
Introduction to General, Organic and Biochemistry
11th Edition
ISBN: 9781285869759
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.32P
13*32 Black-and-white photography is a commercial process that involves a phenol. Black-and-white film is coated with an emulsion containing silver bromide or silver iodide crystals that become activated by exposure to light. The activated silver ions then react with hydroquinone in the developing stage as shown in the following balanced equation.
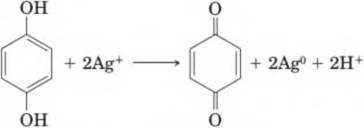
All silver halide not activated by light is removed in the fixing process, and the result is a black image (a negative) left by the deposited metallic silver where the film had been struck by fight. In this
- What is reduced and what is the reducing agent?
- What is oxidized and what is the oxidizing agent?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
None
In the solid state, oxalic acid occurs as
a dihydrate with the formula H2C2O4
C+2H2O. Use this formula to
calculate the formula weight of oxalic
acid. Use the calculated formula
weight and the number of moles
(0.00504mol)
of oxalic acid in each titrated
unknown sample recorded in Table
6.4 to calculate the number of grams
of pure oxalic acid dihydrate
contained in each titrated unknown
sample.
1.
Consider a pair of elements with 2p and 4p valence orbitals (e.g., N and Se). Draw their
(2p and 4p AO's) radial probability plots, and sketch their angular profiles. Then, consider these
orbitals from the two atoms forming a homonuclear л-bond. Which element would have a
stronger bond, and why?
(4 points)
Chapter 13 Solutions
Introduction to General, Organic and Biochemistry
Ch. 13.2 - Prob. 13.1PCh. 13 - Answer true or false. Alkenes, alkynes, and arenes...Ch. 13 - 13-3 What is the difference in structure between a...Ch. 13 - 13-4 Define aromatic compound.Ch. 13 - 13-5 Why are alkenes, alkynes, and aromatic...Ch. 13 - 13-B Do aromatic rings have double bonds? Are they...Ch. 13 - 13-7 Can an aromatic compound be a saturated...Ch. 13 - Draw at least two structural formulas for each of...Ch. 13 - 13-9 Write a structural formula and the name for...Ch. 13 - 13-10 Account for the fact that the six-membered...
Ch. 13 - 13-11 Explain why the compound 1,4-dichlorobenzene...Ch. 13 - 13-12 One analogy often used to explain the...Ch. 13 - 13-13 Answer true or false. A phenyl group has the...Ch. 13 - Prob. 13.14PCh. 13 - 13-15 Draw structural formulas for these compounds...Ch. 13 - 13-16 We say that naphthalene, anthracene,...Ch. 13 - 13-17 Following is the structural formula of...Ch. 13 - 13-18 Answer true or false. Benzene does not...Ch. 13 - 13-19 Suppose you have unlabeled bottles of...Ch. 13 - 13-20 Three products with the molecular formula...Ch. 13 - 13-21 The reaction of bromine with toluene in the...Ch. 13 - 13-22 What reagents and/or catalysts are necessary...Ch. 13 - 13-23 What reagents and/or catalysts are necessary...Ch. 13 - Prob. 13.24PCh. 13 - 13-25 Answer true or false. (a) Phenols and...Ch. 13 - 13-26 Both phenol and cyclohexanol are only...Ch. 13 - 13-27 Define autoxidation.Ch. 13 - 13*28 Autoxidation is described as a radical-chain...Ch. 13 - 13-29 Show that if you add Steps 2a and 2b of the...Ch. 13 - 13-30 How does vitamin E function as an...Ch. 13 - 13-31 What structural features are common to...Ch. 13 - 13*32 Black-and-white photography is a commercial...Ch. 13 - 13-33 Following is the structural formula of...Ch. 13 - 13-34 (Chemical Connections 13A) From what parts...Ch. 13 - Prob. 13.35PCh. 13 - 13-36 (Chemical Connections 13A, Would you expect...Ch. 13 - Prob. 13.37PCh. 13 - 13-38 (Chemical Connections 13A) What is meant by...Ch. 13 - 13-39 (Chemical Connections 13B) What is a...Ch. 13 - 13-40 (Chemical Connections 130 In the absence of...Ch. 13 - Prob. 13.41PCh. 13 - 13-42 (Chemical Connections 13E) What are the...Ch. 13 - 13-43 (Chemical Connections 13E) Which features of...Ch. 13 - 13-44 (Chemical Connections 13E) What color would...Ch. 13 - Prob. 13.45PCh. 13 - Prob. 13.46PCh. 13 - Prob. 13.47PCh. 13 - 13-48 (Chemical Connections 13F, How many...Ch. 13 - 13-49 (Chemical Connections 13F) In what ways is...Ch. 13 - 13*50 The structure for naphthalene given in...Ch. 13 - 13-51 Draw structural formulas for these...Ch. 13 - 13-52 2,6-Di-/ezY-butyl-4-methylphenol (BHT,...Ch. 13 - 13-53 Write the structural formula for the product...Ch. 13 - 13-54 Styrene reacts with bromine to give a...Ch. 13 - 13-55 When toluene is treated with Br, in the...Ch. 13 - 13-56 Four alternatives to the structure of...Ch. 13 - 13-57 Benzene, as we have seen in this chapter, is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the reaction and show the mechanism of the reaction. Include the mechanism for formation of the NO2+ 2. Explain, using resonance structures, why the meta isomer is formed. Draw possible resonance structures for ortho, meta and para.arrow_forwardNonearrow_forward3. A molecular form of "dicarbon", C2, can be generated in gas phase. Its bond dissociation energy has been determined at 599 kJ/mol. Use molecular orbital theory to explain why energy of dissociation for C₂+ is 513 kJ/mol, and that for C2² is 818 kJ/mol. (10 points)arrow_forward
- 9.73 g of lead(IV) chloride contains enough Cl- ions to make ____ g of magnesium chloride.arrow_forward6. a) C2's. Phosphorus pentafluoride PF5 belongs to D3h symmetry group. Draw the structure of the molecule, identify principal axis of rotation and perpendicular (4 points) b) assume that the principal axis of rotation is aligned with z axis, assign symmetry labels (such as a1, b2, etc.) to the following atomic orbitals of the P atom. (character table for this group is included in the Supplemental material). 3s 3pz (6 points) 3dz²arrow_forward2. Construct Lewis-dot structures, and draw VESPR models for the ions listed below. a) SiF5 (4 points) b) IOF4 (4 points)arrow_forward
- 5. Complex anion [AuCl2]¯ belongs to Doh symmetry point group. What is the shape of this ion? (4 points)arrow_forward4. Assign the following molecules to proper point groups: Pyridine N 1,3,5-triazine N Narrow_forward7. a) Under normal conditions (room temperature & atmospheric pressure) potassium assumes bcc lattice. Atomic radius for 12-coordinate K atom is listed as 235 pm. What is the radius of potassium atom under normal conditions? (3 points) b) Titanium metal crystallyzes in hcp lattice. Under proper conditions nitrogen can be absorbed into the lattice of titanium resulting in an alloy of stoichiometry TiNo.2. Is this compound likely to be a substitutional or an interstitial alloy? (Radius of Ti (12-coordinate) is 147 pm; radius of N atom is 75 pm. (3 points)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

ENVIRONMENTAL POLLUTION; Author: 7activestudio;https://www.youtube.com/watch?v=oxtMFmDTv3Q;License: Standard YouTube License, CC-BY