
Concept explainers
(a)
Interpretation:
An acceptable name for the following ether should be determined:
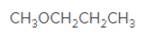
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the
(b)
Interpretation:
An acceptable name for the following ether should be determined:
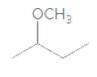
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the IUPAC nomenclature; one alkyl group is named as a hydrocarbon chain and the other part is named as a part of substituent bonded to that chain. The simpler alkyl group with the oxygen atom should name as an alkoxy substituent and the -yl suffix of the alkyl replaced with- oxy suffix. Remaining longest alkyl chain is named as an alkane by considering the alkoxy group as a substituent bonded to the main carbon chain.
(c)
Interpretation:
An acceptable name for the following ether should be determined:
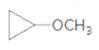
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the IUPAC nomenclature; one alkyl group is named as a hydrocarbon chain and the other part is named as a part of substituent bonded to that chain. The simpler alkyl group with the oxygen atom should name as an alkoxy substituent and the -yl suffix of the alkyl replaced with- oxy suffix. Remaining longest alkyl chain is named as an alkane by considering the alkoxy group as a substituent bonded to the main carbon chain.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, & Biological Chemistry
- SYNTHESIS REACTIONS. For the following reactions, synthesize the given products from the given reactants. Multiple reactions/steps will be needed. For the one of the steps (ie reactions) in each synthesis, write out the mechanism for that reaction and draw an energy diagram showing the correct number of hills and valleys for that step's mechanism. CI b. a. Use acetylene (ethyne) and any alkyl halide as your starting materials Br C. d. "OH OH III. OHarrow_forwardCalculate the pH and the pOH of each of the following solutions at 25 °C for which the substances ionize completely: (a) 0.200 M HClarrow_forwardCalculate the pH and the pOH of each of the following solutions at 25 °C for which the substances ionize completely: (a) 0.000259 M HClO4arrow_forward
- What is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forwardDetermine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. NaN₃arrow_forwardCan I please get help with this?arrow_forward
- Can I please get help with this?arrow_forwardUse the Henderson-Hasselbalch equation to calculate pH of a buffer containing 0.050M benzoic acidand 0.150M sodium benzoate. The Ka of benzoic acid is 6.5 x 10-5arrow_forwardA. Draw the structure of each of the following alcohols. Then draw and name the product you would expect to produce by the oxidation of each. a. 4-Methyl-2-heptanol b. 3,4-Dimethyl-1-pentanol c. 4-Ethyl-2-heptanol d. 5,7-Dichloro-3-heptanolarrow_forward
- What is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forwardCan I please get help with this.arrow_forwardDetermine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. N₂H₅ClO₄arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



