
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 13, Problem 47P
Interpretation Introduction
Interpretation:
Double bonds of the following cycloalkane should be labeled as cis or trans.
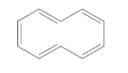
Concept Introduction:
Cis-trans isomers are fall under geometric isomerism.
Cis isomers- two different groups are on the same side of the double bond.
Trans isomers- two different groups are on opposite sides of the double bond.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Part A
Identify each alcohol as primary, secondary, or tertiary
Drag the appropriate items to their respective bins.
CH₂
H₂C-
-C-OH
HO
CH₂
Primary
Он
OH
CH₂
OH
CCH₂OH
CH₂
сн
Secondary
Tertiary
Reset Help
CH,CH₂
(CH)CHCH,OH CH,CH,CH,CCH,
CHOH
CH₂
Different types of alcohol groups
Alcohol and its reaction:
8. Combing two alcohol molecules below and completing the reaction with
Product .( Hint Reaction called etherification as ether is formed and name the
ether once you complete the reaction.
Hint.:
R-O-H+H-O-RR-O-R
Do the reaction:
CH₂OH + CH₂OH---→
+ H-O-H
9. Write the reaction of formation of alcohol from alkene by adding water:
Addition reaction also called hydration reaction as we are adding water
which occur always in presence of acid
Hint: Break the double bond and add H and OH if symmetrical then
add anywhere if unsymmetrical then follow Markovnikov rule H
should go to that double bone carbon which has more hydrogen
CH2=CH2 + H₂O-→
Complete the reaction
hand written please
Predict the major products of this organic reaction:
HBr (1 equiv)
cold
?
Some important notes:
• Draw the major product, or products, of this reaction in the drawing area below.
• You can draw the products in any arrangement you like.
• Pay careful attention to the reaction conditions, and only include the major products.
• Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers.
• Note that there is only 1 equivalent of HBr reactant, so you need not consider the case of multiple additions.
dm
Re
Explanation
Check
©2025 McGraw Hill LLC. All Rights Reserved. Term
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 13.1 - Convert each condensed structure to a complete...Ch. 13.1 - Prob. 13.1PCh. 13.1 - Complete the structure of zingiberene, a component...Ch. 13.2 - Prob. 13.2PPCh. 13.2 - Prob. 13.3PCh. 13.2 - Prob. 13.3PPCh. 13.2 - Prob. 13.4PPCh. 13.3 - Prob. 13.5PPCh. 13.3 - Prob. 13.4PCh. 13.3 - Prob. 13.6PP
Ch. 13.3 - Prob. 13.5PCh. 13.3 - Prob. 13.6PCh. 13.3 - Prob. 13.7PCh. 13.3 - Prob. 13.8PCh. 13.6 - Prob. 13.7PPCh. 13.6 - Prob. 13.9PCh. 13.6 - What products is formed in each of the following...Ch. 13.6 - Prob. 13.9PPCh. 13.6 - What product is formed when l-pentene (CH3CH2CH2CH...Ch. 13.7 - Prob. 13.11PCh. 13.8 - Prob. 13.10PPCh. 13.8 - Prob. 13.12PCh. 13.8 - Prob. 13.13PCh. 13.10 - Prob. 13.11PPCh. 13.10 - Prob. 13.14PCh. 13.11 - Prob. 13.15PCh. 13.12 - Prob. 13.16PCh. 13.13 - Prob. 13.17PCh. 13.13 - Prob. 13.18PCh. 13 - Anethole, the major constituent of anise oil, is...Ch. 13 - Prob. 20PCh. 13 - What is the molecular formula for a hydrocarbon...Ch. 13 - Prob. 22PCh. 13 - Prob. 23PCh. 13 - Prob. 24PCh. 13 - Prob. 25PCh. 13 - Prob. 26PCh. 13 - Give the IUPAC name for each molecule depicted in...Ch. 13 - Give the IUPAC name for each molecule depicted in...Ch. 13 - Give the IUPAC name for each compound. a....Ch. 13 - Give the IUPAC name for each compound. d....Ch. 13 - Give the IUPAC name for each alkene.Ch. 13 - Give the I UP AC name for each alkene.Ch. 13 - Give the IUPAC name for each cyclic compound.Ch. 13 - Give the IUPAC name for each cyclic compound.Ch. 13 - Give the structure