
General, Organic, & Biological Chemistry
3rd Edition
ISBN: 9780073511245
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 14.40P
Answer each question about alcohol B.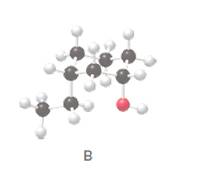
- Draw a skeletal structure for B.
- Classify’ the hydroxyl group as l°, 2°, or 3°.
- Give the RIP AC name for B.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Is this statement true or false and why?
6. Draw the following compounds.
a. 2-pentanol
b. ethylpropylamine
c. methyl propyl ether
d. butyl propyl ketone
abayoqtheo ghidollo
WHO
H3--3
HO
HO HO HO HO
What is the IUPAC name of the structure?
a. ethanoic acid
b. butanoic acid
c. methyl propanoate
d. ethyl propanoate
Chapter 14 Solutions
General, Organic, & Biological Chemistry
Ch. 14.1 - Prob. 14.1PCh. 14.2 - Prob. 14.2PCh. 14.2 - Classify each hydroxyl group in sorbitol as 1°,...Ch. 14.2 - Prob. 14.4PCh. 14.2 - Prob. 14.5PCh. 14.3 - Prob. 14.6PCh. 14.3 - Give the structure corresponding to each name a....Ch. 14.5 - Draw the products formed when each alcohol is...Ch. 14.5 - Prob. 14.9PCh. 14.5 - Prob. 14.10P
Ch. 14.5 - Prob. 14.11PCh. 14.6 - Prob. 14.12PCh. 14.6 - Prob. 14.13PCh. 14.7 - Prob. 14.14PCh. 14.7 - Prob. 14.15PCh. 14.7 - Prob. 14.16PCh. 14.7 - Prob. 14.17PCh. 14.8 - (a) Translate the hall and stick model of...Ch. 14.8 - Prob. 14.19PCh. 14.9 - Prob. 14.20PCh. 14.9 - Prob. 14.21PCh. 14.9 - Prob. 14.22PCh. 14.9 - Prob. 14.23PCh. 14.9 - Prob. 14.24PCh. 14.9 - Prob. 14.25PCh. 14.10 - Prob. 14.26PCh. 14.10 - Prob. 14.27PCh. 14.10 - Prob. 14.28PCh. 14 - Prob. 14.29PCh. 14 - Prob. 14.30PCh. 14 - Prob. 14.31PCh. 14 - Classify each halide hi A as 1°, 2°, or 3°. A is a...Ch. 14 - Prob. 14.33PCh. 14 - Draw the structure of a molecule that fits each...Ch. 14 - Draw the structure of the six constitutional...Ch. 14 - Draw the structure of the four constitutional...Ch. 14 - Prob. 14.37PCh. 14 - Prob. 14.38PCh. 14 - Prob. 14.39PCh. 14 - Answer each question about alcohol B. Draw a...Ch. 14 - Prob. 14.41PCh. 14 - Give the IUAPC name for each alcohol.Ch. 14 - Prob. 14.43PCh. 14 - Prob. 14.44PCh. 14 - Prob. 14.45PCh. 14 - Prob. 14.46PCh. 14 - Draw the structures and give the IUPAC names for...Ch. 14 - Prob. 14.48PCh. 14 - Prob. 14.49PCh. 14 - Prob. 14.50PCh. 14 - Give the structure corresponding to each IUPAC...Ch. 14 - Give the structure corresponding to each IUPAC...Ch. 14 - Which compound in each pair has the higher boiling...Ch. 14 - Rank the compounds in order of increasing melting...Ch. 14 - Rank the following compounds in order of...Ch. 14 - Rank the following compounds in order of...Ch. 14 - Prob. 14.57PCh. 14 - Prob. 14.58PCh. 14 - Prob. 14.59PCh. 14 - Prob. 14.60PCh. 14 - Prob. 14.61PCh. 14 - Prob. 14.62PCh. 14 - Prob. 14.63PCh. 14 - Prob. 14.64PCh. 14 - Prob. 14.65PCh. 14 - Prob. 14.66PCh. 14 - Prob. 14.67PCh. 14 - Prob. 14.68PCh. 14 - Prob. 14.69PCh. 14 - Prob. 14.70PCh. 14 - Prob. 14.71PCh. 14 - Prob. 14.72PCh. 14 - Prob. 14.73PCh. 14 - Prob. 14.74PCh. 14 - Prob. 14.75PCh. 14 - Prob. 14.76PCh. 14 - Prob. 14.77PCh. 14 - Prob. 14.78PCh. 14 - Prob. 14.79PCh. 14 - Prob. 14.80PCh. 14 - Prob. 14.81PCh. 14 - Prob. 14.82PCh. 14 - Prob. 14.83PCh. 14 - Prob. 14.84PCh. 14 - Prob. 14.85PCh. 14 - Prob. 14.86PCh. 14 - With reference to the halogenated organic...Ch. 14 - Prob. 14.88PCh. 14 - Prob. 14.89PCh. 14 - Prob. 14.90PCh. 14 - Write out the chemical reaction that occurs when a...Ch. 14 - Prob. 14.92PCh. 14 - Prob. 14.93PCh. 14 - Lactic acid [CH3CH(OH)CO2H] gives sour milk its...Ch. 14 - Prob. 14.95PCh. 14 - Prob. 14.96PCh. 14 - Prob. 14.97PCh. 14 - Prob. 14.98PCh. 14 - Prob. 14.99PCh. 14 - Answer the following questions about alcohol B....Ch. 14 - Prob. 14.101CPCh. 14 - Dehydration of alcohol C forms two products of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. What are the isomers of hexane. 