
(a)
Interpretation:
An acceptable name for the following ether should be determined:
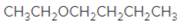
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the
(b)
Interpretation:
An acceptable name for the following ether should be determined:
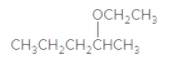
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the IUPAC nomenclature; one alkyl group is named as a hydrocarbon chain and the other part is named as a part of substituent bonded to that chain. The simpler alkyl group with the oxygen atom should name as an alkoxy substituent and the -yl suffix of the alkyl replaced with- oxy suffix. Remaining longest alkyl chain is named as an alkane by considering the alkoxy group as a substituent bonded to the main carbon chain.
(c)
Interpretation:
An acceptable name for the following ether should be determined:
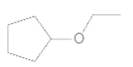
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the IUPAC nomenclature; one alkyl group is named as a hydrocarbon chain and the other part is named as a part of substituent bonded to that chain. The simpler alkyl group with the oxygen atom should name as an alkoxy substituent and the -yl suffix of the alkyl replaced with- oxy suffix. Remaining longest alkyl chain is named as an alkane by considering the alkoxy group as a substituent bonded to the main carbon chain.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, & Biological Chemistry
- Synthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- If possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forwardSynthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forward
- We mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forwardIndicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forward
- Indicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forwardQuestion 3 (4 points), Draw a full arrow-pushing mechanism for the following reaction Please draw all structures clearly. Note that this intramolecular cyclization is analogous to the mechanism for halohydrin formation. COH Br + HBr Brarrow_forwardIndicate the products obtained if 2,2-dimethylpropanal and acetaldehyde are mixed with sodium ethoxide in ethanol.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

