G6P) is to drive the equilibrium reaction to the right by increasing the intracellular concentrations of glucose and Pj. The maximum solubility of glucose is less than 1 M, and the normal physiological concentration of G6P is 250 μM. Assume a fixed concentration of P, at 4.8 mM. The calculated value of K'cq is 4.74 × 10-³ M-¹. Calculate the intracellular concentration of glucose when the equilibrium concentration of glucose 6-phosphate is 250 μM, the normal physiological concentration. [glucose] = 10.99 Correct Answer Would increasing the concentration of glucose be a physiologically reasonable way to increase the concentration of G6P? No. Because the concentration of P,…arrow_forwardCalculate the equilibrium constant for the phosphorylation of glucose to glucose 6-phosphate at 37.0 °C. K'eq = M-' In the rat hepatocyte, the physiological concentrations of glucose and P, are maintained at approximately 4.8 mM. What is the equilibrium concentration of glucose 6-phosphate (G6P) obtained by the direct phosphorylation of glucose by P.? [G6P] = Does this reaction represent a reasonable metabolic step for the catabolism of glucose? Why or why not? Yes, because the value of AG" is positive. No, because the K'eq is too large for the reaction to proceed in the forward direction. Yes, because AG is negative at the calculated value of K'eq No, because [G6P] is likely to be higher than the calculated value. Marrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Cengage Learning


Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning


Enzyme Kinetics; Author: MIT OpenCourseWare;https://www.youtube.com/watch?v=FXWZr3mscUo;License: Standard Youtube License