corresponding to each IUPAC...Ch. 13 - Prob. 36PCh. 13 - Each of the following IUPAC names is incorrect....Ch. 13 - Each of the following IUPAC names is incorrect....Ch. 13 - Prob. 39PCh. 13 - Prob. 40PCh. 13 - Prob. 41PCh. 13 - Prob. 42PCh. 13 - Prob. 43PCh. 13 - Label the carbon-carbon double bond as cis or...Ch. 13 - Prob. 45PCh. 13 - Prob. 46PCh. 13 - Prob. 47PCh. 13 - Prob. 48PCh. 13 - Prob. 49PCh. 13 - Prob. 50PCh. 13 - Prob. 51PCh. 13 - Prob. 52PCh. 13 - Prob. 53PCh. 13 - Prob. 54PCh. 13 - What alkyd halide is formed when each alkene is...Ch. 13 - Prob. 56PCh. 13 - Prob. 57PCh. 13 - Prob. 58PCh. 13 - What alkene is needed as a starting material to...Ch. 13 - Prob. 60PCh. 13 - Prob. 61PCh. 13 - Prob. 62PCh. 13 - Prob. 63PCh. 13 - Prob. 64PCh. 13 - Prob. 65PCh. 13 - Prob. 66PCh. 13 - Prob. 67PCh. 13 - Prob. 68PCh. 13 - Prob. 69PCh. 13 - Prob. 70PCh. 13 - Prob. 71PCh. 13 - Prob. 72PCh. 13 - Prob. 73PCh. 13 - Are o-bromochlorobenzene and m-bromochlorobenzene...Ch. 13 - Give the structure corresponding to each IUPAC...Ch. 13 - Give the structure corresponding to each IUPAC...Ch. 13 - Prob. 77PCh. 13 - Prob. 78PCh. 13 - Prob. 79PCh. 13 - Prob. 80PCh. 13 - Prob. 81PCh. 13 - Prob. 82PCh. 13 - Prob. 83PCh. 13 - Prob. 84PCh. 13 - Prob. 85PCh. 13 - Eleostearic acid is an unsaturated fatty acid...Ch. 13 - Prob. 87PCh. 13 - Prob. 88PCh. 13 - Prob. 89PCh. 13 - Prob. 90PCh. 13 - Prob. 91PCh. 13 - Prob. 92PCh. 13 - Prob. 93PCh. 13 - Prob. 94PCh. 13 - Answer the following questions about compound A,...Ch. 13 - Prob. 96PCh. 13 - Answer the following questions about alkene C,...Ch. 13 - Prob. 98PCh. 13 - Prob. 99PCh. 13 - Prob. 100PCh. 13 - Prob. 101CPCh. 13 - Prob. 102CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- b) Use curved arrows to show the reaction of the radical with hydrogen bromide. Br: Br H .. Answer Bankarrow_forwardIndicate the reaction products when CH3COCH2COOCH2COOC2H5 (ethyl acetoacetoacetate) reacts with 1º OH-/H2O and 2º H3O+arrow_forwardDraw the formula of the compound 4-cyclohexyl butanamide?arrow_forward
- What is the formula of the compound 3-isopropylcyclopentane-1-carbonyl chloride?arrow_forwardIndicate the products of the reaction between CH3COCH2COONa (Sodium acetoacetate) and BrCH2COOC2H5arrow_forwardIndicate whether the product of the reaction between Naphthalene and CrO3 in acetic acid at 25ºC is 1,4 naphthoquinone or phthalic anhydride.arrow_forward
- Indicate the products of the reaction between CH3COCH2COOC2H5 and Na+-OC2H5.arrow_forwardPrimary, Secondary, and Tertiary Alcohols O-H O-H O-H R₁-C-H R₁-C-H R₁-C-R₁ H R₂ R₂ Primary Alcohol Secondary Alcohol ChemistryLearner.com R stands for Carbon group like ethyl methyl propyl Tertiary Alcohol If 1 carbon group with two H attached to alcoholic carbon, then primary If 2 carbon group and 1 H are attached to alcoholic carbon, then secondary IF 3 carbon group and no H attach to alcoholic carbon then tertiary. The bottom line Starting "Weak" oxidant material PCC, DMP, Swern, etc Primary alcohol Aldehyde OH Secondary alcohol Ketone OH "Strong" oxidant KMnO4, H₂CrO4 (or equivalent) OH Carboxylic acid 요 Ketone No reaction No reaction Tertiary alcohol 1. Is ethanol a primary, secondary, or tertiary alcohol? Write out the structures of ethanol and any oxidation products of ethanol. If there is more than one oxidation product, give the structure of each of the products. 2. Is 2-propanol a primary, secondary, or tertiary alcohol? Write out the structures of 2-propanol and any…arrow_forwardFormulate the reaction: Naphthalene with CrO3 in acetic acid at 25ºCarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
