
Concept explainers
Interpretation:
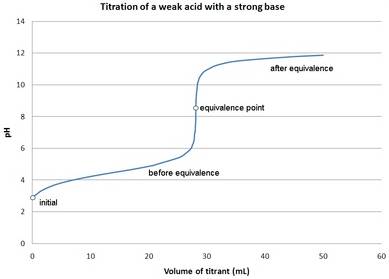
The pH values for the titration of 500 mL of 0.010 M acetic acid with 0.010 M KOH should be calculated and the titration curve should be drawn.
Concept Introduction:
For a weak acid or a weak base, the dissociation reaction is in equilibrium and the equilibrium constant is known as acid or base dissociation constant. For such a solution, concentration of each species is calculated using ICE table.
The pH of solution is calculated as follows:
Here,
For a buffer solution, pH is calculated using Henderson-Hasselbalch equation as follows:
Here, pH is
Pictorial representation:
The titration curve for weak acid and strong base is as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
- Balance the following equation and list of coefficients in order from left to right. SF4+H2O+—-> H2SO3+HFarrow_forwardProblem 15 of 15 Submit Using the following reaction data points, construct Lineweaver-Burk plots for an enzyme with and without an inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Using the information from this plot, determine the type of inhibitor present. 1 mM-1 1 s mM -1 [S]' V' with 10 μg per 20 54 10 36 20 5 27 2.5 23 1.25 20 Answer: |||arrow_forward12:33 CO Problem 4 of 15 4G 54% Done On the following Lineweaver-Burk -1 plot, identify the by dragging the Km point to the appropriate value. 1/V 40 35- 30- 25 20 15 10- T Км -15 10 -5 0 5 ||| 10 15 №20 25 25 30 1/[S] Г powered by desmosarrow_forward
- 1:30 5G 47% Problem 10 of 15 Submit Using the following reaction data points, construct a Lineweaver-Burk plot for an enzyme with and without a competitive inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. 1 -1 1 mM [S]' s mM¹ with 10 mg pe 20 V' 54 10 36 > ст 5 27 2.5 23 1.25 20 Answer: |||arrow_forwardProblem 14 of 15 Submit Using the following reaction data points, construct Lineweaver-Burk plots for an enzyme with and without an inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Using the information from this plot, determine the type of inhibitor present. 1 mM-1 1 s mM -1 [S]' V' with 10 μg per 20 54 10 36 20 5 27 2.5 23 1.25 20 Answer: |||arrow_forward12:36 CO Problem 9 of 15 4G. 53% Submit Using the following reaction data points, construct a Lineweaver-Burk plot by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Based on the plot, determine the value of the catalytic efficiency (specificity constant) given that the enzyme concentration in this experiment is 5.0 μ.Μ. 1 [S] ¨‚ μM-1 1 V sμM-1 100.0 0.100 75.0 0.080 50.0 0.060 15.0 0.030 10.0 0.025 5.0 0.020 Answer: ||| O Гarrow_forward
- Problem 11 of 15 Submit Using the following reaction data points, construct a Lineweaver-Burk plot for an enzyme with and without a noncompetitive inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. 1 -1 1 mM [S]' 20 V' s mM¹ with 10 μg per 54 10 36 > ст 5 27 2.5 23 1.25 20 Answer: |||arrow_forwardProblem 13 of 15 Submit Using the following reaction data points, construct Lineweaver-Burk plots for an enzyme with and without an inhibitor by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Using the information from this plot, determine the type of inhibitor present. 1 mM-1 1 s mM -1 [S]' V' with 10 μg per 20 54 10 36 20 5 27 2.5 23 1.25 20 Answer: |||arrow_forward12:33 CO Problem 8 of 15 4G. 53% Submit Using the following reaction data points, construct a Lineweaver-Burk plot by dragging the points to their relevant coordinates on the graph and drawing a line of best fit. Based on the plot, determine the value of kcat given that the enzyme concentration in this experiment is 5.0 μM. 1 [S] , мм -1 1 V₁ s μM 1 100.0 0.100 75.0 0.080 50.0 0.060 15.0 0.030 10.0 0.025 5.0 0.020 Answer: ||| Гarrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





