
Concept explainers
(a)
Interpretation: To indicate whether the statement “
Concept introduction: Glucose is a monosaccharide with the molecular formula
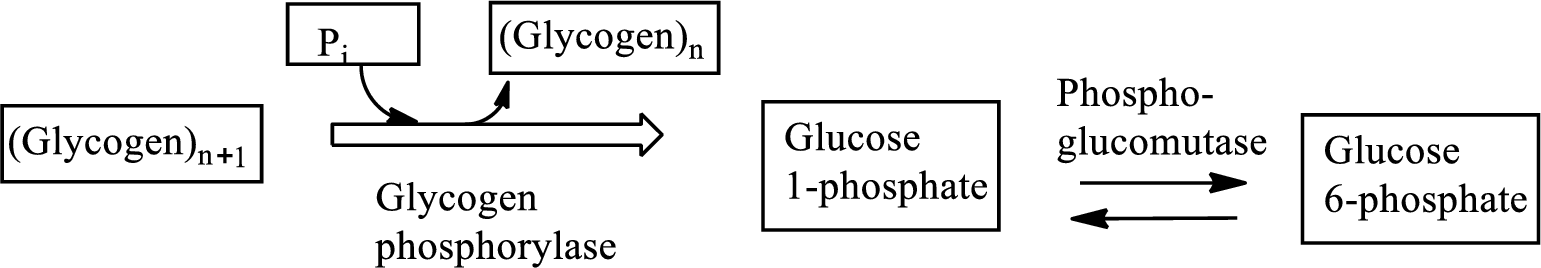
Glycogenolysis is the
An intermediate is defined as the transient species that is formed from the reactants in the preceding step and gets consumed in the subsequent steps to generate the products. An intermediate is formed within a multi-step reaction mechanism.
In the isomerization reaction, a molecule transformed itself to another molecule, having the same number of atoms with a different arrangement.
(b)
Interpretation: To indicate whether the statement “an isomerization reaction changes
Concept introduction: Glucose is a monosaccharide with the molecular formula
Glycogenolysis is the metabolic pathway that converts glycogen to
In the isomerization reaction, a molecule transformed itself to another molecule, having the same number of atoms with a different arrangement.
(c)
Interpretation: To indicate whether the statement “an ATP molecule is used to activate a
Concept introduction: Glucose is a monosaccharide with the molecular formula
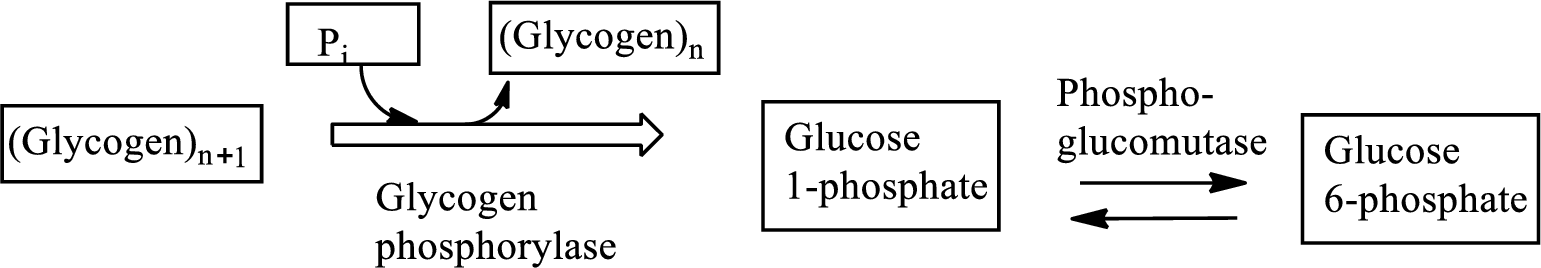
Glycogenolysis is the metabolic pathway that converts glycogen to
Adenosine triphosphate (ATP) is the energy currency of life that provides energy to carry out the metabolic processes in the living cells.
(d)a
Interpretation: To indicate whether the statement “the equivalent of two ATP molecules are consumed” relating to glycogenolysis is true or false.
Concept introduction: Glucose is a monosaccharide with the molecular formula
Glycogenolysis is the metabolic pathway that converts glycogen to
Adenosine triphosphate (ATP) is the energy currency of life that provides energy to carry out the metabolic processes in the living cells.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Organic And Biological Chemistry
- Indicate the product formed in each reaction. If the product exhibits tautomerism, draw the tautomeric structure. a) о + CH3-NH-NH2 CO2C2H5 b) + CoH5-NH-NH2 OC2H5arrow_forwardIndicate the formula of the compound, that is the result of the N- alquilación (nucleofílic substitution), in which an additional lateral chain was formed (NH-CH2-COOMe). F3C. CF3 NH NH2 Br о OMe K2CO3, DABCO, DMFarrow_forwardSynthesis of 1-metilbenzotriazole from 1,2-diaminobenceno.arrow_forward
- Synthesis of 1-metilbenzotriazole.arrow_forwardIndicate the formula of the compound, that is the result of the N- alquilación (nucleofílic substitution), in which an additional lateral chain was formed (NH-CH2-COOMe). F3C. CF3 NH NH2 Br о OMe K2CO3, DABCO, DMFarrow_forwardIdentify the mechanism through which the following reaction will proceed and draw the major product. Part 1 of 2 Br KOH EtOH Through which mechanism will the reaction proceed? Select the single best answer. E1 E2 neither Part: 1/2 Part 2 of 2 Draw the major product formed as a result of the reaction. Click and drag to start drawing a structure. Xarrow_forward
- What is single-point calibration? Provide an example.arrow_forwardDraw the major product formed via an E1 pathway.arrow_forwardPart 9 of 9 Consider the products for the reaction. Identify the major and minor products. HO Cl The E stereoisomer is the major product and the Z stereoisomer is the minor product ▼ S major product minor productarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning
Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning




