
Biochemistry: Concepts and Connections (2nd Edition)
2nd Edition
ISBN: 9780134641621
Author: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 17P
Initial rate data for an enzyme that obeys Michaelis-Menten kinetics are shown in the following table. When the enzyme concentration is 3 nmol ml-1, a Lineweaver-Burk plot of this data gives a line with a y-intercept of 0.00426 (µmol- ml s).
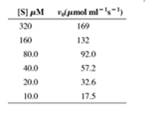
- Calculate kcat for the reaction.
- Calculate KM for the enzyme.
- When the reactions in part (b) are repeated in the presence of 12 µM of an uncompetitive inhibitor, the y-intercept of the Lineweaver-Burk plot is 0.352 (µmol-1 ml s). Calculate K’I for the inhibitor.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
18. Which one of the compounds below is the major organic product obtained from
the following series of reactions?
1. BH3
2. H2O2, NaOH
H₂CrO4
CH2N2
oro ororos
A
B
C
D
E
17. Which one of the compounds below is the major organic product obtained from
the following series of reactions?
CI
benzyl alcohol
OH
PBr3
Mg
1. CO2
SOCl2
?
ether
2. H+, H₂O
CI
Cl
HO
OH
CI
Cl
A
B
C
D
E
14. What is the IUPAC name of this compound?
A) 6-hydroxy-4-oxohexanenitrile
B) 5-cyano-3-oxo-1-pentanol
C) 5-cyano-1-hydroxy-3-pentanone
D) 1-cyano-5-hydroxy-3-pentanone
E) 5-hydroxy-3-oxopentanenitrile
HO.
CN
Chapter 8 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
Ch. 8 - Prob. 1PCh. 8 - The enzyme urease catalyzes the hydrolysis of urea...Ch. 8 - An enzyme contains an active site aspartic acid...Ch. 8 - The folding and unfolding rate constants for a...Ch. 8 - In some reactions, in which a protein molecule is...Ch. 8 - Would you expect an “enzyme” designed to bind to...Ch. 8 - The initial rate for an enzyme-catalyzed reaction...Ch. 8 - a. If the total enzyme concentration in Problem 7...Ch. 8 - Prob. 9PCh. 8 - Prob. 10P
Ch. 8 - The following data describe the catalysis of...Ch. 8 - At 37 oC, the serine protease subtilisin has kcat...Ch. 8 - The accompanying figure shows three...Ch. 8 - The steady-state kinetics of an enzyme are studied...Ch. 8 - The same enzyme as in Problem 14 is studied in the...Ch. 8 - Enalapril is an anti-hypertension “pro-drug"...Ch. 8 - Initial rate data for an enzyme that obeys...Ch. 8 - Prob. 18PCh. 8 - Suggest the effects of each of the following...Ch. 8 - The inhibitory effect of an uncompetitive...Ch. 8 - Prob. 21PCh. 8 - Prob. 22PCh. 8 - Prob. 23PCh. 8 - In kinetics experiments, the hydrolysis of the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- 13. What is the IUPAC name of this compound? A) 5-hydroxy-3,3-dimethylpentanoic acid B) 3,3-dimethylpentanoic acid C) 3,3-dimethyl-1-oxo-1,5-pentanediol D) 1,5-dihydroxy-3,3-dimethylpentanal E) 4-hydroxy-2,2-dimethylbutanoic acid HO OHarrow_forwardHelp me understand how carbon disulfide leads to toxicity in the brain, using terms like distal axonopathy, neurofilaments, covalent cross-linking, adducts, etc.,...please intuitively explain what is happening and where and the effects of it. For example, I know that CS2 reacts with amide and sulfhydryl groups on proteins, but what proteins exactly and where are they located?arrow_forwardWhat is the standard free energy change (in kJ/mole) of the spontaneous reaction between Oxygen and NADH to form H2O2 and NAD+?arrow_forward
- Redox Chemistry: Give standard free energy changes expected for the following reactions:-Succinate -> fumarate (using FAD/FADH2)-Oxaloacetate -> Malate (using NAD/NADH)-NADH --> NAD+ (using FMN/FMNH2)-CoQ --> CoQH2 (using Cytochrome C)arrow_forwardGive examples of balanced redox reactions that match the following:-Catabolic-Anabolic-Oxidative-Reductivearrow_forwardIf there are 20uM of a GLUT2 transporter on the surface of a cell, each able to move 8 per second, and 50mM glucose outside of the cell, what is the flux into the cell in mM/sec?arrow_forward
- A transporter is responsible for antiporting calcium and glucose. The transporter brings glucose into the cell and sends calcium out of the cell. If blood [calcium] = 2.55mM and intracellular [calcium] = 7uM, blood [glucose] = 5.2mM, and intracellular [glucose] = 40uM, what is the free energy of transport? Assume a membrane potential of 62mV (negative inside).arrow_forwardAn ATP-coupled transporter is used to import 1 phosphate from the extracellular environment. Intracellular phosphate exists at 65mM, while it is 2mM outside.Assume a free energy change of ATP hydrolysis of -42.7 kJ/mol. What is the net free energy change of the coupled reaction? Assume a membrane potential of 70mV.arrow_forwardAnother transporter brings 3 chloride ions into the cell. Outside, chloride has a concentration of 107mM, and 4mM inside the cell. Assuming a membrane potential of 62mV (negative inside), what is the free energy of transport of these ions?arrow_forward
- For the Oxaloacetate -> Malate reaction, assume the normal ratio of NAD/NADH, what is the maximum ratio of Malate/Oxaloacetate that will allow reaction progress?arrow_forwardA particular particle is trying to cross a membrane by simple diffusion from a high concentration of 20mM to a low concentration of 20uM. If a membrane is 15uM in width, and the diffusion coefficient of the particle is 5 uM/sec, what is the influx in uM/sec?arrow_forwardMechanisms: 1. Give a full arrow-pushing mechanism for the hydrolysis of the gamma phosphate of ATP by an ATPase. 2. Give a full arrow pushing mechanism of the spontaneous redox reaction between NAD+/NADH and oxaloacetate/malate.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning



Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning

Enzyme Kinetics; Author: MIT OpenCourseWare;https://www.youtube.com/watch?v=FXWZr3mscUo;License: Standard Youtube License