2. Arrange the isomers of hexane in order of increasing boiling point.arrow_forwarda. Draw two different structures with the molecular formula C,H,0. b. Name the functional group in each structure. c. Which one will have the higher boiling point, and why?arrow_forwardAlcohols with two or more - OH groups have higher boiling point.I. Alcohols with more than two -OH groups are more water soluble than similar alcohols with only one -OH group.A.BOTH statements are CORRECTB. BOTH statements are INCORRECTC. FIRST statement is CORRECT. SECOND is INCORRECT D.FIRST statement is INCORRECT, SECOND is CORRECT 2. l. An alcohol is an organic compound with at least one hydroxyl (-OH) group bound to unsaturated carbon atom. II. Alcohols have higher boiling points than corresponding alkynes.A.FIRST statement is CORRECT. SECOND is INCORRECTO FIRST statement is INCORRECT SECOND Is CORRECTAre INCORKI© bUlk statements are CORRECTIarrow_forward
- True or False. Organic compounds synthesized in the laboratory have the same chemical and ohysical properties as those synthesized in living organisms. True or False. An alcohol, an aldehyde, and a ketone each contain a single oxygen atom. Multiole choice 1. Which of the following molecules contains a carbonyl group? A. 2-Propanol B. Diethyl ether C. 3,7-Dimethyloctanal D. 2-Methyl-1-butene 2. Methanol has all the following properties except? A. Colorless liquid B. Poisonous C. Boils below 100°C D. Does not hydrogen bondarrow_forwardClassify each alkyl halide and alcohol as 1°, 2°, or 3°.arrow_forward2. Classify each compound as an alcohol, aldehyde, sugar, ketone, methyl ketone, or hydrocarbon (there should only be one of each type of compound). Hexane Ethanol Benzaldehyde Cyclohexanone Acetone (Propanone) Glucose emplearrow_forward
- 1. Which functional group is found in aldehydes? a. CHO b. -CH;OH c. -COOH d. RCOR' 2. Which functional group is found in ketones? a. -CHO CH:OH c. -COOH RCOR' 3. Which structural feature is common to aldehydes and ketones? a, an oxygen atom bonded to both a carbon atom and a hydrogen atom b. an oxygen atom bonded to two carbon atoms c. an oxygen atom double bonded to a carbon atom d. two oxygen atoms bonded to the same carbon atom 4. What is the IUPAC name for the following compound? a. I-pentaldehyde b. l-pentanal c. pentanal d. pentanealdehyde Cetoa of Fiarrow_forwardPlease explain the chosen letter. Which is NOT a physical property of alcohols or phenols? a. Phenols are generally only slightly soluble in water. b. The solubilities of normal primary alcohols in water decrease with increasing molecular weight. c. The hydroxyl group of an alcohol is nonpolar. d. Due to hydrogen bonding, boiling points of alcohols are much higher than those of corresponding alkanes.arrow_forwardWhat is the IUPAC name of the structure? a. propyl methanoate b. methyl propanoate c. propanoic acid d. 2-methyl propanoic acidarrow_forward
- Part A: Write the structure for methyl propyl ether, an anesthetic known as Neothyl. Part B: Write the structure for dimethyl ether, used as a compressed gas to "freeze" warts from the skin. What alcohol is an isomer of dimethyl ether?arrow_forwardIdentify the IUPAC name of the given structure. A. 2 - propylpropanal B. hexanal C. 2 - methylpentanalarrow_forwardQuestion 7arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY